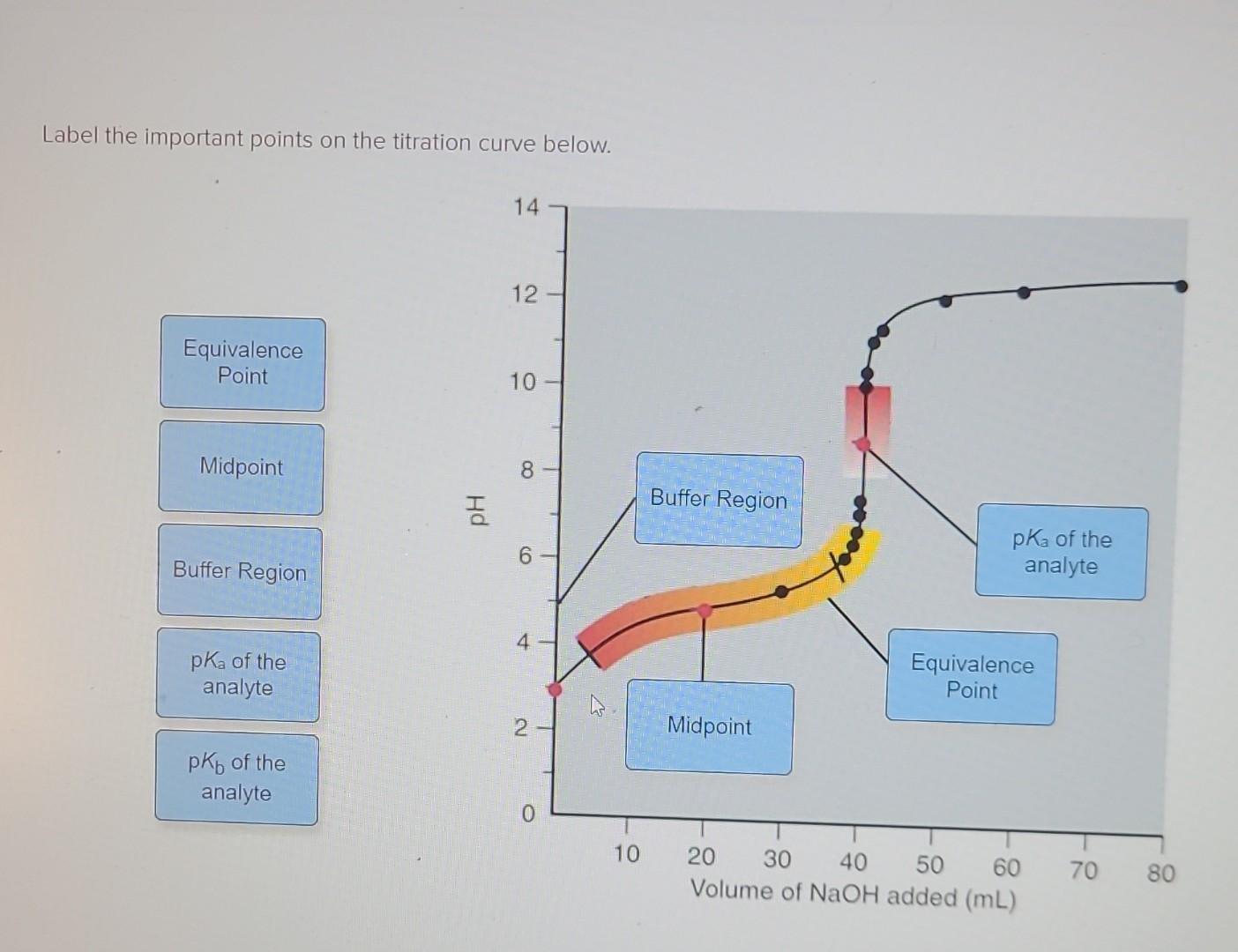
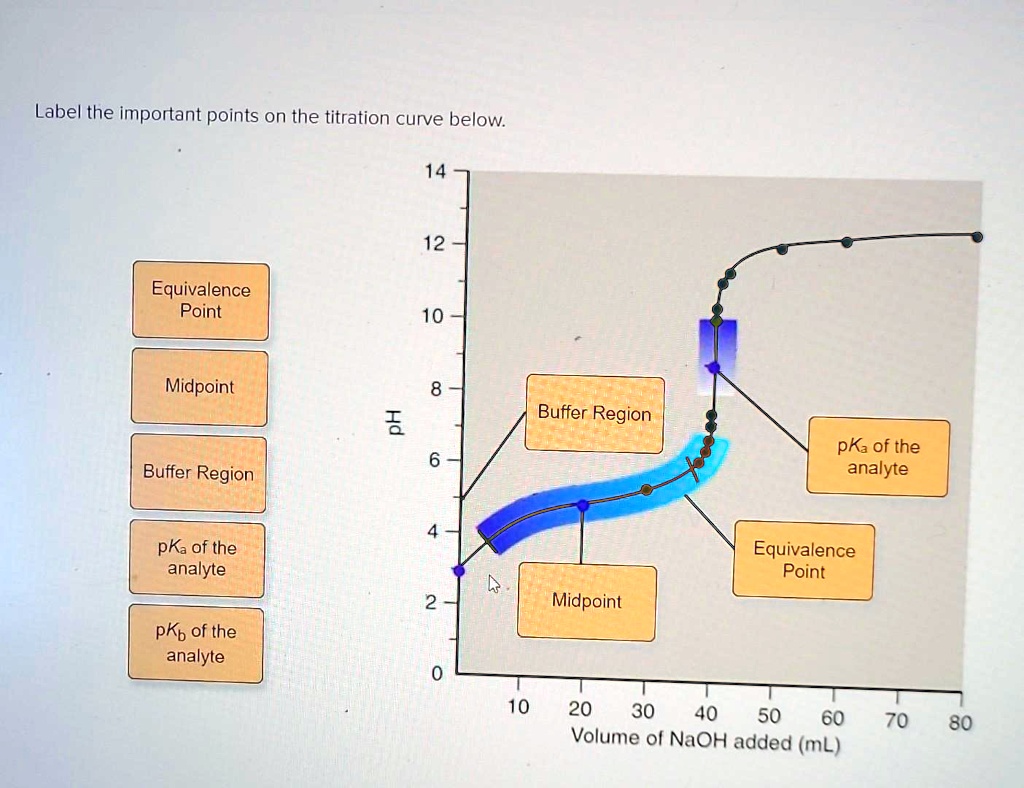
Label The Important Points Of The Titration Curve Below.
Hey there, fellow explorers of the subtly complex! Ever found yourself staring at a graph and feeling like you're deciphering ancient hieroglyphics? We get it. Science, sometimes, can feel a tad intimidating. But what if we told you that understanding a titration curve is actually way more chill than you think, and can even offer some cool insights into everyday life? Think of it as a scientific roadmap, guiding us through the sweet spot of chemical reactions. No need to panic; grab a coffee (or your beverage of choice), settle in, and let's break down this fascinating visual, one point at a time. It’s less about memorizing formulas and more about appreciating the ebb and flow of a chemical dance.
Imagine you're making the perfect cup of tea. You've got your water, your tea leaves, and then you add sugar. A titration curve is kind of like charting how the sweetness changes as you add sugar, but with chemicals. It’s a visual story of a reaction reaching its perfect balance, or as we scientists like to call it, its equivalence point. Pretty neat, right?
This journey will help you label the important points on a titration curve, making you feel like a true lab wizard – or at least someone who can confidently nod along at a science documentary. We'll sprinkle in some relatable analogies, maybe a nod to a favorite rom-com where a perfect match is key, and even a little bit of fun trivia. So, let's dive into the wonderful world of titration curves, shall we? It's going to be a smooth ride, promise!
