Lewis Dot Formula Unit & Naming Practice Sheet

Hey there, future chemistry rockstar! Ever feel like atoms are just tiny, mysterious blobs? Well, buckle up, buttercup, because we're about to unlock some serious atomic secrets. And it all starts with these little dudes called Lewis Dot Formulas. Think of them as the atom's bling. Seriously!
So, what's the big deal? Lewis Dot Formulas are basically a super-simple way to show how atoms like to party. They focus on the outermost electrons, the ones that actually get invited to the atomic dance. These are called valence electrons. Each dot you see around an atom's symbol? That's a valence electron. Easy peasy, right?
The Dot-tastic World of Atoms
Imagine atoms as introverts or extroverts. Some are perfectly happy chilling with their current electron situation. Others? They're totally craving more friends to feel complete. Lewis Dots help us see who's feeling the electron FOMO (Fear Of Missing Out).
We're talking about elements like carbon, oxygen, nitrogen – the usual suspects in the chemistry party scene. They all have different numbers of valence electrons, and that dictates how they'll bond. It's like each element has its own unique accessory count. Gold chains for some, friendship bracelets for others.
And get this: drawing these is surprisingly therapeutic. It's like a tiny, scientific coloring book. Plus, understanding them is the key to knowing how molecules are actually built. Think of it as learning the alphabet before you can write a novel. Except, you know, a novel about explosions and weird goo.
Why All the Dots? It's About Stability, Dudes!
Atoms are basically chasing a dream: a full outer shell. They want eight valence electrons (except for hydrogen and helium, they're rebels and settle for two). Why eight? It's like a magic number for atomic happiness. When an atom has a full outer shell, it's super stable. It’s not looking to make any new friends, or break up with its current ones. It's content. It's done. It's the atomic equivalent of a cozy blanket and a good book.

So, how do they get these magic eight? They share! They loan! They even steal (but we call that ionic bonding, which is a whole other juicy topic for another day). Lewis Dots visually show you these electron arrangements. You can see a lone electron just hanging out, or a pair snuggled up together. It's like peeking into their personal electron lives.
This is where the fun really kicks in with a Lewis Dot Formula Unit & Naming Practice Sheet. It’s your personal playground to get hands-on with these concepts. You’re not just reading about atoms anymore; you’re drawing them. You’re predicting how they’ll behave. It’s like being a tiny chemist inventor!
From Dots to Names: The Grand Unveiling
But wait, there's more! Once you've mastered drawing those little electron dots, you can take it a step further. You can actually start naming the molecules you create. Yep, those weird, long names you see on chemical bottles? They have a logic! And Lewis Dots are a stepping stone to understanding that logic.

The way atoms bond, the number of bonds they form – it all influences the final name. It's like a secret code. You crack the Lewis Dot code, and suddenly you can decipher the chemical language. Imagine the power! You could be the person at parties who casually explains why water is H₂O and not, say, O₂H. Mind. Blown.
A good practice sheet will walk you through this. It’ll give you an atom, you draw its dots. Then it might give you a couple of atoms, and you figure out how they’ll bond using those dots. Finally, it’ll reveal the name, and you’ll be like, "Aha! I see it now!" It’s a beautiful moment of scientific revelation.
Quirky Facts and Fun Stuff You Didn't Know You Needed
Did you know that the element with the most Lewis dots is often the life of the party? Looking at you, Group 18 noble gases! They’re already chilling with a full house, so they're super unreactive. They're the ultimate introverts of the element world. They just don't need any more electron friends.

