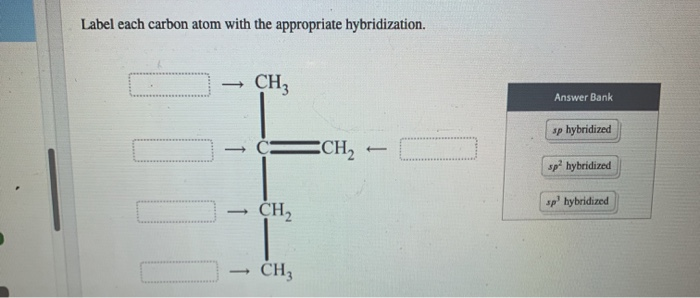
Label Each Carbon Atom With The Appropriate Hybridization
Alright, settle in, grab your latte, and let's talk about something that sounds drier than a week-old croissant but is actually, dare I say it, kinda fun? We're diving into the wild world of carbon atoms and their oh-so-fashionable hybridization. Think of it as carbon's personality makeover, where it decides to dress up in different outfits depending on who it's hanging out with. It's like deciding whether to wear a ballgown for a fancy dinner or ripped jeans for a chill movie night. Carbon does that, but with orbital configurations. Wild, right?
So, what's this "hybridization" all about? Basically, it's how carbon atoms get ready to bond with other atoms. They take their plain ol' atomic orbitals – those imaginary spaces where electrons hang out – and smoosh them together to make new, hybrid orbitals. These new orbitals are just right for making strong, stable connections. It's like having a bunch of separate, clunky tools and then inventing a super-tool that does ten jobs at once. Efficiency, baby!
Now, carbon has three main styles, three main outfits it likes to wear: sp³, sp², and sp. Each one dictates how many friends (bonds) carbon can make and what shape its little molecular party will take. It’s like being a social butterfly at a cocktail party – you can mingle with one person deeply, a couple of people in a small group, or a whole crowd! Let's break it down, shall we?
The All-Star, Always-Ready: sp³ Hybridization
First up, we have our reliable, workhorse, the sp³ hybridized carbon. This is carbon in its most enthusiastic, "let's-be-friends-with-everyone" mode. It takes one 's' orbital and all three 'p' orbitals and mixes them up. Poof! Four brand new, identical sp³ orbitals. These orbitals point out in all directions like a perfectly symmetrical asterisk, ready to form four single bonds.
Think of methane (CH₄). That's the simplest organic molecule, basically a carbon atom with four hydrogen buddies. Every single one of those carbon-hydrogen bonds is a happy, single bond, facilitated by the sp³ orbitals. It's like a group hug, totally inclusive. The shape? A perfect tetrahedron. Imagine a pyramid with a triangular base, but upside down. Super stable, super common. This is the carbon you’ll find in all those saturated fats your doctor is probably telling you to cut down on (oops!).

This sp³ hybridization is the backbone of so many molecules. It's the friendly handshake, the firm nod. It’s everywhere! It's so prevalent, it’s practically the default setting for carbon. If you see a carbon atom that's only making single bonds, chances are, it’s rocking the sp³ look. And honestly, it looks good on them. It’s like the classic little black dress of the molecular world – always appropriate, always stylish.
The Double-Duty Daredevil: sp² Hybridization
Next, things get a little more interesting. Enter the sp² hybridized carbon. This one decides, "You know what? Four single bonds is a bit much. Let's spice things up." It takes one 's' orbital and two of the 'p' orbitals, smashes them together, and voilà – three sp² orbitals. These three orbitals arrange themselves in a flat triangle, like a perfectly laid-out picnic blanket. But wait, what happened to that third 'p' orbital? It's still there, unhybridized, just chilling, and ready to do something special.

This unused 'p' orbital is the star of the show for sp² carbons. It’s used to form a double bond. Yep, you heard that right. Two atoms sharing not one, but two pairs of electrons! Think of ethene (C₂H₄). Each carbon atom in ethene is sp² hybridized. They each form two single bonds with hydrogen atoms, and then they get together for a double bond between themselves. It's like they're holding hands and giving each other a high-five simultaneously. Intense!
The geometry around an sp² carbon is trigonal planar. Flat as a pancake, literally. And that double bond? It's a bit more rigid than a single bond. You can't just spin around freely. This rigidity is super important in biological molecules. It's like the difference between a floppy noodle and a nice, crisp piece of spaghetti. This sp² character is responsible for all those unsaturated fats (the "good" kind, thankfully!) and a whole heap of reactive molecules. They’re the life of the party, ready to engage in new friendships (reactions).
