Ions And Isotopes Practice Worksheet Answer Key

Hey there, fellow curious minds! Ever find yourself staring at a science worksheet, perhaps with the tantalizing promise of an "answer key" lurking just out of sight, and feel a vague sense of… something? Maybe it's the hum of the universe, or perhaps it's just the faint echo of a forgotten chemistry lesson. Whatever it is, today we’re going to dive into the wonderfully accessible world of ions and isotopes. Think of this not as a dry textbook chapter, but as a chill chat over a perfectly brewed cup of your favorite beverage, unraveling some of the fundamental building blocks of everything around us.
You know, it's a funny thing. We often think of science as this complex, intimidating beast, locked away in sterile labs with people in white coats doing… well, science. But at its heart, science is just about understanding the world. And the world is made up of tiny, fascinating things. Like atoms. And those atoms? They’re not always as straightforward as they seem. Sometimes they’re feeling a bit charged, and sometimes they’re just… slightly different versions of themselves. Enter ions and isotopes, our stars of the show today.
The Electrifying World of Ions
Let's kick things off with ions. Imagine an atom as a tiny, bustling city. In the center, you have the nucleus, the town hall, packed with protons (positive citizens) and neutrons (the neutral bystanders). Whizzing around the outskirts, in ever-expanding orbits, are the electrons (the energetic youngsters, always on the move and carrying a negative charge). Now, for an atom to be truly happy and stable, it usually likes to have an equal number of positive protons and negative electrons. It's like a perfectly balanced neighborhood watch committee.
But sometimes, things get a little… energetic. An atom might lose one or more of its electrons. Think of it as a teenager deciding they need more personal space and ditching some of their younger siblings (electrons) to go explore. When an atom loses negatively charged electrons, it ends up with more positive protons than negative electrons. And what happens when you have more positives than negatives? You get a positive ion, also known as a cation. It’s like the town hall suddenly having a surplus of positive energy. These guys are often found in things like table salt (sodium chloride) – that sodium atom lost an electron and became a happy little cation.
On the flip side, an atom can also gain electrons. This is like a busy household deciding to take in a few stray kittens (electrons). When an atom gains negatively charged electrons, it now has more negatives than positives. This results in a negative ion, or an anion. These are common in things like the chlorine in table salt, which readily scoops up an extra electron. So, you've got your cations and anions, always looking for a stable balance, sometimes forming beautiful crystalline structures like salt or, on a grander scale, contributing to the electric charges in our bodies.
Why should you care about these charged little guys? Well, ions are absolutely everywhere and do everything. They are the unsung heroes of our nervous system, allowing signals to zap from your brain to your toes. Ever felt that tingle when you touch a static-charged balloon? That's ions at play, baby! They’re also crucial for muscle contractions, maintaining fluid balance in your cells (think electrolytes in your sports drinks – yep, those are ions!), and even in the vibrant colors you see in fireworks. It’s a charged life, indeed!
For those of you tackling practice worksheets, identifying ions is usually about looking at the number of electrons compared to protons. If electrons > protons, it’s an anion (negative). If electrons < protons, it’s a cation (positive). Simple, right? It's like a little detective game. And the answer key? It’s your reward for cracking the case!
