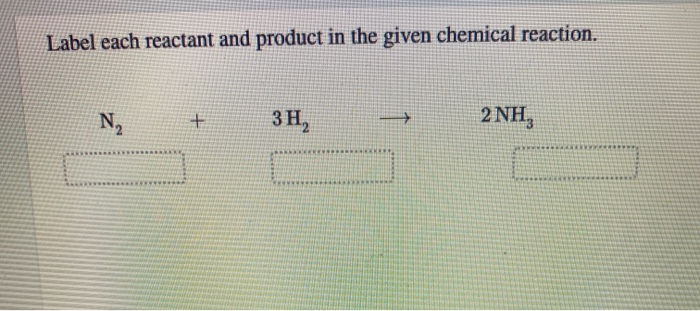
Label Each Reactant And Product In The Given Chemical Reaction
Hey there, fellow explorers of the universe! Ever found yourself staring at a chemical reaction, maybe something you saw in a science class or a cool documentary, and thought, "Whoa, what's actually going on here?" It's like a tiny, invisible dance, right? And just like any good dance, there are partners, and then there are the people who become something new by the end of the song. Today, we're going to break down a super fundamental concept in chemistry: figuring out who's who in the reaction zoo. We're talking about labeling the reactants and the products.
Think of it like baking a cake. You don't just magically end up with a delicious chocolate masterpiece, do you? Nope. You start with a bunch of individual ingredients: flour, sugar, eggs, cocoa powder. These are your starting materials. In the world of chemistry, we call these the reactants. They're the OG ingredients, the things that are there before the magic happens.
And what happens after you mix all those ingredients and pop them in the oven? You get a cake! That cake is totally different from the raw flour or the liquid eggs, right? It’s a brand new creation. In chemistry, this brand new creation is called a product. It's what you end up with after the reaction has done its thing. Pretty straightforward, huh?
So, How Do We Actually "Label" Them?
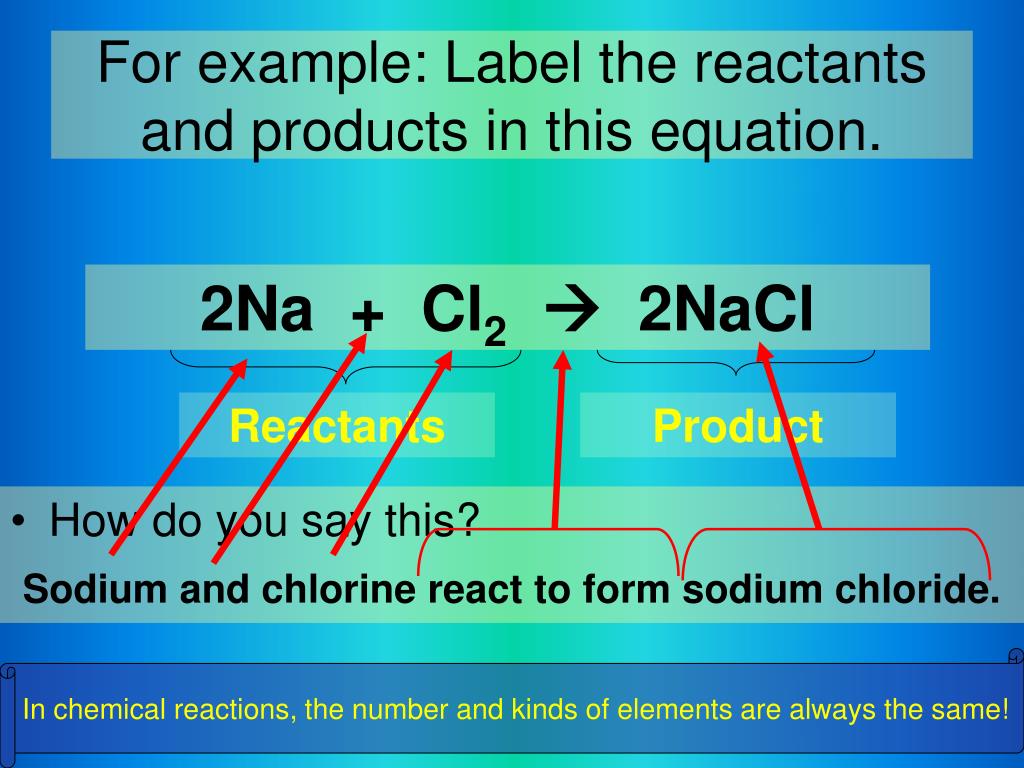
When scientists write down a chemical reaction, it's usually in a special shorthand. It's like a recipe, but for atoms and molecules. You'll see a bunch of chemical formulas, and crucially, an arrow. This arrow is like the chef's "bake me!" instruction. It points from the ingredients to the final dish.