And then there's hydrogen. It's like the eager intern. It only needs one more electron to feel complete. It's always looking to make one quick bond. So humble, so efficient. It’s the tiny car of the element world, getting the job done with minimal fuss.
The concept of Lewis dots was first introduced by Gilbert N. Lewis in 1916. Can you imagine trying to explain electrons back then? It must have felt like teaching people how to use a smartphone before the internet existed. Revolutionary stuff!
Seriously, there’s a whole universe of tiny structures and interactions happening all around us, all the time. And Lewis Dots are your ticket to getting a glimpse. They’re not just for chemists in lab coats; they're for anyone who’s ever wondered what stuff is actually made of.
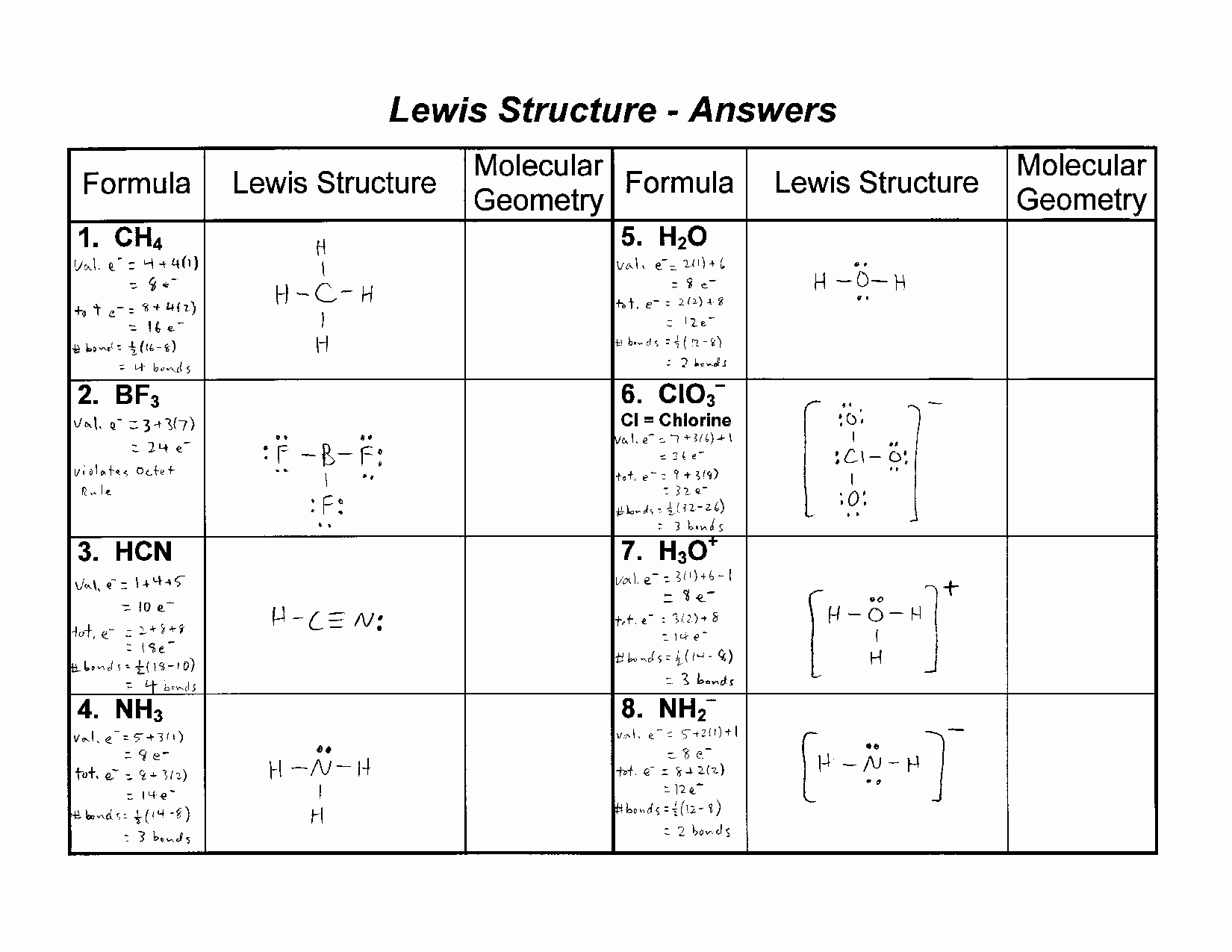
Why This Practice Sheet is Your New Best Friend
Think of this practice sheet as your cheat sheet to the atomic underworld. It’s a safe space to mess up, to doodle, to draw electron pairs that look more like chaotic spaghetti than neat dots. Nobody's judging! That's how you learn.
You'll get to practice drawing dots for all sorts of elements. You'll see how oxygen loves to grab two electrons. You'll see how chlorine is always on the lookout for one more electron to complete its party of eight. It’s like a dating app for atoms, and you’re the matchmaker.
And when you move on to naming, you’ll realize it’s not arbitrary. There are rules! There are patterns! It’s like a puzzle, and each correctly named molecule is a piece that clicks into place.
So, if you're looking for a way to make chemistry a little less intimidating and a lot more fun, grab yourself a Lewis Dot Formula Unit & Naming Practice Sheet. It’s your first step into understanding the building blocks of everything. And who knows, you might just discover you’re a chemistry whiz in the making. Now go forth and dot!
