Is The Autoionization Of Water Endothermic Or Exothermic

Hey, you! Yeah, you, the one staring at that suspiciously clear glass of water. Ever stop and think about what's really going on in there? Like, beyond just being wet and refreshing, you know?
So, water. H2O. We all know the basics. Two hydrogen atoms, one oxygen. Simple, right? But what if I told you this humble liquid is actually a bit of a drama queen? It's constantly doing this little dance, this... autoionization thing. Sounds fancy, I know. Basically, a few water molecules are like, "You know what? Let's shake things up!"
They do this thing where one water molecule steals a hydrogen ion (a proton, if you wanna get technical) from another. Poof! You get a hydronium ion (H3O+) and a hydroxide ion (OH-). Pretty wild, huh? It's like a molecular breakup and makeup, all in one go. And get this, it happens all the time. Billions and billions of these little H2O drama queens are busy at work in your average glass of water.
Now, for the million-dollar question, or at least the coffee-and-a-pastry question: is this whole autoionization shindig an endothermic affair, or is it exothermic? Let’s break it down, shall we? Because understanding this is kinda like knowing the secret handshake for understanding a whole lot of chemistry. No biggie.
Endo vs. Exo: The Energy Showdown
Okay, so we've got our two main players in the energy game: endothermic and exothermic. Think of it like this. Endothermic means "inward heat." It’s like a plant needing sunlight to grow. It has to take in energy from its surroundings to make the magic happen. So, an endothermic process needs energy to go. If you’re trying to make it happen, you gotta give it some juice. It gets colder around it because it’s sucking up all that warmth. Brrr.
Then there’s exothermic. This one's the opposite. "Exo" means "outward." So, exothermic processes release energy. Think of a campfire. It gives off heat and light, right? It’s like a generous friend who’s always giving stuff away. These reactions tend to make their surroundings warmer. Hot stuff!
So, is water's little ionization party an energy-guzzler (endo) or an energy-giver (exo)? This is where it gets interesting.
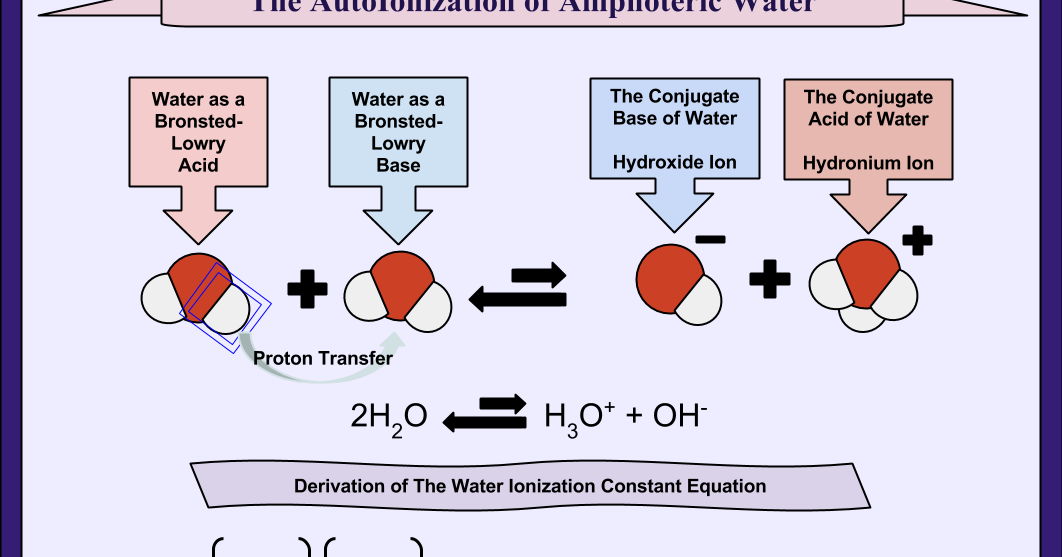
The Case for Endothermic
When water molecules decide to go their separate ways and form those ions, it’s not exactly a walk in the park. Breaking bonds, man. That’s tough work! Think about trying to snap a sturdy twig. You gotta put some effort into it, right? You gotta exert force. That force is, in a way, energy.
So, to break apart those H2O molecules and form the H3O+ and OH- ions, a little bit of energy has to be put in. It’s like a tiny investment. The universe has to cough up some energy to get this reaction started and keep it going. This is the argument for the autoionization of water being endothermic.
Scientists have actually measured this. They’ve looked at how the equilibrium of this reaction changes with temperature. And guess what they found? When you heat up water, this autoionization reaction shifts a bit more towards forming those ions. That’s a classic sign of an endothermic process. If you add heat, the reaction goes, trying to use up that extra heat. It’s like, "Ooh, thanks for the energy boost! I can make more ions now!"
It’s a small effect, don't get me wrong. We’re not talking about your water turning into a miniature ice pack or a boiling inferno just from this. But the tiny shift is there. The evidence points towards needing that energy input.
But Wait, There's a Twist!
Now, here’s where things get a little cheeky. While the overall process of autoionization is considered endothermic, it's not as simple as just breaking bonds. There’s another step involved, and this is where things get a bit more nuanced. And honestly, it's what makes chemistry so darn fun!

