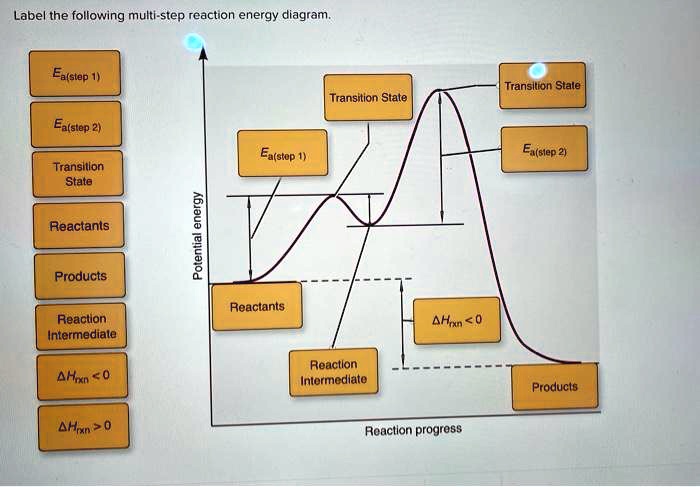
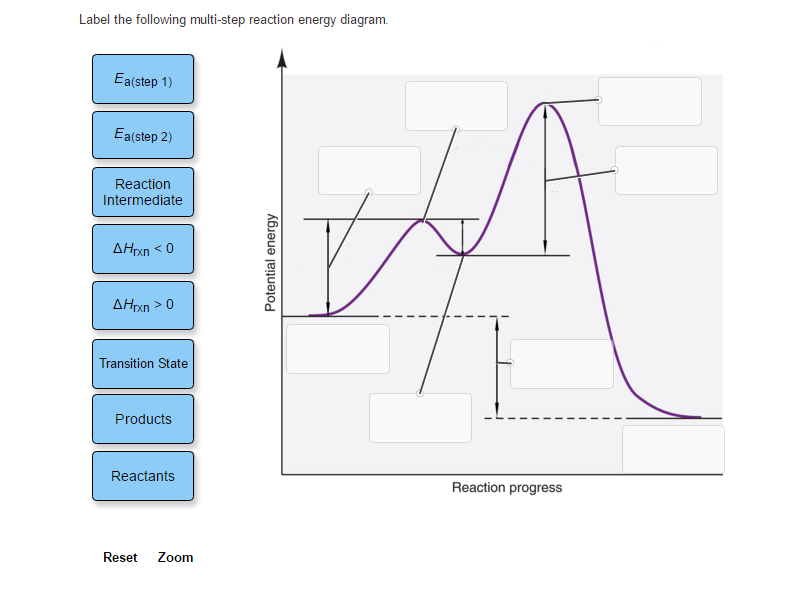
Label The Following Multi Step Reaction Energy Diagram
Hey there, science enthusiasts and curious minds! Ever looked at a chemical reaction and wondered what’s really going on behind the scenes? We’re not just talking about things mixing and bubbling, oh no. There’s a whole energetic dance happening, a kind of invisible rollercoaster that reactants go on before they become those new, exciting products. Today, we’re going to peek at a special kind of map that shows us this journey: a multi-step reaction energy diagram. Think of it as the GPS for a chemical transformation, showing us all the twists, turns, and even the little rest stops along the way.
So, what is this magical diagram, anyway? Imagine you’re trying to get from your couch to the kitchen for a snack. That’s your overall reaction. Easy peasy, right? But what if there are a few hurdles in the way? Maybe you have to navigate past a sleeping cat (don’t disturb the cat!), open a tricky cabinet, or even do a little dance to reach the cookie jar. Each of those little actions is a step in your journey. A multi-step reaction is just like that – it’s not a direct flight from A to B. It’s a journey with a few intermediate stops, a few little bumps and humps.
And our energy diagram? It’s like a profile of that journey, plotting how much energy you need to put in or how much you get out at each stage. On one axis, we have progress of the reaction – basically, how far along the chemical change is. On the other axis, we have the energy level. It’s like climbing a mountain, right? The higher you go, the more energy you’ve expended. Or maybe it’s more like a rollercoaster! Some bits are uphill, requiring effort, and some are downhill, where energy is released.
Now, for the fun part: labeling the diagram! Let’s break down what all those squiggles and peaks actually mean. First, you'll see your starting point. This is where all your reactants hang out, full of potential energy, just waiting to get the party started. Think of them as the ingredients before you start baking. They’re all there, but they haven’t transformed into a delicious cake yet.
