Lead Ii Nitrate Sodium Chloride Balanced Equation
Hey there, science adventurers! Ever looked at a bunch of seemingly random chemicals and thought, "What's the big deal?" Well, get ready to have your mind gently nudged open because we're diving into something that might sound a tad… unglamorous at first glance: the balanced equation for Lead(II) Nitrate and Sodium Chloride. Sounds like a mouthful, right? But stick with me, because this little dance of molecules is actually a tiny spark of wonder that can add a dash of fun to your day!
Let's break it down, shall we? We've got Lead(II) Nitrate. Think of it as a fancy salt, but with a lead atom chilling at its core. And then there's Sodium Chloride. Yep, you guessed it – that's your everyday table salt! Who knew such common ingredients could team up for a chemical party?
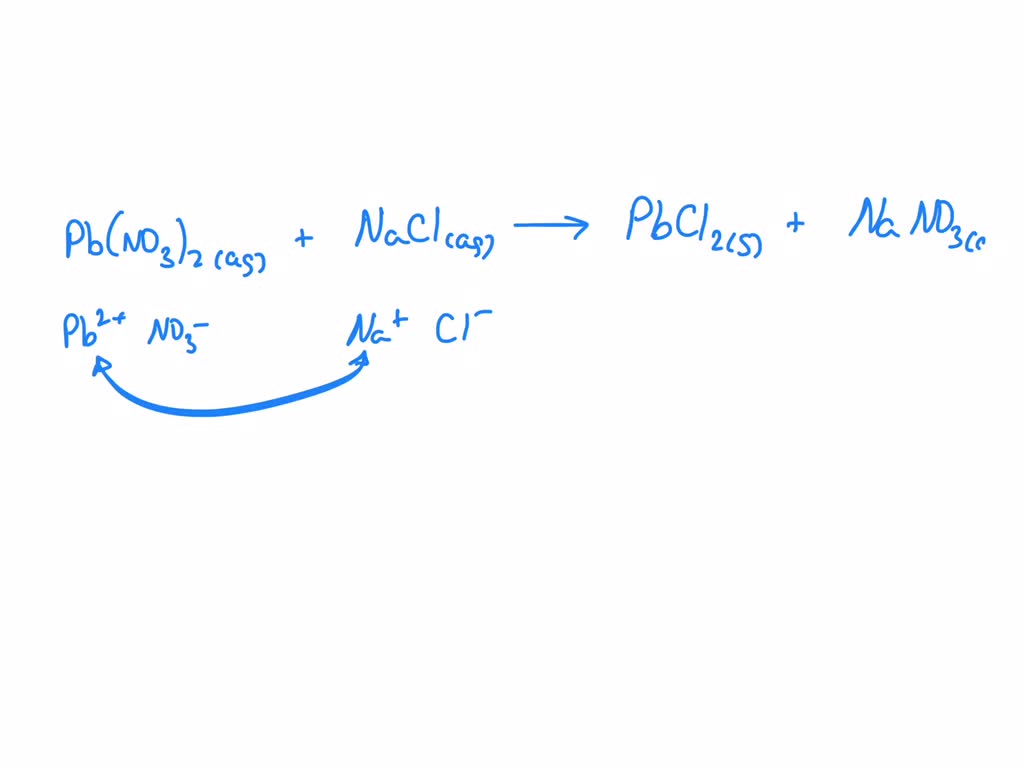
Now, what happens when these two get together? It's like a matchmaking service for ions! Lead(II) Nitrate, when dissolved in water (because most of these reactions happen in solution, like a tiny, invisible swimming pool for molecules), splits into its components: lead ions (Pb²⁺) and nitrate ions (NO₃⁻). Similarly, Sodium Chloride, also in water, breaks into sodium ions (Na⁺) and chloride ions (Cl⁻).
So, you've got these four types of ions floating around. They're all looking for a partner! And guess what? They're pretty particular. It turns out that lead ions and chloride ions have a major crush on each other. They just can't resist forming a really strong bond. We call this a precipitate – basically, they decide to ditch the water party and form a solid together.
Meanwhile, the sodium ions and nitrate ions are like, "Eh, we're okay together, but not that into it." They prefer to stay dissolved and keep swimming around. It's like when you go to a party and some people hit it off instantly, forming a tight-knit group, while others are happy to mingle solo. Chemistry, am I right?
The "balanced equation" part? That's just the chemist's way of keeping score. It's like saying, "Okay, for every one of this thing we start with, we're going to end up with exactly that much of that other thing." Conservation of matter, people! It's the universe's way of saying, "You can't create or destroy stuff, you can only rearrange it." Pretty neat, huh?