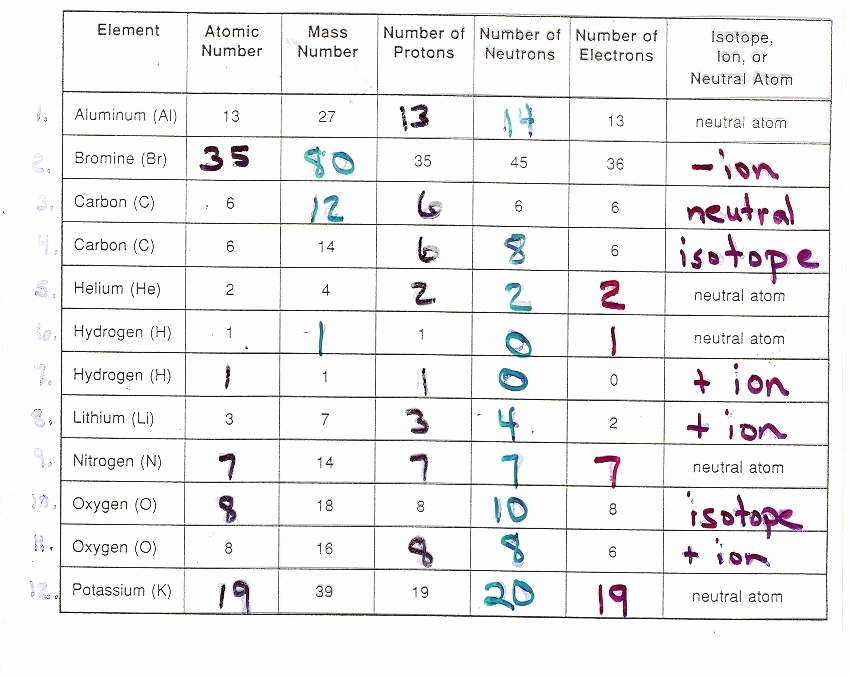
The Slightly-Different Siblings: Isotopes
Now, let’s shift gears and talk about isotopes. If ions are about charge, isotopes are about… a subtle variation on a theme. Imagine you have a family of identical twins. They look the same, they have the same core characteristics, but maybe one of them has a slightly different appetite, or a slightly different sleep schedule. Isotopes are like that for atoms. They are atoms of the same element – meaning they have the same number of protons (which defines the element, like the fingerprint of a chemical identity) – but they have a different number of neutrons in their nucleus.
Remember those neutrons we mentioned? They’re the neutral party in the nucleus, just hanging out. While the number of protons is fixed for a given element (e.g., every carbon atom has 6 protons), the number of neutrons can vary. So, you can have carbon-12 (6 protons, 6 neutrons) and carbon-14 (6 protons, 8 neutrons). They're both carbon, but one has two extra neutrons. This difference in the number of neutrons gives them slightly different masses. That’s why we call them isotopes – 'iso' meaning same, and 'tope' meaning place, so they occupy the same place on the periodic table but have different masses.
Why does this matter? Well, it turns out that these subtle differences can have huge implications. Some isotopes are stable, like regular water molecules. Others are radioactive, meaning their nucleus is a bit unstable and they tend to decay over time, emitting energy and particles. Think of it like a ticking clock that eventually runs out of wind.
The most famous example is probably carbon-14. This radioactive isotope of carbon is naturally present in living organisms. When an organism dies, it stops taking in new carbon, and the carbon-14 it contains starts to decay at a predictable rate. Scientists can measure the amount of carbon-14 remaining in an ancient artifact or fossil to determine its age. This is called radiocarbon dating, and it's revolutionized our understanding of history and archaeology. Imagine being able to tell if a mummy is thousands of years old just by looking at its carbon content! Pretty neat, huh?

Another cool application is in medicine. Radioactive isotopes are used in imaging techniques like PET scans to diagnose diseases, and in treatments for cancer. They can be precisely targeted to destroy cancer cells while minimizing damage to healthy tissue. So, while they might sound a bit abstract, isotopes are directly impacting our health and our ability to understand the past.
On your practice worksheets, identifying isotopes usually involves looking at the mass number, which is the sum of protons and neutrons. If you're given an element name and a mass number (like "Carbon-14"), you know the protons (from the element name) and can calculate the neutrons. Or, you might be given the number of protons and neutrons and asked to identify the isotope. It’s all about that neutron count making the difference!
The Answer Key: Your Friendly Guide
Let's talk about that elusive "answer key." Think of it not as a shortcut to avoid learning, but as a trusty sidekick on your learning adventure. It’s the experienced friend who’s already navigated the tricky parts and can offer a guiding hand. When you’re working through your ions and isotopes practice, reaching for the answer key is like checking your GPS on a road trip – it helps you confirm you’re on the right path and points out any scenic detours you might have missed.
The best way to use an answer key is to try the problems yourself first. Really wrestle with them! Make your best guess. Then, use the key to check your work. If you got it right, awesome! High five yourself. If you got it wrong, don't despair. Instead, look at the correct answer and try to understand why it’s correct. What did you miss? Was it a simple calculation error, or a misunderstanding of a concept? The answer key, when used thoughtfully, becomes a powerful learning tool, helping you to identify your strengths and weaknesses.