The Speedy, Zingy One: sp Hybridization
Finally, we arrive at the sp hybridized carbon, the rockstar of the bunch. This carbon is all about efficiency and, frankly, a bit of a minimalist. It takes one 's' orbital and just one 'p' orbital, mixes them up, and creates two sp orbitals. These two orbitals are linear, stretched out like a tightrope walker on a wire, 180 degrees apart. But the real action? It leaves two unhybridized 'p' orbitals hanging out.
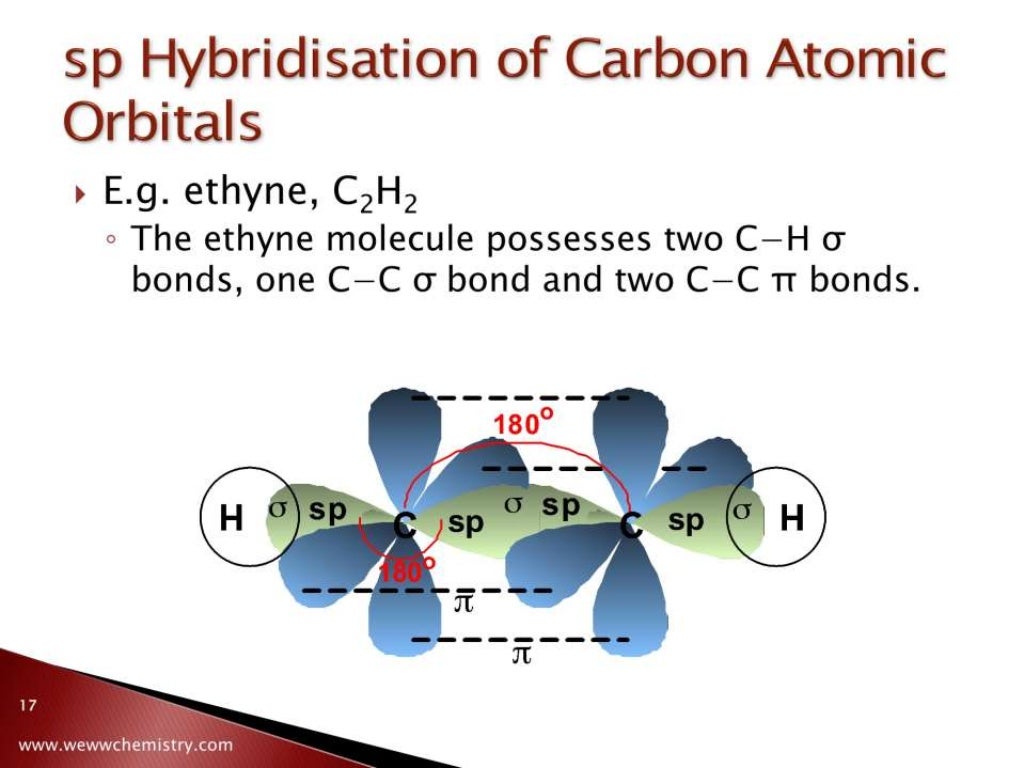
What do those two extra 'p' orbitals do? They form not one, but two double bonds! Or, one triple bond. Mind. Blown. Take ethyne (acetylene, C₂H₂). Each carbon atom in ethyne is sp hybridized. They form a single bond to one hydrogen atom each, and then they have a triple bond between them. That’s three pairs of electrons being shared! It’s like a super-hug, a triple-threat connection. This is one of the strongest types of covalent bonds you'll find.
The geometry around an sp carbon is linear. Straight as an arrow. No curves allowed. These molecules are often quite reactive and can be found in things like natural gas (though usually in slightly more complex forms) and are crucial intermediates in many chemical processes. They are the sprinters of the molecular world, quick and to the point. They’re not afraid to go all-in on a bond, making for some seriously potent chemistry.
So, How Do We Know Which is Which?
Here's the fun part, the detective work! You just need to look at how many other atoms a carbon is directly bonded to.
- If a carbon is bonded to four other atoms (all single bonds), it's sp³. Think of a carbon surrounded by four little friends.
- If a carbon is bonded to three other atoms (one double bond and two single bonds), it's sp². Think of a carbon making a double bond with one friend and single bonds with two others.
- If a carbon is bonded to two other atoms (either one triple bond and one single bond, or two double bonds), it's sp. Think of a carbon that’s super focused, making a triple bond with one friend and a single bond with another, or two double bonds with two separate friends.
It’s like a simple counting game, but the stakes are the very structure of the universe! Seriously, these little hybridization tricks are what allow carbon to form the incredible diversity of molecules that make up life, from the DNA in your cells to the caffeine in your coffee. So next time you see a carbon atom, give it a little mental nod. It's not just a boring old building block; it's a shape-shifting, bond-loving marvel, rocking its hybridization like a true star.
And there you have it! Carbon atoms, not so scary after all, right? They're just dressing for the occasion, ready to bond and build the amazing world around us. Now, who needs another coffee? All this talk of electrons has made me thirsty!
