Label The Energy Diagram For A Two Step Reaction
Have you ever wondered what goes on behind the scenes when a chemical reaction happens? It’s not just a splash and a puff of smoke, you know! There’s a whole hidden world of energy at play. And when we talk about a two-step reaction, things get even more interesting. Think of it like a mini-adventure for molecules. It’s not one smooth ride; it’s got a couple of little bumps and twists along the way. And guess what’s the best way to see this adventure unfold? You guessed it – an energy diagram!
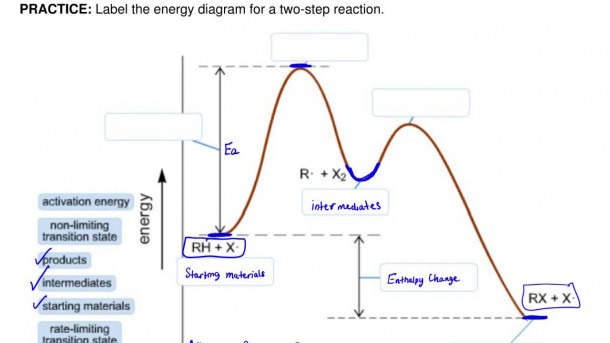
Now, an energy diagram might sound a bit like homework, but trust me, it’s way more fun than that. It’s like a secret map that shows us exactly how much energy is needed to get things moving and how much is released. For a two-step reaction, this map has a special charm. It’s not a simple uphill and then downhill. Oh no, it's got a little plateau or a mid-point peak that makes it extra special. It’s like watching a skilled acrobat perform. They don't just jump from here to there; they might do a little flip or a pause mid-air. That’s kind of what’s happening with the energy.
Let’s break down this exciting map. On the bottom, you've got what we call the reaction coordinate. Think of this as the path the reaction is taking, step by step. It’s like the road you’re driving on, showing how far you’ve gone on your journey. On the side, the tall part, that’s where the energy lives. This shows you how much oomph, how much power, is needed or is let out at each stage.
So, for our two-step reaction, the diagram will show you two distinct “humps” or “hills” to climb. Each hump represents a step, a little phase in the overall process. The first hump is the energy needed to get the first part of the reaction going. This is called the activation energy for the first step. It's the initial push required to start the ball rolling. Imagine pushing a heavy box; you need a good shove to get it moving. That shove is the activation energy!
And then, after climbing that first hill, the molecules might not immediately go to the final destination. They’ll often pause at a little “resting spot” in between. This resting spot is super important, and we call it the intermediate. It's like a pit stop on your road trip. The molecules are changed, but they’re not quite done yet. This intermediate is a key player, a temporary character in our chemical drama. You’ll often see a dip in the energy diagram after the first peak, showing this intermediate state. It's stable enough to exist for a bit, but not forever.

Then comes the second act! From this intermediate resting spot, the molecules need to climb another hill to reach the final product. This second climb also has its own activation energy. It might be higher or lower than the first one, making the second step easier or harder to complete. Think of it as the final sprint to the finish line. This second peak is crucial because it tells us how much energy is needed to get us to the very end of our reaction.
What makes this so captivating? It’s the unveiling of the unseen. We get to peek into the energy rollercoaster. The highest point on the entire diagram is the most challenging part of the whole journey. It's the transition state for the step with the highest activation energy. Whoever conquers this peak has overcome the biggest hurdle in the reaction. It's like the boss level in a video game!
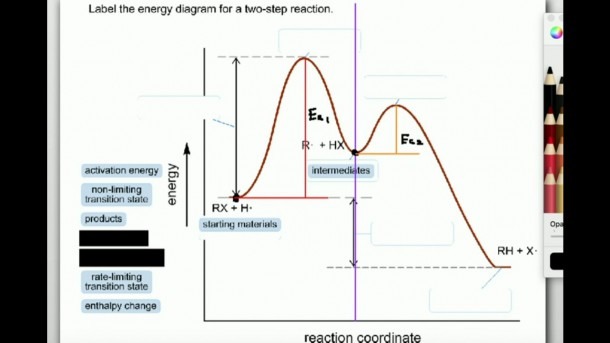
The difference in energy between where the reaction starts (the reactants) and where it ends (the products) is also super revealing. This is the overall energy change. If the products have less energy than the reactants, the reaction releases energy (an exothermic reaction, often feeling warm, like a cozy blanket). If the products have more energy, the reaction absorbs energy (an endothermic reaction, feeling cool, like a refreshing drink). It's like seeing if your adventure ended with a treasure chest full of energy or if you had to spend a lot of your own to get there.
The beauty of labeling an energy diagram for a two-step reaction lies in its detail. You're not just looking at a simple start and end. You're appreciating the journey, the mini-challenges, and the temporary pauses. It’s like reading a book with a compelling plot twist instead of just a summary. Each label – reactants, activation energy 1, intermediate, activation energy 2, products, and the overall energy change – tells a part of the story. It’s a visual narrative of molecular transformation, and honestly, it’s pretty neat to be able to understand it.
So, next time you hear about a reaction having “steps,” picture that energy diagram. Imagine those hills, those resting spots, and the thrilling climb. It’s a way to make the invisible world of chemistry visible and surprisingly engaging. It’s a map to understanding how much effort and how much reward is hidden within every chemical change. It’s like having a cheat sheet to the universe’s energy secrets. And who wouldn’t want to look at that?
