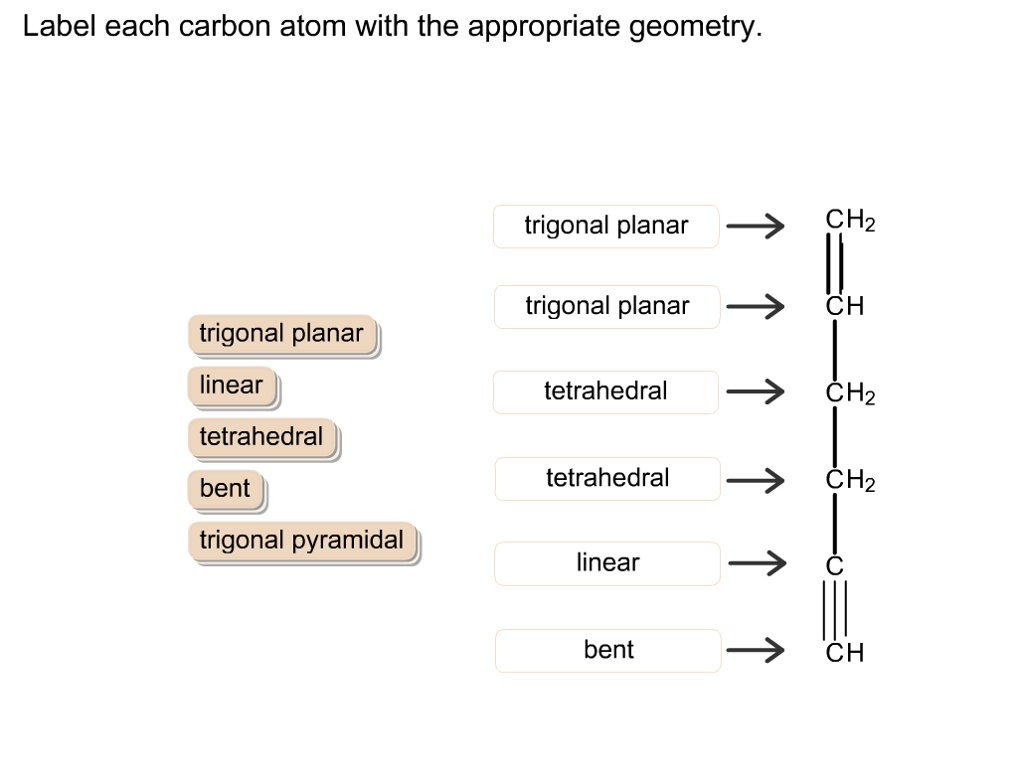
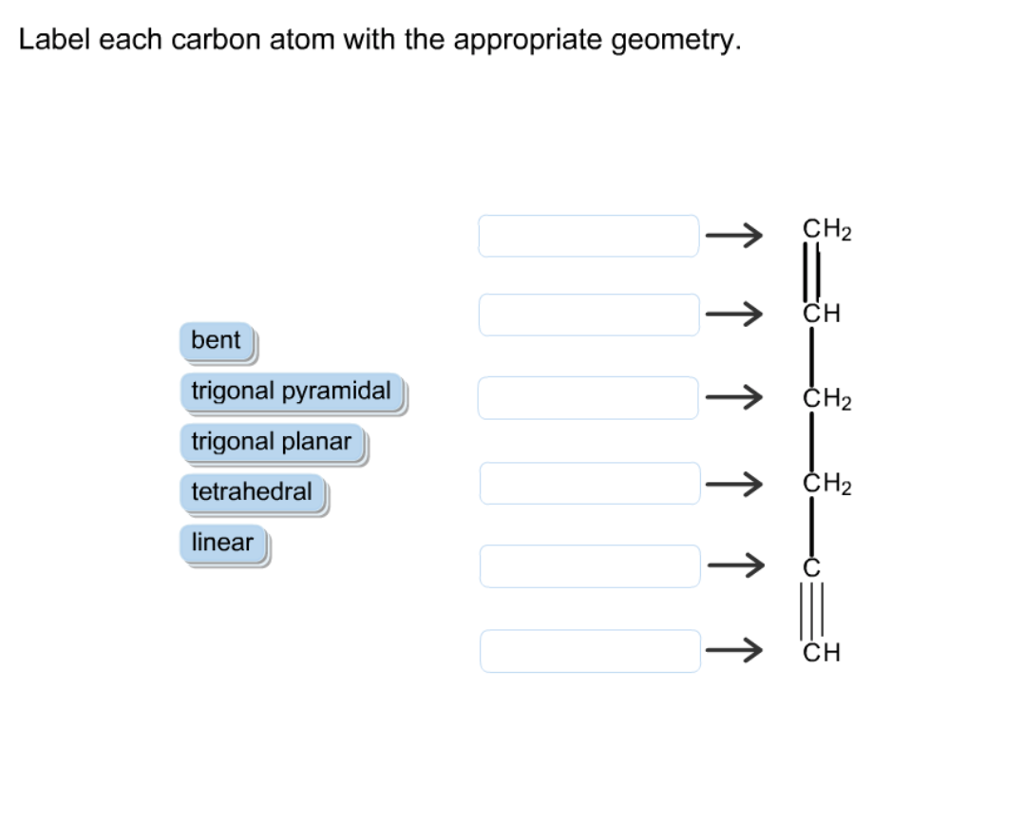
Label Each Carbon Atom With The Appropriate Geometry
Imagine you're at a big family reunion, and everyone has a little name tag on. Well, in the wild world of chemistry, those amazing carbon atoms, the building blocks of pretty much everything we can see and touch – from the pizza you ate last night to the air you're breathing right now – also get their own special "name tags." But instead of saying "Hi, I'm Sarah, I like gardening!", these tags tell us about their shape, their geometry. It's like giving each carbon atom its own personality trait based on how it's hanging out with its friends!
Think about it: a carbon atom is like a super friendly guy who loves to make friends. It usually wants to hold hands with four other atoms. Now, how those hands are arranged is where the magic, and the geometry, comes in. It’s not just random; there are specific ways these connections happen, and it makes a huge difference to the overall shape of the molecule, kind of like how the arrangement of furniture in a room can totally change the vibe.
Let’s meet our first carbon character. This one is a real straight shooter. It likes to keep things nice and orderly, all in a line. When a carbon atom is holding hands with two other atoms in a particular way, forming a straight-as-an-arrow line, we call its geometry linear. Think of it like a perfectly aligned row of dominoes, all set up to fall. These carbons are the backbone of long, skinny molecules, and they're super important in things like the gases we exhale, like carbon dioxide. Not the most exciting shape, perhaps, but undeniably essential!
Then we have the more relaxed, spread-out carbon. This one prefers to make three friends, and it arranges itself like a little flat triangle, all spread out evenly. This is called trigonal planar geometry. Imagine three friends sitting around a perfectly round table, each with an equal amount of space. This kind of carbon is often found in molecules that have a bit of a double bond going on, like in the delicious fats that make your cookies so wonderfully chewy. It's a bit more dynamic than the linear carbon, a little more social, if you will.
