Lead Nitrate Sodium Sulfide Molecular Equation

Ever feel like you're just mixing random things together in life and hoping for the best? Like when you’re trying to whip up a gourmet meal with only a can of beans and a dream, or attempting to assemble IKEA furniture without looking at the instructions? Well, believe it or not, chemistry has its own version of those delightful kitchen (or workshop) disasters. Today, we're diving into a little chemical shindig involving lead nitrate and sodium sulfide. Don't let the fancy names scare you; it's all about what happens when these two meet up.
Think of it like this: you've got your best friend, let’s call her Lead Nitrate. She’s a bit of a drama queen, always looking to pair up with someone new. And then there’s Sodium Sulfide, who's kind of the quiet, steady type, but secretly a bit of a show-off when the right ingredients come along. They’re both dissolved in water, which is like them being at the same big, slightly crowded party. Water is the ultimate social butterfly of the chemical world, letting everyone mingle and get to know each other.
Now, when Lead Nitrate and Sodium Sulfide are in the same watery bash, things get interesting. They start eyeing each other up, and it turns out they’re a much better match with different partners. It’s like when you go to a party and suddenly realize your friend looks way better with that person across the room than with the one they dragged along. Chemistry’s got its own version of awkward matchmaking.
So, Lead Nitrate, being the energetic one, decides to ditch her original buddies (nitrate ions, which are totally chill and just float around) and pair up with Sulfide. And guess what? They form this unholy union called lead sulfide. Now, lead sulfide is not like your average party guest who just mingles. Oh no. It’s the kind of guest that shows up in a blindingly white, pristine outfit and then immediately decides to just sit down and not move. It solidifies. It precipitates. It basically makes a statement by becoming a solid, insoluble blob.
Imagine trying to dance at a party and suddenly your shoes get glued to the floor. That’s kind of what lead sulfide does. It’s so not into the whole dissolving-and-mingling thing that it just clumps together, forming a thick, inky black precipitate. Think of it as the ultimate party pooper, but in a visually dramatic way. It’s the visual equivalent of someone dropping a mic and storming off stage. Except, in this case, it's dropping into the solution and forming a solid.
And what about Sodium Sulfide’s original partner, Sodium? Well, it’s now free and single again, and it spots Lead Nitrate’s old pal, the nitrate ion. They get together and form sodium nitrate. Now, sodium nitrate is the opposite of lead sulfide. It’s like the life of the party, the one who’s totally fine with dancing all night, mingling with everyone, and generally being a good sport. It stays dissolved in the water, happily splashing around and not causing any fuss. It’s the friend who’s always up for another round of karaoke.
So, in the grand scheme of things, when you mix a solution of lead nitrate with a solution of sodium sulfide, you end up with a visual spectacle. You get this dramatic, inky black cloud forming at the bottom of your beaker, and the rest of the liquid stays clear. It's like watching a magic trick where a dark shadow suddenly appears out of nowhere.
The molecular equation is just the fancy way scientists write down this whole drama. It's like the script for our chemical play. It tells you exactly what molecules went in and what molecules came out. It’s not just a jumble of letters and numbers; it’s a precise record of the reactants and products.
The Molecular Lowdown
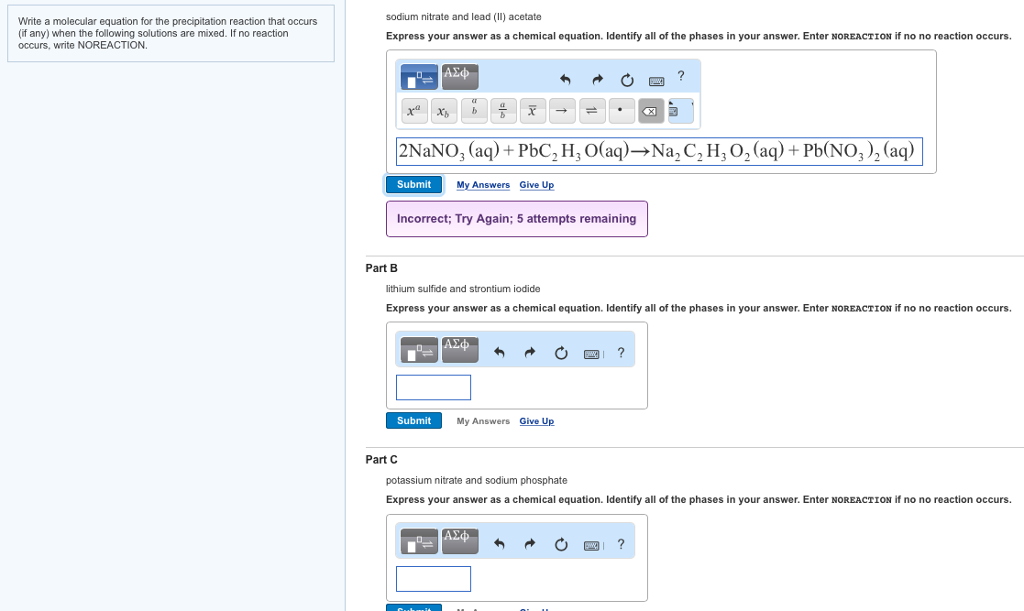
Let's break down the official jargon, shall we? The molecular equation for this whole kerfuffle looks something like this:
Pb(NO3)2 (aq) + Na2S (aq) → PbS (s) + 2NaNO3 (aq)
Don’t freak out. We’re going to translate this into plain English, with a few helpful analogies thrown in.
On the left side of the arrow (that’s the "reactants" side, the stuff you start with, like your ingredients before you bake a cake), we have Pb(NO3)2 (aq). This is our Lead Nitrate. The "Pb" is for lead, and the "NO3" is the nitrate. The "(aq)" just means it's dissolved in water, so it's all watery and ready to mingle. Think of it as a bottle of really fancy, clear liquid that you bought at the store.
Next to it, we have Na2S (aq). This is our Sodium Sulfide. "Na" is for sodium (think of that salty stuff you put on your fries), and "S" is for sulfur. Again, "(aq)" means it’s dissolved in water. This is like another bottle of clear liquid, maybe a little less fancy, but equally ready for action.