The Grand Entrance: Where It All Begins
So, picture this: you're at the start of the curve. This is the initial point. Think of it as the moment before the magic happens. In our tea analogy, this is your plain hot water, before any sugar or tea leaves have been added. It's the baseline, the starting canvas. Chemically speaking, this is where we have our initial solution, which could be an acid or a base, all by itself. It's pure potential, waiting for its reaction partner.
No complex chemistry here, just the humble beginnings. It’s like the opening scene of a movie, setting the stage for everything that’s about to unfold. This point is crucial because it gives us our starting pH (or potential of hydrogen), a measure of how acidic or basic our initial solution is. It’s the foundation upon which the entire curve is built. So, when you see that graph starting off, remember: it’s just the beginning of a beautiful story.
The Steep Ascent: Picking Up the Pace
As we start adding our titrant – that’s the solution we’re carefully dripping in, drop by drop – things start to get interesting. We move into the region of the buffer region. This is where the pH changes gradually. Think of it like dating. At the beginning, small gestures of kindness can have a big impact. In chemistry, this is where a weak acid and its conjugate base (or a weak base and its conjugate acid) are working together, buffering the solution against drastic pH changes. It’s their way of saying, “We’ve got this!”
This buffer region is super important, especially in biological systems. Our bodies, for instance, rely on buffer systems to keep our blood pH stable, around 7.4. Imagine if your blood pH swung wildly every time you ate something slightly acidic or alkaline – that would be a recipe for disaster, or at least a very uncomfortable afternoon! This is nature’s elegant solution, and it’s mirrored in these chemical curves.
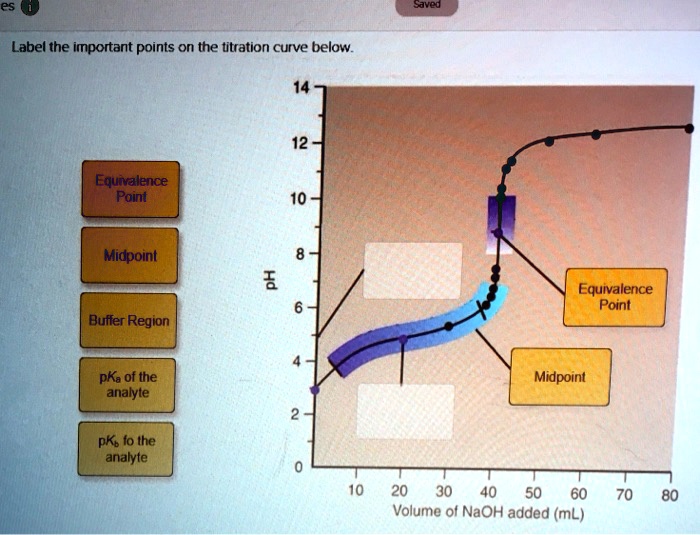
Culturally, this buffer region reminds us of those moments in life where things are stable, predictable, and comfortable. It's the calm before the storm, or more accurately, the calm before the perfect point. It’s where the characters in a rom-com might be having a delightful, but slightly predictable, few dates before the real sparks fly. The pH is doing its thing, resisting major change, keeping things steady.
The Game Changer: The Equivalence Point
Ah, the star of the show! The equivalence point. This is the moment when the moles of the added titrant have exactly reacted with the moles of the substance in the initial solution. In our tea analogy, this is where you’ve added just the right amount of sugar to make your tea perfectly sweet, not too sugary, not too bland. It’s the sweet spot, the ideal balance.
Chemically, this is where the reaction is complete. If you're titrating an acid with a base, at the equivalence point, all the acid has been neutralized by the base, or vice versa. The pH at this point will depend on the strength of the acid and base used. For a strong acid and strong base titration, the equivalence point is at pH 7. For a weak acid and strong base, it’ll be above 7, and for a strong acid and weak base, it’ll be below 7. It's like the perfect chord in a song, hitting just the right note.
