Lead Ii Nitrate And Sodium Sulfate Net Ionic Equation

Hey there, science buddies! Ever feel like chemistry class is just a bunch of fancy words and equations that make your brain do a little pretzel impression? Yeah, me too! But guess what? Sometimes, the coolest stuff is hiding in plain sight, and today, we're going to tackle something that sounds a bit intimidating but is actually pretty darn neat: the net ionic equation for lead(II) nitrate and sodium sulfate. Don't worry, no complicated lab coats required for this adventure, just your curious mind and maybe a comfy seat!
So, imagine you've got two clear liquids chilling in separate beakers. One has lead(II) nitrate, which, despite its slightly ominous name, is just a soluble salt. Think of it as a happy little dissolved molecule, ready to mingle. The other beaker has sodium sulfate, another friendly soluble salt just waiting for its chance to party. When you pour these two together, something magical happens. Or, you know, scientifically interesting.
The reason we're even talking about this is because when these two solutions meet, a reaction occurs. Not all reactions are like a big bang, you know. Sometimes, it’s more like a subtle shift, a rearrangement of atoms. And in this case, we get a precipitate. What's a precipitate, you ask? Well, it’s like when you’re at a party, and suddenly, a bunch of people decide to form their own little group and stand off to the side. In chemistry terms, it's a solid that forms when two solutions are mixed. And here, our precipitate is lead(II) sulfate. Fancy, right?
Now, to understand why this precipitate forms, we gotta break it down. This is where our fancy-pants "net ionic equation" comes into play. It's like the X-ray vision of chemistry, showing us exactly what’s going on at the microscopic level, stripping away all the spectator ions – the ones that are just chilling, not really doing much.
The Players in Our Little Drama
Let's meet our main characters. We have lead(II) nitrate, which has the chemical formula Pb(NO₃)₂. The "(II)" in lead(II) nitrate tells us that the lead atom has a +2 charge. Nitrates (NO₃) always have a -1 charge, so we need two of them to balance out the lead's charge. Simple enough, right? Like a balanced diet, but with ions.
Then we have sodium sulfate, with the formula Na₂SO₄. Sodium (Na) atoms are like the happy-go-lucky folks of the periodic table, usually sporting a +1 charge. Sulfates (SO₄) are a bit more complex, carrying a -2 charge. So, we need two sodium ions to balance out that single sulfate ion. It's all about keeping things balanced, like a tightrope walker.
The Grand Mixing Ceremony
When we mix these two solutions, what we actually have floating around are individual ions. Think of the water as a giant dance floor, and all these ions are ready to groove. So, lead(II) nitrate in water becomes:
Pb(NO₃)₂(aq) → Pb²⁺(aq) + 2NO₃⁻(aq)
See that "(aq)"? That means "aqueous," which is just a fancy way of saying "dissolved in water." So, our lead ions (Pb²⁺) and nitrate ions (NO₃⁻) are now happily swimming around, separate but together. It’s like a bunch of friends at a party, still in their original groups, but mingling.
And sodium sulfate in water does the same:

Na₂SO₄(aq) → 2Na⁺(aq) + SO₄²⁻(aq)
So now, in our mixed solution, we have four types of ions floating around:
- Lead ions (Pb²⁺)
- Nitrate ions (NO₃⁻)
- Sodium ions (Na⁺)
- Sulfate ions (SO₄²⁻)
It's like a giant chemical mixer! Everyone's introduced, and things are about to get interesting.
The Chemistry Dance: The Full Equation
When we write the overall or molecular equation for this reaction, it looks like this:
Pb(NO₃)₂(aq) + Na₂SO₄(aq) → PbSO₄(s) + 2NaNO₃(aq)
Let's break this down. On the left side, we have our reactants: lead(II) nitrate and sodium sulfate, both dissolved in water. On the right side, we have our products. Notice that PbSO₄(s)? That "(s)" means "solid," and that's our precipitate, our lead(II) sulfate, forming a cloudy solid at the bottom of the beaker. It's like some of our partygoers decided to team up and become something new and solid!
We also have 2NaNO₃(aq). This is sodium nitrate, and it's still dissolved in water. So, while the lead and sulfate ions decided to get together and form a solid, the sodium and nitrate ions decided to remain partners and stay dissolved. They're like the couple at the party who are just happy to chill together.

Peeling Back the Layers: The Net Ionic Equation
Now, here's where the magic of the "net ionic equation" comes in. Remember those spectator ions? They're the ones that were dissolved at the beginning and are still dissolved at the end, unchanged. They didn't really participate in forming the new solid. They're just… there. Like the background music at the party.
In our reaction, the sodium ions (Na⁺) and the nitrate ions (NO₃⁻) are the spectators. They were floating around as ions in the reactants, and they are still floating around as ions in the product (sodium nitrate). They didn't get incorporated into the solid precipitate.
So, to get the net ionic equation, we take our full molecular equation and remove the spectator ions from both sides.
Let's look at the dissociated ions of our full equation:
Pb²⁺(aq) + 2NO₃⁻(aq) + 2Na⁺(aq) + SO₄²⁻(aq) → PbSO₄(s) + 2Na⁺(aq) + 2NO₃⁻(aq)
Now, let’s cross out the ions that appear on both sides:
Pb²⁺(aq) + 2NO₃⁻(aq) + 2Na⁺(aq) + SO₄²⁻(aq) → PbSO₄(s) + 2Na⁺(aq) + 2NO₃⁻(aq)