But the real party animal of the carbon world has to be the one that goes for the full four friends, and arranges them in a super neat, almost pyramid-like shape, but with a flat base. This is our tetrahedral carbon. Think of a perfectly formed diamond, or a cute little four-sided pyramid. Each of its hands is pointing in a slightly different direction, spreading out as much as possible to avoid bumping into its neighbors. This is the most common type of carbon geometry, and it’s responsible for the incredible diversity of life on Earth. From the DNA in your cells to the protein in your muscles, these tetrahedral carbons are holding everything together in a wonderfully intricate 3D dance. It’s like the carbon atom saying, "Let's give everyone some personal space and reach for the stars!"
It’s truly amazing how these simple geometric arrangements can lead to such complex and varied substances. It’s not just about what atoms are present, but how they are positioned relative to each other. A slight change in geometry can be the difference between a molecule that’s good for your health and one that’s… well, not so good. It’s like how the same set of LEGO bricks can be used to build a simple tower or a magnificent castle, all depending on how you connect them.
So, the next time you’re looking at something, anything at all, take a moment to think about the carbon atoms inside. They’re not just passive participants; they’re actively shaping the world around you with their inherent geometry. They’re the silent architects, the invisible artists, constantly building and rebuilding the universe, one perfectly arranged connection at a time. It’s a little bit like a secret code, a hidden language that tells us about the very nature of matter. And understanding this "shape language" helps scientists not only understand what things are made of but also how they work, how they react, and even how to create new materials with incredible properties. It’s a reminder that even the smallest, seemingly insignificant details can have a profound impact on the grand scheme of things. The humble carbon atom, with its geometric flair, is a true marvel, a testament to the elegance and ingenuity of nature. And it’s all happening, all the time, right under our noses!