So, the equation for our little lead and salt shindig looks like this:
Pb(NO₃)₂(aq) + 2NaCl(aq) → PbCl₂(s) + 2NaNO₃(aq)
Let's decode this masterpiece.

Pb(NO₃)₂ is our Lead(II) Nitrate. The (aq) after it means it's dissolved in aqueous solution – fancy talk for water.
2NaCl is our Sodium Chloride. Again, (aq) means it's dissolved in water. Notice that little '2' in front? That's important! It tells us we need two units of sodium chloride to balance out the lead nitrate. It's like having a plus-one to the party!
The arrow (→) means "reacts to form" or "yields." It's the point of transformation.
On the other side, PbCl₂ is our solid precipitate. See the '(s)'? That means solid! This is the cool part where you'd actually see something form – a cloudy white solid, like magic!
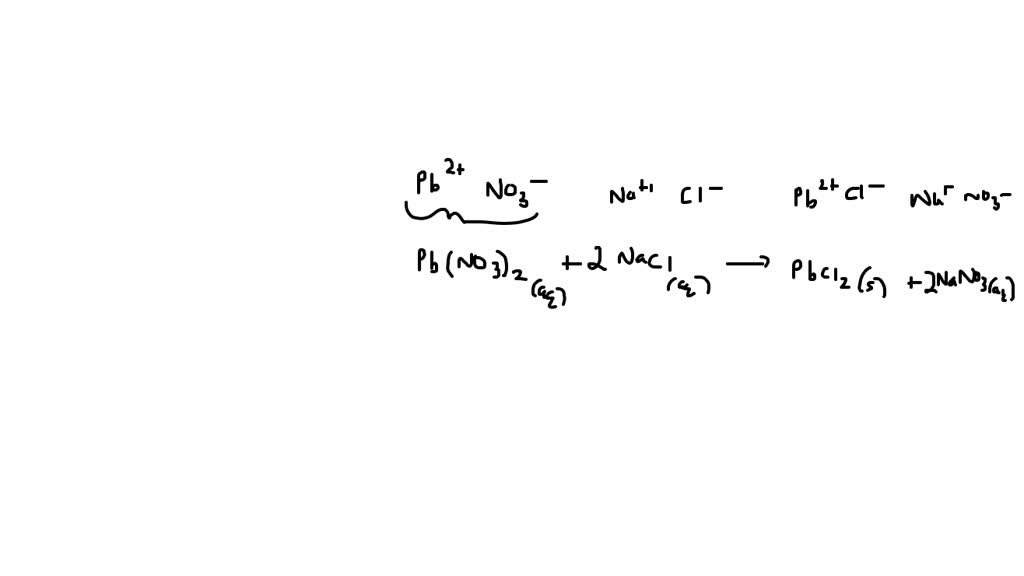
And then we have 2NaNO₃, our dissolved sodium nitrate. The '(aq)' tells us it's still floating around in the water, perfectly happy.
Why is this fun? Well, first off, it's like solving a tiny puzzle! You're understanding how the world around us works at a fundamental level. And when you can predict what will happen when you mix things, that's pretty darn powerful. It’s the foundation for so many amazing things we use every day, from medicines to materials.
Imagine you're a budding alchemist, or maybe a detective of the molecular world. You're given these two clear liquids, and you know that if you mix them in the right proportions, something visible will happen. That's not just chemistry; that's a miniature science show happening right before your eyes! It’s a chance to witness the unseen forces that shape our reality.

And think about the possibilities! While this specific reaction might not be for your kitchen experiments (lead is a no-go, folks!), the principle of balanced equations applies to countless safe and fascinating reactions. Understanding these basic building blocks can unlock a whole universe of creative potential. You could be designing new materials, concocting amazing dyes, or even understanding how life itself functions.
It’s also a fantastic way to build your problem-solving skills. Learning to balance equations hones your ability to think logically and systematically. It’s like a mental workout that makes you sharper and more capable in all sorts of areas, not just science.
So, the next time you hear about a chemical equation, don't shy away. Embrace it! Think of it as a secret code waiting to be cracked, a tiny window into the incredible order and beauty of the universe. Every balanced equation is a testament to the consistent and predictable nature of matter, a whisper of the grand design.
This seemingly simple equation between Lead(II) Nitrate and Sodium Chloride is just the tip of the iceberg. It’s an invitation to explore further, to ask more questions, and to discover the endless wonders of chemistry. Who knows what exciting discoveries await you? The world is a giant laboratory, and you've got the potential to be an amazing explorer. So, go forth, be curious, and let the magic of science inspire you!
