Is Si A Molecular Metallic Ionic Or Covalent-network Solid

Ever found yourself staring at the periodic table, wondering what makes one element behave so differently from another? It's like a cosmic casting call, and each element gets assigned a specific role in the grand drama of chemistry. Today, we're pulling back the curtain on one particularly intriguing character: Silicon (Si). Is it a flashy rockstar in the world of metallic bonding, a delicate dancer in the realm of covalent networks, or perhaps a bit of a chameleon, showing traits of something else entirely? The answer to whether Si is a molecular, metallic, ionic, or covalent-network solid isn't just an academic exercise; it's key to understanding why our smartphones are so smart, why solar panels harness the sun's energy, and a whole lot more!
Understanding the bonding behavior of elements like Silicon is super useful because it directly dictates their physical and chemical properties. Think about it: the difference between a brittle salt crystal (ionic) and a bendable piece of copper (metallic) is all down to how their atoms hold hands. For Si, this understanding unlocks its incredible versatility. The world of electronics, from the tiny transistors in your computer to the large solar arrays powering our cities, relies heavily on the unique semiconductor properties of Silicon. These properties, in turn, are a direct consequence of its specific solid-state structure. So, when we ask, "Is Si molecular, metallic, ionic, or covalent-network solid?", we're really asking about the building blocks of modern technology!
So, What Kind of Solid is Silicon?
Let's break down the contenders. We have molecular solids, where discrete molecules are held together by weaker forces. Think of ice – water molecules (H₂O) sticking together. Then there are metallic solids, like gold or iron, where electrons are delocalized and flow freely, giving them their characteristic shine and conductivity. Ionic solids, like table salt (NaCl), are formed by the electrostatic attraction between positively and negatively charged ions. Finally, we have covalent-network solids, a super strong category where atoms are linked by a continuous network of strong covalent bonds. Diamond is the classic example here, with its incredibly rigid, three-dimensional structure.
Now, where does Silicon (Si) fit in? Looking at its position on the periodic table, Silicon is in Group 14, right below carbon. Carbon, in its diamond form, is the epitome of a covalent-network solid. This proximity already gives us a big hint! Silicon, like carbon, has four valence electrons, which it loves to share to form strong covalent bonds.
In its most common and stable solid form, elemental Silicon exists as a covalent-network solid. Each Silicon atom is covalently bonded to four other Silicon atoms in a tetrahedral arrangement, forming an extended, rigid, three-dimensional network. This is very similar to the structure of diamond, though the Si-Si bonds are slightly longer and weaker than the C-C bonds in diamond.
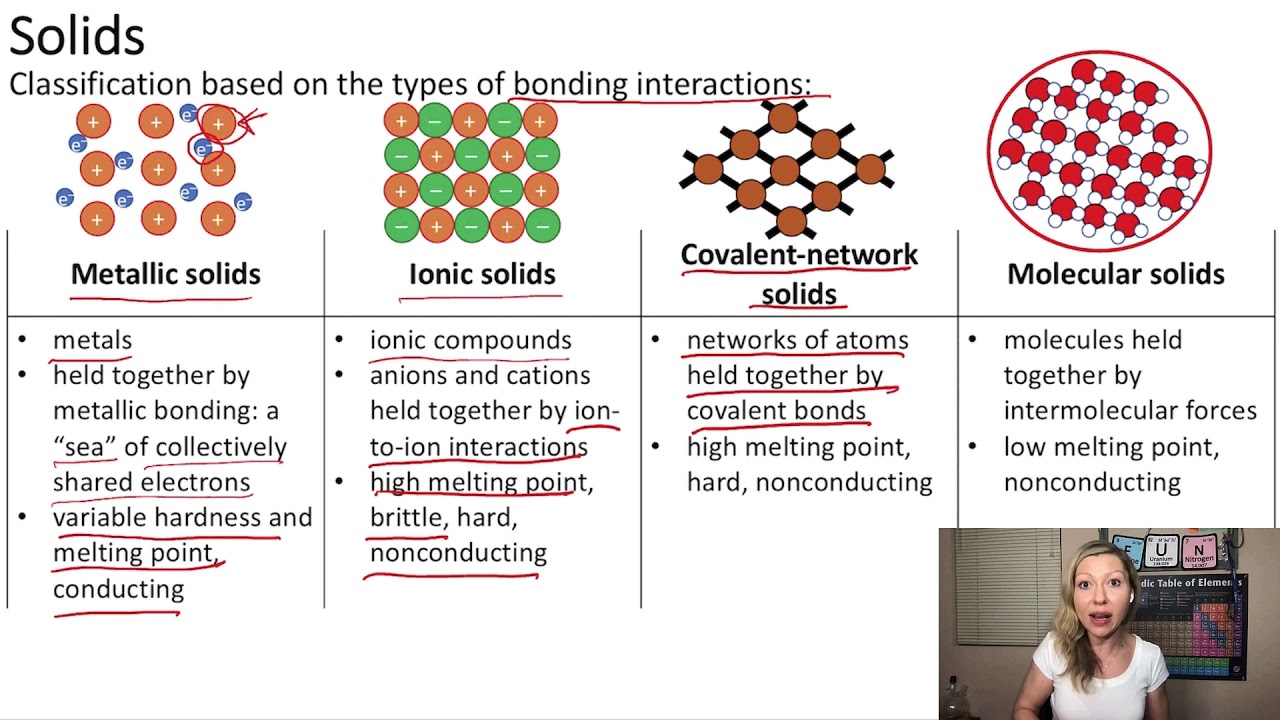
This covalent-network structure is responsible for many of Silicon's key properties. It's very hard and brittle, just like diamond. It also has a very high melting point because a lot of energy is required to break these strong covalent bonds. But here's where Silicon gets really interesting and sets itself apart from pure diamond:
Unlike diamond, which is an electrical insulator, Silicon is a semiconductor. This means it can conduct electricity, but not as well as a metal. This unique property arises from the specific arrangement of its electrons within the covalent-network structure. At absolute zero temperature, Silicon behaves like an insulator. However, as the temperature rises, or when impurities (called dopants) are added, some electrons gain enough energy to break free from their covalent bonds and move through the crystal lattice, allowing for electrical conductivity. This ability to control conductivity by adding impurities or changing temperature is the magic behind all semiconductor devices!

So, to directly answer the question: Silicon (Si) is best classified as a covalent-network solid. It doesn't form discrete molecules, so it's not a molecular solid. While it has some metallic characteristics like being a solid at room temperature, its bonding is predominantly covalent, not metallic. And it's definitely not an ionic solid; there's no significant transfer of electrons to form ions.
The fact that Silicon is a covalent-network solid with semiconductor properties is a happy accident of nature that has revolutionized our world. From the very first transistors to the latest processors and memory chips, Silicon has been the workhorse of the digital age. So, the next time you're using your phone, browsing the internet, or enjoying a solar-powered gadget, remember the humble Silicon atom and its incredible covalent-network structure!
