Iron Iii Sulfate And Sodium Hydroxide Balanced Equation
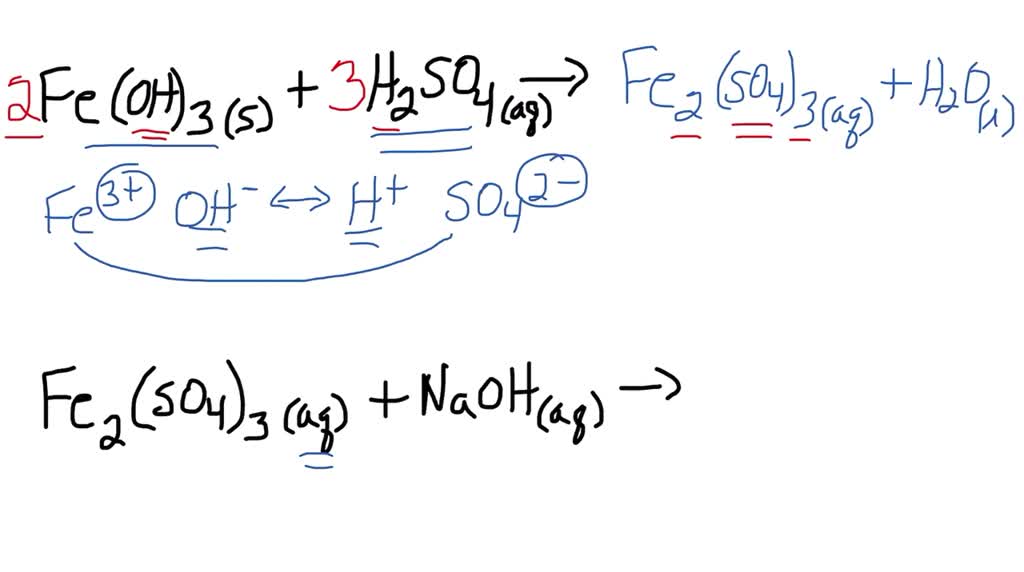
Hey there, science enthusiasts and curious minds! Ever feel like chemistry is this big, scary monster lurking in the shadows of textbooks? Well, I'm here to tell you it's more like a friendly neighborhood wizard, just waiting to sprinkle a little magic into your everyday life! And today, we're going to pull back the curtain on a seemingly dry topic that's actually got a surprising amount of pizzazz: the balanced chemical equation for iron(III) sulfate and sodium hydroxide. Sounds intimidating, right? Trust me, by the end of this, you'll be thinking, "Wow, that's actually kinda cool!"
So, what exactly are these fancy-sounding chemicals? Let's break it down. We've got Iron(III) sulfate. Think of iron as that strong, sturdy metal we see everywhere. The "(III)" just tells us it's in a particular, rather energetic, state. And sulfate? That's just a group of oxygen and sulfur atoms hanging out together. Easy peasy, lemon squeezy, right?
Then we have Sodium hydroxide. You might have heard of sodium – it's in table salt! And hydroxide? That's a little duo of oxygen and hydrogen. Sodium hydroxide is actually super useful. It’s a strong base, meaning it likes to make things… well, basic! Think of it as the opposite of an acid. It's used in everything from making soap to cleaning drains. Pretty neat, huh?
Now, what happens when these two pals meet? When iron(III) sulfate and sodium hydroxide get together, it's not a wild party with explosions (thank goodness!), but rather a precise and elegant dance of molecules. They react to form something new. And this is where the balanced chemical equation comes in. It’s like the recipe or the choreography for this molecular ballet.
Why do we need a balanced equation, you ask? Ah, that's the secret sauce of chemistry! It all comes down to a fundamental law of nature: the law of conservation of mass. Basically, what goes in must come out. You can't create matter out of thin air, and you can't make it disappear. So, our equation needs to show that we have the exact same number of each type of atom on both sides of the reaction.

Let's get to the nitty-gritty of the equation itself. On one side, we have our reactants: iron(III) sulfate and sodium hydroxide. On the other side, we have our products – what's formed after the reaction. When iron(III) sulfate and sodium hydroxide mix, they create iron(III) hydroxide and sodium sulfate.
So, the unbalanced equation might look something like this, just to get our bearings: Fe₂(SO₄)₃ + NaOH → Fe(OH)₃ + Na₂SO₄. See? It's like a puzzle where the pieces aren't quite lining up yet. We have two iron atoms on the left but only one on the right. That’s not balanced!
This is where we get to play chemist and adjust those little numbers, called coefficients, in front of each chemical formula. It's like adding more ingredients to your recipe to make sure everything is just right. We want to ensure that for every iron atom we start with, we end up with one iron atom. Same for sulfur, oxygen, hydrogen, and sodium.

Let's tackle the iron first. We have two iron atoms in Fe₂(SO₄)₃. To get two iron atoms on the product side, we need to put a '2' in front of Fe(OH)₃. So now we have: Fe₂(SO₄)₃ + NaOH → 2Fe(OH)₃ + Na₂SO₄.