This point is like the climax of a movie, or the moment the protagonist realizes their true feelings. It’s the culmination of all the effort and anticipation. In chemistry, it’s the most information-rich point on the curve. It tells us exactly how much of the unknown substance we started with, based on how much titrant we needed to reach this perfect neutralization. It’s precision at its finest. Think of Sherlock Holmes finally piecing together all the clues – that’s the equivalence point for a chemist.
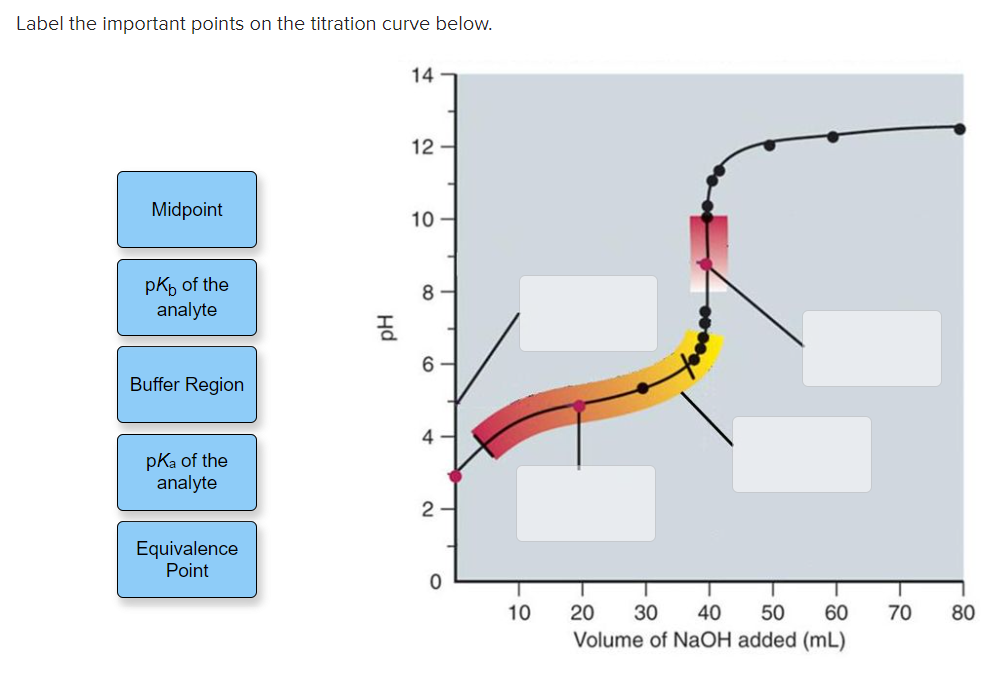
The Indicator's Whisper: The Endpoint
Now, here’s where things get a little nuanced, and honestly, kind of cool. We can’t always see the exact equivalence point with our naked eyes. That's where our trusty indicator comes in! An indicator is a substance that changes color at a specific pH range. The point where the indicator changes color is called the endpoint.
Ideally, we want our indicator to change color as close as possible to the equivalence point. If our indicator is well-chosen, the endpoint will be our best visual approximation of the equivalence point. It’s like having a helpful friend point out the exact moment the magician’s trick is completed. The color change is the signal, the flashing neon sign that says, "We're there!"
Think of it as a surprise party. You might have planned the perfect moment for the reveal, but sometimes things happen a little sooner or a little later. The indicator is just letting us know when our planned reveal moment has occurred. The trick is to choose an indicator whose color change range overlaps significantly with the steep change in pH around the equivalence point. It’s a marriage of chemical prediction and visual cue. It’s the applause after the perfect performance.
A fun fact: Litmus paper, that classic red and blue stuff you might have seen in school labs, is actually a mixture of natural dyes derived from lichens. It's one of the oldest and most widely used acid-base indicators. So, next time you see litmus, give a nod to ancient scientific traditions!
Beyond the Peak: When Enough is Enough (and Then Some)
After the equivalence point, even if we continue to add titrant, the pH won't change as dramatically. This region is sometimes referred to as the excess titrant region. In our tea analogy, this is like adding way too much sugar. Your tea is already perfectly sweet, and adding more sugar just makes it cloying and unpleasant. The sweetness (or in this case, the pH) has reached its maximum potential for change with respect to the original substance.