And voilà! What's left is our net ionic equation:
Pb²⁺(aq) + SO₄²⁻(aq) → PbSO₄(s)
This equation tells us the real story of the reaction. It shows us that it's the lead(II) ions and the sulfate ions that are coming together to form the solid precipitate of lead(II) sulfate. The sodium and nitrate ions were just there for the ride, so to speak.
Why Bother With This Net Ionic Thingy?
You might be thinking, "Why go through all this trouble? The full equation tells us what's happening!" And you're right, it does. But the net ionic equation gives us a deeper understanding. It highlights the essential chemical change. It tells us that any soluble lead(II) compound reacting with any soluble sulfate compound will produce a lead(II) sulfate precipitate, because it's the interaction between the Pb²⁺ and SO₄²⁻ ions that drives the formation of the solid.
Think of it like this: if you're trying to understand the core of a joke, you focus on the punchline, not all the filler words that led up to it. The net ionic equation is the punchline of this reaction!
It's also super useful for predicting reactions. If you know that lead(II) sulfate is insoluble, and you're mixing a solution containing lead(II) ions with a solution containing sulfate ions, you can confidently predict that you'll form a precipitate, even if you don't know the exact counter-ions involved. It simplifies things, making chemistry a bit less of a puzzle and a bit more of a clear picture.
A Little About Solubility Rules (The Party Guest List)
You might be wondering how we know that lead(II) sulfate is insoluble, while sodium nitrate is soluble. That's where solubility rules come in. These are like the universal guidelines for which ionic compounds will dissolve in water and which will not. They're incredibly handy!
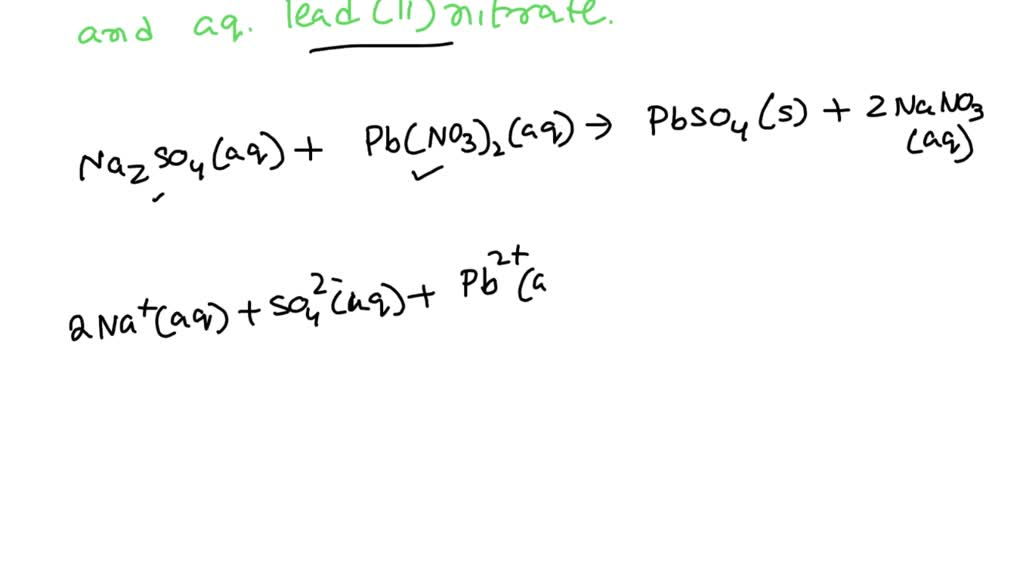
Some general rules that are helpful here:
- Nitrates (NO₃⁻) are almost always soluble. So, Pb(NO₃)₂ is soluble.
- Group 1 metal cations (like Na⁺, K⁺, Li⁺) form soluble compounds. So, Na₂SO₄ is soluble.
- Sulfates (SO₄²⁻) are generally soluble, BUT… there are some exceptions, and lead(II) sulfate (PbSO₄) is one of them! It's considered insoluble.
These rules are like a cheat sheet for predicting whether a precipitate will form. They help us determine which ions are going to team up and become solid.
Putting It All Together with a Smile
So, to recap our little journey: we mixed lead(II) nitrate and sodium sulfate. They dissociated into their ions in water. The lead ions and sulfate ions decided they made a great match and formed an insoluble solid, lead(II) sulfate. The sodium and nitrate ions, meanwhile, just continued on their merry way, staying dissolved.
The full story, the molecular equation, shows all the players: Pb(NO₃)₂(aq) + Na₂SO₄(aq) → PbSO₄(s) + 2NaNO₃(aq).
But the real action, the core chemical transformation, is captured by the net ionic equation: Pb²⁺(aq) + SO₄²⁻(aq) → PbSO₄(s). This is where the chemistry truly happens!
And you know what? Understanding this, even just a little bit, is a fantastic step. Chemistry isn't about memorizing a million facts; it's about understanding how things interact and change. Every time you grasp a concept like this, you're unlocking a new way to see the world around you. From the bubbles in your soda to the formation of rocks, these chemical principles are at play.
So, next time you're feeling a bit overwhelmed by chemical equations, just remember our little precipitate party. Remember the ions dancing, the partners forming, and the spectators just chilling. You’ve got this! Keep that curiosity alive, keep asking questions, and keep exploring the amazing world of chemistry. You're doing great, and every little bit of understanding is a step towards a brighter, more informed future. Keep shining, science star!
