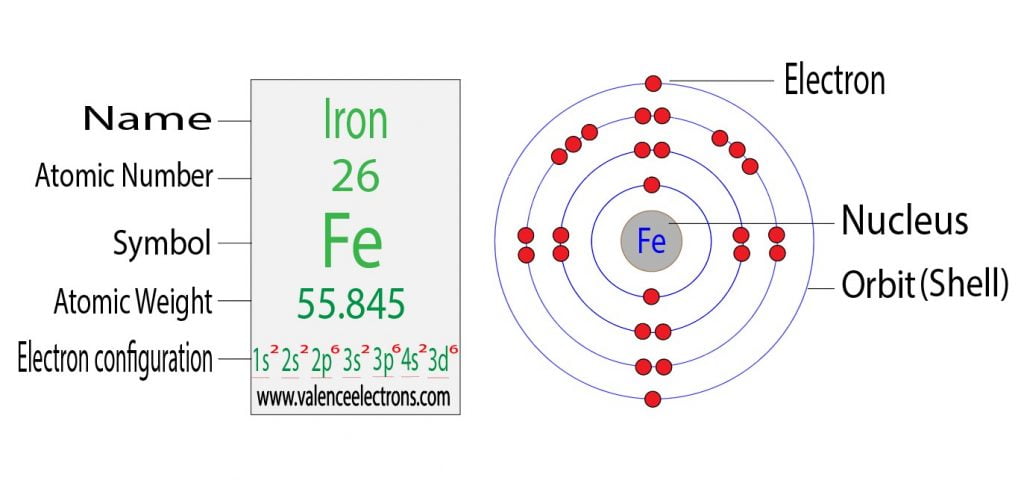
Iron Fe Ar 4s23d6 Core Electrons Valence Electrons

Hey there, science curious folks! Ever wonder what makes the world around us tick? We’re not talking about your toaster or your phone (though those are pretty cool too!). We’re diving deeper, into the super-tiny building blocks of everything. Today, we’re spotlighting a star of the show: Iron!
You know, the stuff that makes your blood red, your pans strong, and your magnets… well, magnetic! But the real magic of Iron, which we scientists often call Fe for short, isn't just what it does. It's about how it's put together. Think of it like a tiny, super-organized apartment building.
Every atom has a center, like the lobby of our apartment building. This is the nucleus. Then, around the nucleus, there are different floors where electrons hang out. These electrons are like the residents of our building. They zip around, each on their own designated floor or energy level.
Now, the awesome thing about Iron (Fe) is its unique setup. It’s got a bunch of floors, but let’s talk about the most important ones for our story. There’s a floor that’s pretty packed and cozy, and we call this the core electrons. These are the electrons that are snuggled up tight to the nucleus. They’re the long-term residents, the ones who have been there forever and don’t really interact with the outside world much. They’re like the folks who are happy to stay home all day, minding their own business.