As the reaction begins, the reactants start to rearrange. This is where things get interesting! For each individual step in a multi-step reaction, there’s a little uphill climb. This peak represents what we call the transition state. Imagine you’re trying to push a heavy box over a small hill. You have to exert energy to get it to the very top, right? That peak is the point of highest energy for that specific step. It’s a fleeting, unstable arrangement of atoms, kind of like a tightly wound spring, just about to spring into action.
And right at the top of that little hill, that’s your activation energy for that step. It's the minimum amount of energy needed to get the reaction moving. If you don't give it enough push, the box won't go over the hill. Simple as that! For a multi-step reaction, you’ll often see multiple of these little uphill climbs and transition states, one for each step the reaction takes.
After reaching the transition state and overcoming the activation energy, the reaction then tumbles down to a lower energy level. This is called an intermediate. Think of it as a little resting spot on your journey to the kitchen. You've opened the cabinet and are holding a bag of chips, but you haven't eaten them yet. This intermediate is a more stable species than the transition state, but it's not your final product. It's like a mini-product, ready to undergo the next step in the transformation.
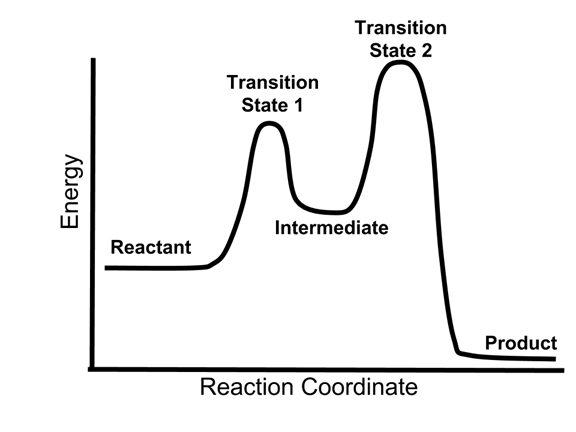
And guess what? This whole process of climbing a hill (activation energy) to a transition state, and then rolling down to an intermediate, happens multiple times in a multi-step reaction. It’s like a series of mini-rollercoasters, each with its own set of hills and valleys. Each intermediate is like a point where the reaction pauses before embarking on its next energetic adventure.
So, how do we label these multiple stages? Well, you’ll see the energy levels of these intermediates. Some might be a bit higher than others, meaning they’re a bit more energetic and less stable. Others might be lower, making them more stable. It’s like some rest stops are more comfortable than others!
What about the final destination? That’s where our products reside. After all the steps, all the climbing and rolling, the atoms have rearranged into their final form. The difference in energy between the reactants (our starting point) and the products (our end point) tells us whether the overall reaction releases energy (exothermic) or absorbs energy (endothermic). If the products are at a lower energy level than the reactants, it’s like finding a treasure chest – energy has been released! If the products are at a higher energy level, it means energy was absorbed, like having to pay for that treasure chest.
The Fastest Way: Rate-Determining Step
Now, here’s a really cool thing about multi-step reactions. Not all the steps are created equal. Some steps are like a leisurely stroll, while others are like a sprint. The step that takes the longest, the one that requires the most energy to get over its activation energy hump, is called the rate-determining step. This is the bottleneck! No matter how fast the other steps are, the overall reaction can only go as fast as this slowest step. It’s like waiting in line at a theme park – the whole queue moves at the pace of the slowest person at the front.
On our energy diagram, this rate-determining step will have the highest activation energy peak among all the individual steps. It’s the biggest mountain to climb in the entire journey. Identifying this step is super important in chemistry because it helps scientists figure out how to speed up or slow down a reaction. If you want to get your snack faster, you might need to find a shortcut past the sleeping cat, or a quicker way to open that cabinet!

So, when you see a multi-step reaction energy diagram, don’t just see a bunch of lines and bumps. See a story! See the journey of molecules transforming. See the effort they put in, the little pauses they take, and the energy they either release or absorb along the way. It’s a visual representation of a dynamic, energetic dance that’s happening all around us, even in the simplest of chemical changes.
It’s pretty neat, isn’t it? The next time you encounter one of these diagrams, you'll know you're not just looking at abstract lines, but at a detailed roadmap of a chemical adventure. You'll be able to point out the reactants, the transition states, the intermediates, the products, and even the sneaky rate-determining step. It’s like having a secret decoder ring for the world of chemistry!
Keep exploring, keep asking questions, and keep appreciating the amazing energy transformations that make our world go 'round!
