Is Seawater A Homogeneous Or Heterogeneous Mixture
Ever gaze out at the vast, shimmering ocean and wonder about its secrets? Beyond the waves and the salty air, there's a little bit of science happening right before your eyes! Today, we're diving into a fun and surprisingly useful question: is seawater a homogeneous or heterogeneous mixture? It might sound like something from a chemistry textbook, but understanding this helps us appreciate the world around us, from the smallest tide pool to the mightiest ocean currents. Plus, it’s a great way to spark curiosity, especially for families and budding scientists!
For beginners, this is a fantastic entry point into the world of mixtures. You’ll learn the difference between things that are perfectly blended and things that you can still see the individual parts of. Families can use this as a springboard for fun experiments, like comparing saltwater to a salad – one is mixed, the other clearly isn't! Hobbyists, whether you're into aquariums, fishing, or even just collecting seashells, knowing about the composition of seawater can offer deeper insights into the environments you love. Imagine understanding why certain fish thrive in specific areas – it all relates back to what’s dissolved in the water!
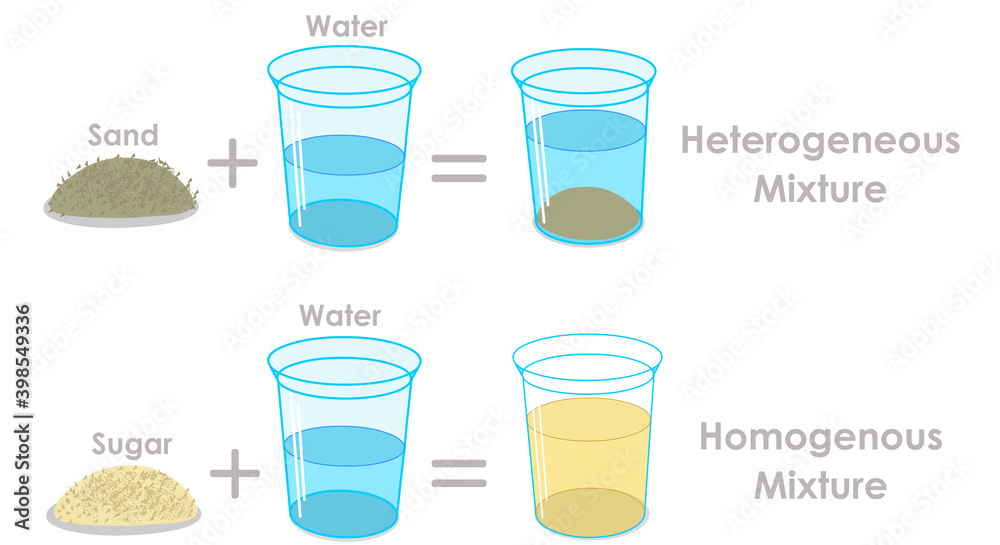
So, what exactly is the answer? Drumroll please… Seawater is considered a homogeneous mixture. This means that its components are uniformly distributed throughout. Think of it like dissolving sugar in water. Once it's all mixed in, you can't see the individual sugar crystals anymore, and every sip of the sugary water tastes the same. Seawater is packed with all sorts of dissolved salts, minerals, and gases – like sodium chloride (your common table salt), magnesium, calcium, potassium, and oxygen. These are so finely dispersed that no matter where you sample the ocean, the proportion of these dissolved substances remains remarkably consistent. You can't pick out individual salt grains with your eyes!
But wait, you might be thinking, "What about the little bits of sand or shells I see on the beach?" Ah, that’s a great observation! Those are actually separate heterogeneous components that are not part of the seawater mixture itself. A heterogeneous mixture is one where you can clearly see the different parts, like sand in water or oil and vinegar in salad dressing. So, while the ocean floor might have heterogeneous elements, the water itself, in its pure form, is wonderfully homogeneous.

Getting started with this concept is super easy. The next time you're at the beach, take a moment to observe. Think about the water itself versus the sand and pebbles. For a simple home experiment, try dissolving salt in a glass of tap water. Stir it well. Does it look clear? Does it taste uniformly salty? That's a great example of a homogeneous mixture, just like the ocean! You could even try adding some sand to another glass of water and see how it settles – that's your heterogeneous example.
Understanding whether seawater is homogeneous or heterogeneous isn't just about memorizing facts; it's about developing a keener eye for the world around you. It’s a simple concept with profound implications, reminding us of the intricate and beautifully blended nature of our planet's vast oceans. So next time you’re near the sea, you’ll have a little extra scientific magic to ponder!