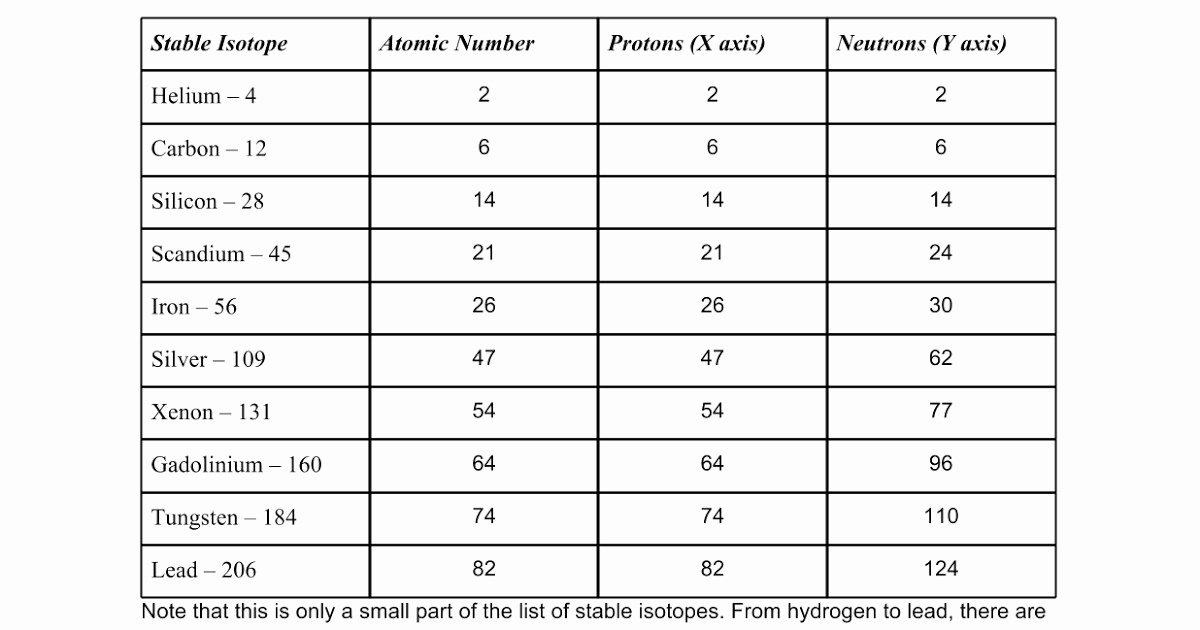
Imagine you’re learning to bake. You follow a recipe, taste your creation, and then compare it to a picture in a cookbook. If it looks and tastes a little off, you check the recipe again, maybe looking at the picture for clues. The answer key is that cookbook picture – it shows you the intended outcome and helps you adjust your technique. It’s all about building that intuition and confidence.
Fun Facts and Cultural Touches
Did you know that the element with the most commonly known isotopes is Hydrogen? It has three main ones: Protium (no neutrons), Deuterium (one neutron, often used in "heavy water"), and Tritium (two neutrons, radioactive, used in some watches for illumination!).
The concept of elements and their transformations has fascinated humans for centuries, from the alchemists of old trying to turn lead into gold (they were, in a way, playing with nuclear physics without realizing it!) to modern scientists exploring the vastness of the universe. Even the word "atom" comes from the ancient Greek word 'atomos', meaning indivisible – little did they know how much more there was to discover!
And speaking of cultural connections, the periodic table itself is like a giant, organized family tree of elements. Each element has its own unique personality, and its isotopes are like different family members with slightly varied traits. It’s a beautiful testament to the order and complexity of the universe.