Now let’s look at the sulfate (SO₄) group. We have three sulfate groups on the reactant side. On the product side, we currently only have one in Na₂SO₄. To balance the sulfates, we need to put a '3' in front of Na₂SO₄. Our equation is shaping up: Fe₂(SO₄)₃ + NaOH → 2Fe(OH)₃ + 3Na₂SO₄.
Almost there! Let's check out the sodium (Na). On the product side, with the '3' in front of Na₂SO₄, we now have a total of 3 * 2 = 6 sodium atoms. So, on the reactant side, we need to have 6 sodium atoms. To achieve this, we put a '6' in front of NaOH: Fe₂(SO₄)₃ + 6NaOH → 2Fe(OH)₃ + 3Na₂SO₄.
+sulfate+and+potassium+sulfide+react..jpg)
Finally, let’s do a quick check of the hydroxide (OH) groups. On the reactant side, with the '6' in front of NaOH, we have 6 hydroxide groups. On the product side, we have 2 * 3 = 6 hydroxide groups in 2Fe(OH)₃. And the oxygen and hydrogen atoms within the sulfate groups are already balanced because we balanced the whole sulfate group as one unit.
Ta-da! We have successfully balanced the equation! The final, glorious, balanced equation is: Fe₂(SO₄)₃ + 6NaOH → 2Fe(OH)₃ + 3Na₂SO₄.
Why should this make your life more fun, you ask? Because understanding this is like unlocking a secret code to the world around you! Think about it: this same principle of balancing equations is at play when your laundry detergent cleans your clothes, when batteries power your gadgets, or even when your body digests food. It’s the underlying language of how matter transforms!
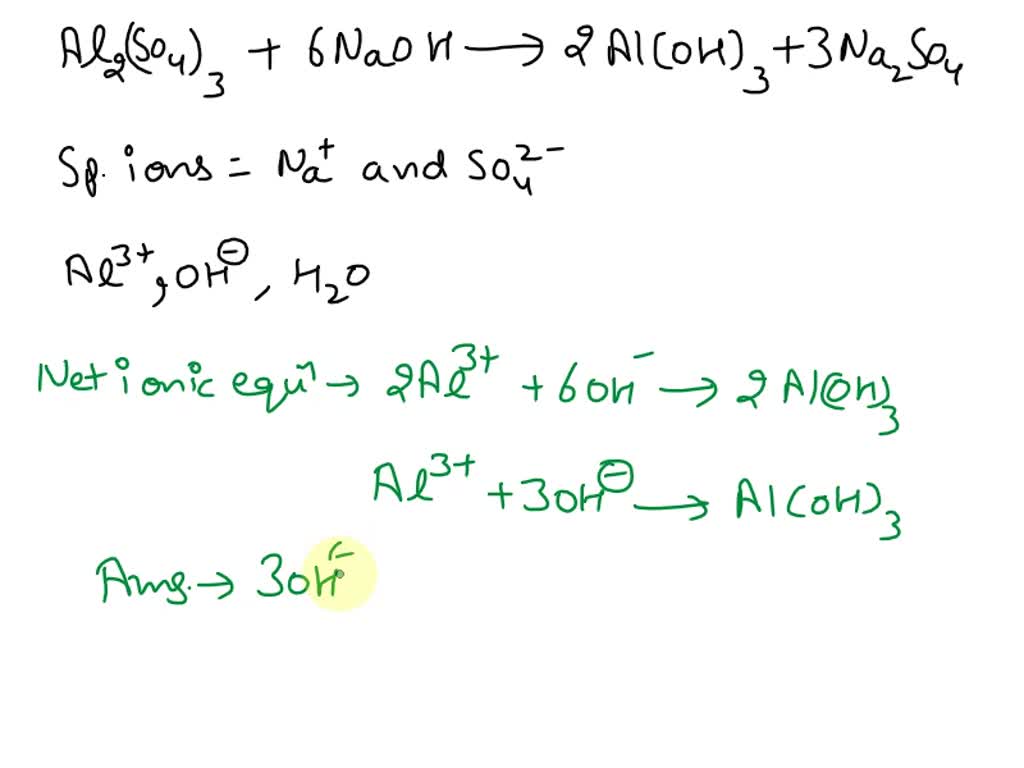
Knowing how to balance equations can make you a super-powered problem solver. It’s like having a superpower that allows you to predict what will happen when certain substances mix. Imagine being able to impress your friends with your newfound chemical knowledge or understanding the science behind your favorite DIY projects. It’s not just about memorizing formulas; it’s about grasping the fundamental order and elegance of the universe.
And the best part? This is just the tip of the iceberg! Chemistry is a vast and fascinating universe waiting to be explored. Each balanced equation is a tiny story of transformation, a testament to the predictable and beautiful laws that govern our physical world. It’s about seeing the magic in the mundane and realizing that even the most seemingly complex processes are built on simple, logical principles.
So, the next time you encounter a chemical equation, don't shy away from it. Embrace it! See it as a challenge, a puzzle, or even a friendly conversation between atoms. Because when you start to understand these basic building blocks, you start to understand so much more. Keep exploring, keep questioning, and keep that spark of curiosity alive. The world of science is an incredible adventure, and you're just getting started!