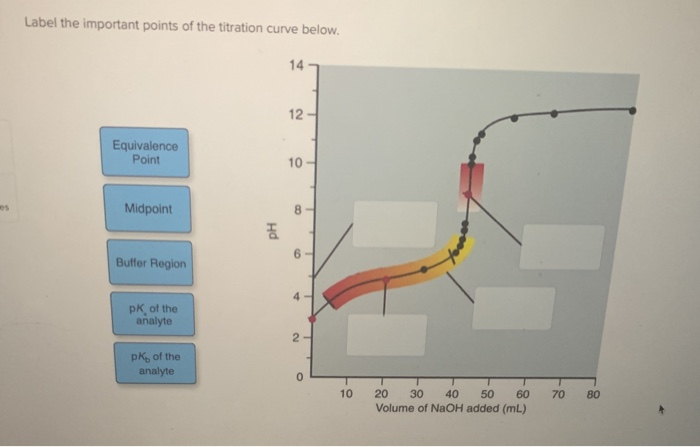
Chemically, this means that all of the original substance has been reacted, and now we're just adding more of the titrant to a solution that's already dominated by the titrant. The pH is now primarily dictated by the excess titrant itself. For example, if you're titrating an acid with a base and you add a lot of excess base, the solution will become very basic.
This part of the curve is less about finding a perfect balance and more about understanding the properties of the excess reagent. It’s the dénouement of the story, where the characters have moved past the main conflict and are dealing with the aftermath. It's the "happily ever after," or in some cases, the slightly overdone "happily ever after." It helps us confirm that we’ve indeed passed the critical point and are now in a different chemical landscape.
The Inflection Point: The Heart of the Curve
Within the steep region leading up to and through the equivalence point, there’s a specific point called the inflection point. This is the point where the rate of change of the pH is at its maximum. Imagine a roller coaster: the inflection point is where the track is steepest as you're going over the crest of a hill. It’s the point of greatest curvature on the graph.
In titration curves, the inflection point is often very close to, or coincides with, the equivalence point. It’s the visual representation of the most rapid pH shift occurring. While the equivalence point is a theoretical measure of moles reacted, the inflection point is a graphical characteristic you can visually identify on the curve. It’s the sharpest turn in the road, the most dramatic moment of transition.
Understanding the inflection point can be particularly useful when dealing with less precise measurements. It's a highly sensitive area of the curve, indicating that even a tiny addition of titrant is causing a significant pH change. It’s like noticing the exact second a celebrity’s expression shifts from polite to genuinely surprised – that’s the inflection point of their reaction!
The Takeaway: More Than Just Lines on Paper
So, there you have it! We’ve navigated the key points of a titration curve: the humble initial point, the stable buffer region, the highly anticipated equivalence point, the visually signaled endpoint, the region of excess titrant, and the dynamic inflection point. Each point tells a part of the story of a chemical reaction, a story of balance, change, and precise measurement.
Think of it like this: When you’re cooking, you’re constantly adjusting. You taste the sauce, add a pinch of salt, stir, taste again. You're looking for that perfect balance of flavors. That’s essentially what a titration curve maps out, but with the precise language of chemistry. The curve shows us the journey to that perfect flavor, that ideal chemical state.
In our daily lives, this concept of finding a perfect balance is everywhere. It's in maintaining a healthy work-life balance, finding the right mix of ingredients for your signature dish, or even in relationships, where finding that sweet spot of give-and-take is crucial. Titration curves, in their own scientific way, are a beautiful reminder that sometimes, the most profound insights come from observing gradual changes and identifying that critical point of equilibrium.
So, the next time you see a graph, whether it’s in a lab report or a business presentation, remember the titration curve. It’s a testament to how even the most complex processes can be understood by carefully observing the journey and pinpointing the crucial moments. It’s about recognizing when you’ve reached that perfect point, and appreciating the journey it took to get there.