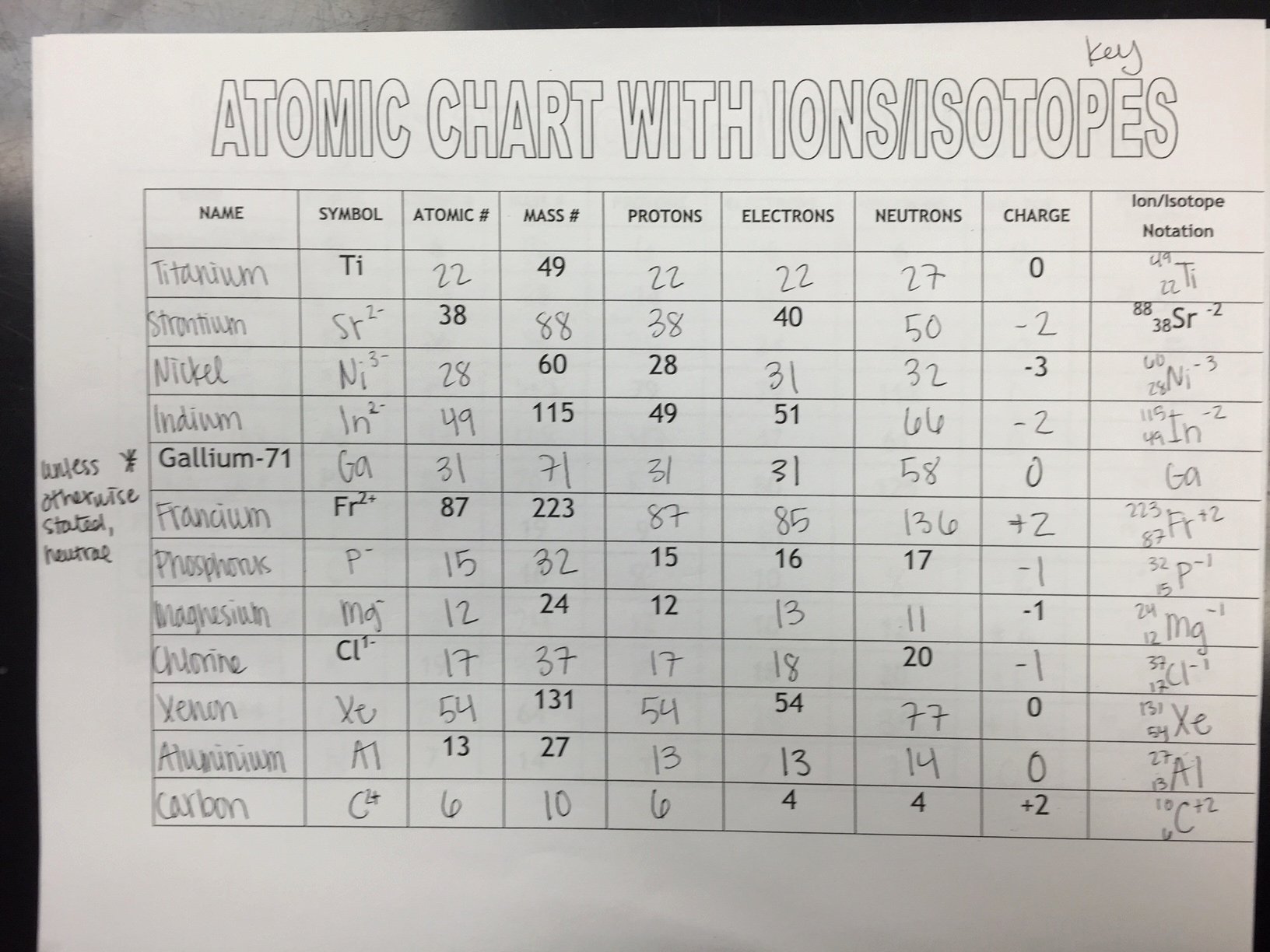
Putting It All Together: A Daily Dose of Ions and Isotopes
So, how does all this connect to our everyday lives, beyond the classroom? Well, those ions we talked about are essential for almost every biological process. When you’re feeling energized, thank those positively charged sodium and potassium ions zipping along your nerves. When your muscles contract to lift a coffee cup, thank the calcium ions. Even the air you breathe has ions in it – atmospheric ions can affect our mood and well-being, though the exact mechanisms are still being studied. It’s a subtle, constant dance of charged particles that keeps us going.
And isotopes? They're quietly at work too. The electricity powering your devices might be generated using nuclear power, which relies on the controlled decay of isotopes like Uranium. The medical scans that can detect early signs of illness? Isotopes. Even the way we date ancient artifacts to understand our human story? Isotopes.
Next time you’re looking at your ions and isotopes practice worksheet, remember that you’re not just memorizing facts. You’re gaining a glimpse into the fundamental workings of the universe. You're understanding why the world behaves the way it does, from the smallest atom to the grandest celestial body. And with a little practice and the thoughtful use of your answer key, you'll be navigating these concepts with ease, feeling confident and connected to the amazing science all around you.
It’s a beautiful thing, this journey of discovery. So, keep asking questions, keep exploring, and remember that even the most complex topics can be approached with a sense of wonder and an easy-going attitude. Happy calculating!