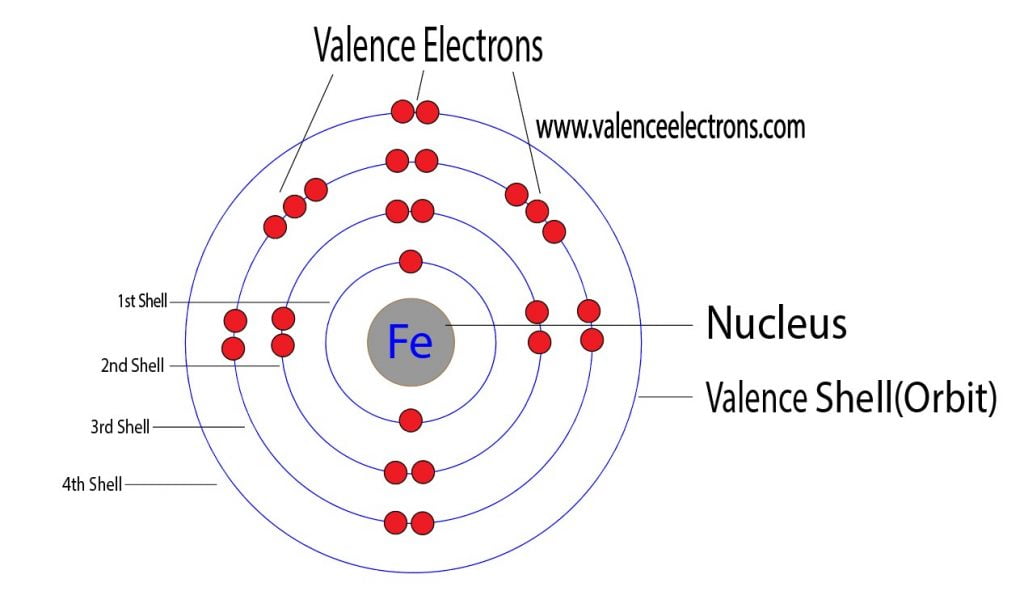
But then, there’s the really exciting part! The outer floors are where the action happens. These are the valence electrons. These are the cool kids, the ones who love to mingle and interact. They’re the ones who decide how Iron behaves, what it can stick to, and what kind of cool stuff it can help create. They’re the ones that make Iron so versatile and, frankly, so much fun to study!

For Iron (Fe), the valence electrons are particularly interesting. They’re not all on the same floor, which makes things even more dynamic. We’re talking about electrons chilling on the 4s level and then some very special ones on the 3d level. Imagine you have a few residents on the fourth floor (the 4s) and then a whole bunch of super-energetic residents on the third floor (the 3d). This arrangement is what gives Iron its superpowers.
The Ar part in the whole picture is like a shorthand for a previous, smaller apartment building that Iron is a bigger, more complex version of. Think of it as Iron inheriting some basic furniture and layout from a smaller, simpler place. The electrons represented by Ar are essentially the inner, core electrons. They’re already accounted for in this "inherited" setup.
So, when we talk about Iron (Fe) with its Ar 4s² 3d⁶ electron configuration, it's like saying: "Okay, we’ve got this base structure from Ar, then we’ve got 2 electrons on the 4s floor, and a whopping 6 electrons on the 3d floor." It’s this specific number and arrangement of electrons, especially those on the 3d floor, that makes Iron so special. It’s like having a secret stash of energy and potential!
Why is this so entertaining? Because it’s the key to understanding so much! These valence electrons, those on the 4s and 3d floors, are the ones that get involved in chemical reactions. They’re the ones that can be shared, swapped, or even lost. This ability to change how many electrons it has available makes Iron a master of disguise in the chemical world.
It can form strong bonds, like in steel, which is a mix of Iron and Carbon. That’s the valence electrons at work, holding things together. It can also lose or gain electrons to form different ions, which are basically Iron atoms with a different electrical charge. This is what happens when Iron rusts – it’s reacting with oxygen! The 3d electrons are particularly flexible, allowing Iron to be quite adaptable.
Think about the colors you see in fireworks or pigments. Often, the vibrant colors come from transition metals like Iron. Why? Because of those 3d electrons! When light hits them, they can absorb certain colors and reflect others, giving us those amazing hues. It's like they're playing a beautiful game of light with us.
This whole electron dance, the way they are arranged and how they interact, is what makes chemistry so endlessly fascinating. For Iron (Fe), the combination of its inner core electrons (represented by Ar) and its outer, active valence electrons (specifically the 4s² 3d⁶ configuration) gives it a unique personality. It’s strong, it’s versatile, and it’s essential to life as we know it.
So, next time you see a rusty bridge, admire a strong metal beam, or even just feel the pulse in your wrist, remember Iron (Fe) and its amazing electron apartment building. It’s a tiny world of wonder, and the way its electrons are arranged, especially those energetic 4s and 3d electrons, is a little piece of scientific magic that makes our world a much more interesting place. Isn't that just the coolest?