Let's imagine a super simple example. What happens when you burn wood? You start with wood, right? And you need oxygen from the air to make it burn. These are your reactants. And what do you get? Smoke, ash, and heat. In a chemical sense, the smoke and ash are the main products (along with some gases we can't easily see, like carbon dioxide). We could represent this with an arrow.
On the left side of the arrow, you'll find all the reactants. They're the guys who are about to go through a transformation. On the right side of the arrow, you'll find all the products. These are the brand-new things that have been cooked up by the chemical reaction.

Why is This Labeling Stuff Even Important?
Well, besides satisfying our curiosity about what's what, it's the foundation of understanding chemistry. It’s like knowing the difference between a guitar and a drum kit before you try to play a song. You need to know what you're starting with and what you're aiming for.
Imagine you're trying to build with LEGOs. You have a pile of different bricks (your reactants). You want to build a spaceship. The spaceship is your ultimate goal, your product. You wouldn't just grab random bricks and hope for a spaceship; you'd follow instructions, or at least have a plan of what you're trying to assemble. Chemical reactions are similar, but instead of bricks, we have atoms and molecules rearranging themselves.
Knowing the reactants tells us what chemicals are going into the "mixing bowl." Knowing the products tells us what we're getting out. This is super important for so many things:
- Predicting what will happen: If you know the reactants, and you know the general rules of chemistry, you can often predict what products you're likely to get. This is how scientists discover new reactions and materials!
- Controlling reactions: If you want to make a specific product, you need to start with the right reactants and make sure the reaction goes in the direction you want it to.
- Understanding the world around us: From the way your body digests food to how plants make their own food (photosynthesis!), chemical reactions are happening all the time. Being able to label reactants and products helps us understand these amazing natural processes.
Let's Get a Little More Specific (But Still Chill!)
Okay, let's take another look at that baking analogy. When you mix flour, eggs, and sugar, they are the reactants. When you bake them, they undergo a chemical change. The heat causes new bonds to form and break, transforming them into the delicious cake. That cake, with its fluffy texture and sweet taste, is the product. It's a completely different substance than the individual ingredients.
In a chemical equation, you'll see something like this:
Reactant A + Reactant B → Product C + Product D
See the arrow? It's the key! Everything to the left of the arrow (Reactant A and Reactant B) are the starting materials, the things that are going to react. Everything to the right of the arrow (Product C and Product D) are the results, the new things that are formed.

Sometimes, there are multiple reactants or multiple products. Think about making a sandwich. You start with bread, cheese, and maybe some ham. Those are your reactants. What's the product? A delicious cheese and ham sandwich! We can have more than one "thing" we put in, and we end up with one "thing" we can eat.
A Fun Little Real-World Example
Let's consider the reaction that makes water. We all know water, right? H₂O. It's pretty essential. How do we make it from scratch (in a chemistry lab, of course)? Well, we can take hydrogen gas (H₂) and oxygen gas (O₂). These are our starting ingredients, our reactants.
If we mix them and add a little spark (like a tiny explosion!), they react to form water! So, in this case:
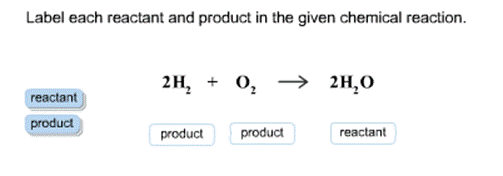
- Reactants: Hydrogen gas (H₂) and Oxygen gas (O₂)
- Product: Water (H₂O)
The chemical equation looks like this:
2H₂ + O₂ → 2H₂O
Even with the numbers in front (which tell us how many of each molecule are involved), we can still easily see who's who. The things before the arrow are the ones doing the reacting (the reactants), and the thing after the arrow is what's made (the product). Cool, right?
So, the next time you see a chemical reaction, don't be intimidated! Just look for that arrow. The stuff on the left are the reactants, ready to party and transform. The stuff on the right are the products, the awesome new creations that emerge from the chemical dance. It’s like being a detective, spotting the clues and figuring out the story of what happened. Keep exploring, keep asking questions, and keep enjoying the amazing chemistry all around you!