So, you’ve got two clear liquids, right? You pour them into the same container, and BAM!
The Grand Reveal
On the right side of the arrow (that’s the "products" side, what you end up with, like the delicious (or questionable) cake you baked), we have PbS (s). This is our dramatic Lead Sulfide. "Pb" for lead and "S" for sulfur. The crucial part here is the "(s)". This little "s" stands for "solid". This is the inky black stuff that sinks to the bottom. It’s the precipitate. It’s the reason your clear liquid is no longer clear. It's the thing that makes you go, "Whoa, what just happened?"
And then, we have 2NaNO3 (aq). These are our Sodium Nitrate pals. The "2" in front? That's just there to make sure everything balances out. It's like making sure you have the right number of dance partners so no one is left standing awkwardly by the punch bowl. The "Na" is sodium, and "NO3" is nitrate. And the "(aq)" means this stuff is still dissolved in water, happily floating around, having a great time. It’s the clear part of the liquid that remains.
So, in essence, the lead and sulfide ions, who were previously sort of "paired up" with their nitrate and sodium friends, decided to swap partners. Lead grabbed onto Sulfide, and they formed this solid, insoluble compound, lead sulfide. Meanwhile, Sodium happily hooked up with the leftover nitrate ions and they stayed dissolved as sodium nitrate. It’s a classic case of chemical partner-swapping, with one couple forming a solid, dark precipitate and the other remaining happily in solution.
Why does this happen? Well, lead sulfide is like that one friend who absolutely loathes being in a crowd of strangers. It prefers to stick with its own kind, and when it finds its chemical soulmate (sulfide, in this case), it forms a very strong bond that makes it want to crystallize out of the solution. It’s like it’s saying, "Nope, not mingling anymore. We're doing our own thing, separately."
Sodium nitrate, on the other hand, is the social butterfly. It’s perfectly happy to stay dissolved, interacting with the water molecules and not forming any solid structures. It’s the kind of compound that’s always like, "Hey, let's keep this party going!"
Everyday Analogies for the Win
Think about making a really strong cup of coffee. You add coffee grounds (the solids) to hot water. The water (our solvent) extracts the tasty stuff from the coffee grounds. In our chemical reaction, the lead sulfide is like the coffee grounds that don't dissolve; they just sink to the bottom, leaving the clear liquid behind. The sodium nitrate is like the dissolved flavor compounds in your coffee that make it taste good.
Or, consider making a batch of homemade ice cream. You mix all your yummy ingredients together (reactants). When you freeze it, some things become solid, like the ice crystals, while others remain mixed in the creamy liquid. Lead sulfide is the ice crystal that solidifies and stands out, while sodium nitrate is the smooth, dissolved creaminess.
It’s also a bit like when you’re sorting through a pile of Lego bricks. You’ve got red bricks and blue bricks all mixed up. Then you decide to sort them, and you put all the red ones together and all the blue ones together. Lead sulfide is like a clump of all the red bricks that stick together so strongly that they form a solid block, while sodium nitrate is like all the blue bricks that are now neatly sorted but still loose and ready to be used for something else.
The funny thing is, sometimes these reactions are used in practical ways. For instance, lead sulfide is a very dark pigment. So, in the olden days, chemists might have used this reaction to create black paint or dyes. Imagine a chemist in a dusty old lab, carefully mixing these liquids, and a cloud of blackness forms. It's like they're brewing up a potion!

And sodium nitrate? Well, that has its own uses, too. It's used in fertilizers, making plants grow big and strong. So, from a dramatic black precipitate to a plant-growing booster, this simple reaction is quite the multitasker!
The whole point of writing out that molecular equation is to have a clear, concise way to describe these transformations. It’s like a chemical shorthand. If you were trying to tell someone how to make lead sulfide, you wouldn’t just say, "Mix the stuff from that bottle with the stuff from the other bottle and see what happens." You’d give them the precise recipe, just like the molecular equation does.
It’s also important for making sure the reaction actually works. Chemists need to know the exact ratios of the ingredients to get the desired result. If you put in too much lead nitrate and not enough sodium sulfide, you might end up with some leftover lead nitrate floating around, which, let's be honest, is probably not ideal. It’s like trying to bake a cake with too much flour and not enough eggs – it might turn out… interesting.
So, next time you hear about a chemical reaction, don't just picture confusing formulas. Think about the everyday scenarios it can be compared to: the messy kitchen, the party, the sorting of toys. Chemistry is happening all around us, even when we're not in a lab coat. And sometimes, it’s just about a few dissolved substances deciding to swap partners and make a dramatic, solid exit. It’s the little dramas of the molecular world, playing out in a beaker near you!
It's pretty neat, right? These invisible particles are constantly interacting, forming new things, and driving the world around us. And the molecular equation is our way of understanding and documenting these tiny, but mighty, transformations. It's the chemical gossip column, if you will, telling us who's pairing up with whom and what the outcome is. And in the case of lead nitrate and sodium sulfide, the outcome is a visually striking, inky black precipitate that reminds us that even simple mixing can lead to something quite dramatic.
