Kinetic Energy Of Gas Molecules Is Directly Proportional To
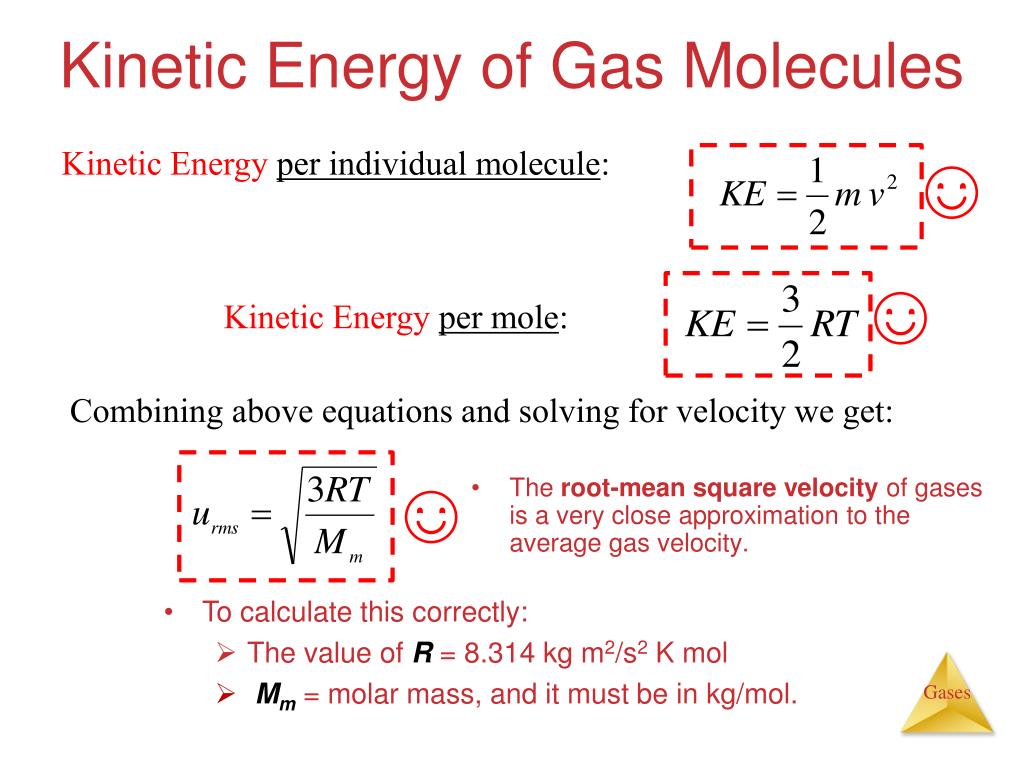
Imagine you're at a party. You've got a bunch of people, right? Some are chatting quietly in a corner, others are dancing like crazy, and a few are pacing around, looking for snacks. It's a bit chaotic, but that's what makes it fun! Now, think about the air around you. It’s not just empty space. It's packed with tiny, invisible things called gas molecules.
These little guys are always on the move. They're bumping into each other, bouncing off walls, and generally zipping around like a swarm of excited gnats. This constant dance of movement is what we call their kinetic energy. And guess what? There's a super cool secret about this kinetic energy, and it’s tied to something we all understand: temperature.
Here's the mind-blowing part: the kinetic energy of gas molecules is directly proportional to... wait for it... the temperature! Yep, it's as simple and as fascinating as that. Think of it like this: the hotter it gets, the more energetic our little gas molecules become. They start dancing faster, bumping into each other with more gusto, and basically throwing a bigger party.
So, when you feel a hot day, it's not just the sun warming you up. It's also the air molecules around you getting a serious energy boost. They're not just sitting there; they're vibrating, spinning, and zooming around at incredible speeds. It’s like they’ve had too much sugary soda and are now bouncing off the walls!
Conversely, on a chilly day, those same gas molecules are moving a lot slower. They’re more like folks having a leisurely stroll, not a full-on rave. Their kinetic energy is lower, and they’re not bumping into things as hard. It’s a much calmer, quieter gathering of molecules.
This direct proportionality is a fundamental concept in science, and it’s surprisingly relatable. It explains so many everyday phenomena. Ever wondered why a balloon expands when it gets warmer? It's because the gas molecules inside are getting more kinetic energy and pushing outwards more forcefully. They're basically saying, "Whoa, it's getting crowded in here, let's spread out!"
And what about that satisfying hiss when you open a can of soda? That's the gas molecules, full of bouncy energy, eager to escape their confined space. They’ve been cooped up, and the warmer they are, the more they want to break free and join the wider party.