Remember how we said one water molecule gives a hydrogen to another? Well, that doesn't just happen in the void. The ions that are formed, H3O+ and OH-, don't just float around on their own. They interact with other water molecules.
The hydronium ion (H3O+), that’s like a positively charged proton hanging out with three water molecules. It’s actually stabilized by these water molecules. They sort of hug it, making it less… chaotic. And the hydroxide ion (OH-)? It’s got that negative charge, and it also gets cozy with other water molecules. They're like little water molecule blankets.
And guess what? The formation of these ion-water clusters, the solvation of the ions, that part actually releases energy. It’s like a little burst of warmth when the ions find their watery friends. This solvation process is actually exothermic.
So, What's the Verdict?
This is where your brain might start doing a little somersault. If breaking bonds is endothermic, and forming those stabilized ion-water things is exothermic, what gives? Which one wins the energy battle? Ah, the eternal question!
Well, it turns out that the energy needed to break the bonds in the water molecules to initiate the ionization is slightly greater than the energy released when the resulting ions are solvated by other water molecules.
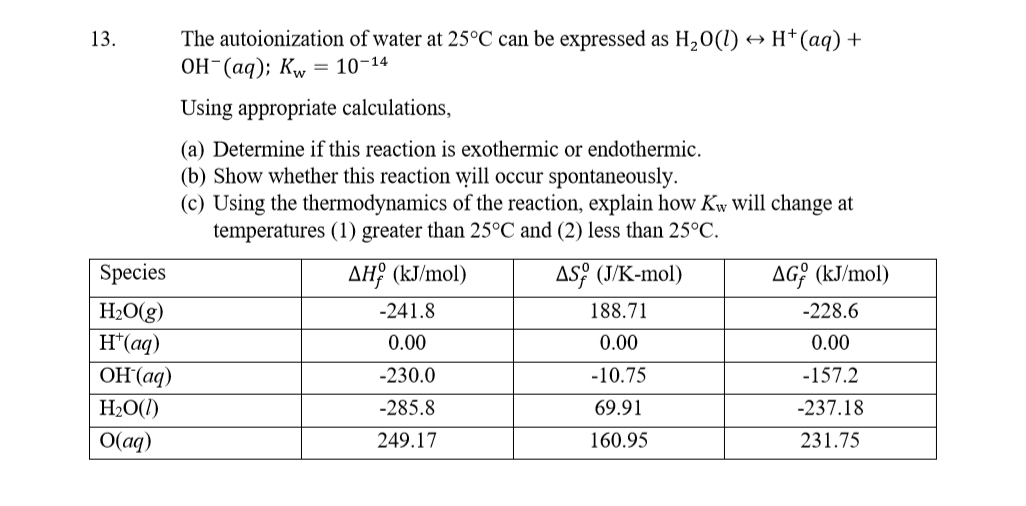
So, the net effect, the overall energy change for the autoionization of water, is that it requires a small amount of energy. It’s a net endothermic process. It’s like you pay a little bit for the initial separation, and then you get a little bit back from the bonding, but the initial payment was just a smidge higher. You’re still down a tiny bit of energy.
Think of it like this: you’re trying to start a business. You have to invest money to get it off the ground (endothermic). Then, once it's running, it starts making some money back (exothermic). If your initial investment is a tiny bit more than what you make back immediately, your business is technically still in a state where it needed that initial capital infusion to get going. It's not a "money-making machine" from minute one.
The enthalpy change for the autoionization of water (often symbolized as ΔH) is a small positive value. Positive ΔH? That's the chemist's way of saying "endothermic." It's a tiny positive value, mind you. We're talking maybe around +55 to +57 kilojoules per mole, depending on the exact conditions and how you measure it. That's not a huge amount of energy. It won't make your glass of water spontaneously freeze over, but it's there.
Why Does This Even Matter?
Okay, so why should you care about this subtle energy dance within your water? Because it has ripple effects, my friend! This endothermic nature of water autoionization is actually a big deal for a lot of chemical and biological processes.
For starters, it explains why the pH of water changes with temperature. Remember pH? That scale that tells you if something is acidic or basic? Well, the concentration of H+ ions (and OH- ions) in pure water is directly related to this autoionization process. Since the process is endothermic, heating water increases the extent of ionization. More ions mean a slightly different pH. So, that pure water you're sipping? Its acidity is actually a little more pronounced when it's warm than when it's cold. Mind. Blown.

This temperature dependence is super important in things like industrial processes, and even in how enzymes and other biological molecules behave. Life itself, you know, happens in water! And water's behavior, even at this fundamental level, plays a crucial role.
Imagine trying to run a reaction in a beaker. If you’re heating it up, you’re not just speeding up the reaction; you might also be subtly changing the fundamental properties of the water solvent itself because of this endothermic ionization. It’s a whole domino effect!
The Little Things Matter
So, the next time you're enjoying a glass of water, give it a little nod of appreciation. It’s not just sitting there being bland. It’s a dynamic, energetic little universe. And that little bit of energy it needs to keep its ions in play? That endothermic kick? It’s a testament to the intricate balance of forces that make our world work.
It’s a reminder that even the most seemingly simple things, like water, have layers of complexity. And understanding those layers? That’s where the real fun of science lies. So yeah, endothermic. The autoionization of water. Who knew that clear liquid could be so… energetically involved?
It’s the little things, you know? The stuff you don’t always see. The subtle energy changes. The molecular breakups and makeups. It all adds up. It makes water, well, water. And it makes everything else in the universe possible. Pretty cool, right? Now, about that coffee refill…