It's this invisible world of energetic particles that makes gases so dynamic and interesting. They're not static things; they're alive with motion. And the temperature is like the conductor of their orchestra, dictating the tempo of their dance.
What makes this so special? Well, it’s the idea that something as simple as feeling warm or cold is directly linked to the frantic, invisible jig happening all around us. It’s a constant reminder that even the air we breathe is a bustling metropolis of activity.
This concept, that kinetic energy of gas molecules is directly proportional to temperature, is a cornerstone of understanding how gases behave. It’s the secret sauce behind everything from weather patterns to how engines work. It’s a bit like knowing the secret handshake to unlock a whole world of scientific understanding.
Think about it: the next time you feel the warmth of the sun, you can picture those tiny air molecules around you, energized and dancing their little hearts out. Or when you shiver, you can imagine them huddled together, moving at a more leisurely pace.
It's this inherent liveliness of gases that makes them so captivating. They’re not just passive elements; they’re active participants in the world. And their energy, their very essence, is tied to something as fundamental as heat.

It's a simple relationship, but its implications are vast. It's a little spark of understanding that can ignite a curiosity for more. It's the kind of scientific tidbit that makes you nod your head and think, "Wow, that's actually really cool!"
So, the next time you're thinking about gases, don't just think of them as invisible stuff. Think of them as tiny, energetic dancers, their every move dictated by the temperature. It’s a party that’s always happening, and we’re all invited!
And remember, that little phrase – kinetic energy of gas molecules is directly proportional to temperature – is the key. It’s the magic spell that explains so much of the unseen world around us. It’s a simple, elegant truth that reveals a universe of constant motion and energy.
It's a beautiful reminder that even the most ordinary things, like the air we breathe, are full of extraordinary activity, all thanks to the energetic dance of gas molecules.

Isn't that just wonderfully entertaining?
The hotter it gets, the more energetic our little gas molecules become. They start dancing faster, bumping into each other with more gusto, and basically throwing a bigger party.
This understanding is like having a secret decoder ring for the physical world. It allows you to look at a cup of steaming tea and instantly grasp the energetic chaos happening within. It's not just hot water; it's a whirlwind of excited water vapor molecules!
And that feeling of a cool breeze on a hot day? It’s the result of those less energetic molecules doing their best to cool things down, a constant effort to reach a balance. It’s a never-ending tug-of-war between hot and cold, all played out by these microscopic movers and shakers.
The beauty lies in its simplicity. No complex formulas or abstract theories are needed to grasp the core idea. Just a simple, direct link between something we can easily feel – temperature – and the unseen world of molecular motion.
This is what makes science so delightful. It's about finding these elegant, underlying principles that explain the world around us. And in the case of gases, it’s a constant, energetic ballet.

So, go ahead, feel the warmth, feel the chill, and know that you're experiencing the grand molecular dance. It’s a performance that’s always on, and the ticket is simply your curiosity.
And remember the main act: kinetic energy of gas molecules is directly proportional to temperature. It's a catchy tune, and once you hear it, you'll start noticing its rhythm everywhere.
It's a fundamental truth, a guiding star in the realm of gases. It's the reason for so much of the dynamism we observe in our world.
It's special because it connects the macro to the micro, the observable to the invisible, in such a clear and compelling way. It's a fundamental building block of our understanding of the universe.
So, embrace the heat, embrace the cold, and appreciate the unseen, energetic world that makes it all happen!
