Ionic Compounds Polyatomic Ions Worksheet Answers
You know, I was recently rummaging through a box of old stuff, and I stumbled upon a binder from my high school chemistry class. Dust bunnies, a rogue pencil stub, and then, BAM! A thick stack of worksheets. I swear, my heart did a little thump-thump of dread mixed with morbid curiosity. And there it was, staring me in the face: "Ionic Compounds and Polyatomic Ions: Practice Problems." Oh, the memories! I distinctly recall the feeling of being both completely fascinated by the concept of ions and utterly bewildered by some of those polyatomic things. Like, how can multiple atoms stick together and act as a single, charged unit? It felt like a chemistry magic trick. And then, the dreaded "worksheet answers." That's what we're diving into today, folks! Because let's be honest, sometimes you just need to see those answers to make the magic, or at least the chemistry, make sense.
I remember feeling like I was decoding ancient hieroglyphs sometimes. You know that moment when you’ve spent an hour wrestling with a problem, convinced you’ve nailed it, only to look at the answer and realize you missed a tiny, crucial detail? Yeah, that was my life with ionic compounds and polyatomic ions. So, consider this your friendly neighborhood guide, a peek behind the curtain, and hopefully, a little less stressful than my original binder experience. We’re not going to get bogged down in super-formal jargon, more like a chat over a (hypothetical) cup of coffee about why these things are cool and how to not pull your hair out when doing the practice.
So, what even ARE ionic compounds, anyway? Think of it like a perfectly balanced dance. You've got your metals, which are usually eager to give away electrons – they're the generous ones, the sharers. And then you have your nonmetals, who are often all too happy to take those electrons. When they come together, they form this electrostatic attraction, like a super strong magnet, holding them in this beautiful, ordered crystal lattice. It’s all about achieving that happy, stable electron configuration. Pretty neat, right?
The Dance of Ions: Metals and Nonmetals
Let’s take a classic example: sodium chloride (NaCl). Yep, that’s table salt! Sodium (Na) is a metal, it’s in Group 1, so it has one valence electron it’s itching to get rid of. Chlorine (Cl), on the other hand, is a nonmetal in Group 17, desperate for one electron to complete its outer shell. So, sodium gives its electron to chlorine. Sodium becomes a positively charged ion (Na+), and chlorine becomes a negatively charged ion (Cl-). These opposite charges attract like crazy, forming the ionic bond. And voilà! You have salt. Simple, elegant, and probably the most important compound in my kitchen.
This whole electron transfer thing is key. Metals tend to form cations (positive ions), and nonmetals tend to form anions (negative ions). The charges they get are usually predictable based on their position on the periodic table. It’s like they have a designated role. Metals in Group 1 form +1 ions, Group 2 form +2 ions. Nonmetals in Group 17 (the halogens, remember them?) usually form -1 ions, and Group 16 elements form -2 ions. See? A pattern!
Now, the tricky part starts when we introduce... wait for it... polyatomic ions. These guys are the rebels of the ionic compound world. They're not just a single atom with a charge; they're a group of atoms that have bonded together covalently (sharing electrons, like pals) but, as a whole unit, carry an overall charge. It’s like a little, tightly-knit family that has decided to go out and get a positive or negative vibe.
Polyatomic Ions: The Rebel Gangs of Chemistry
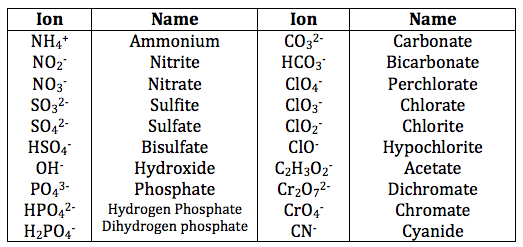
These polyatomic ions are what really make worksheets feel like a puzzle. You've got to know their names, their formulas, and their charges. Think of them like special nicknames and secret handshakes you need to learn. Common examples include nitrate (NO3-), sulfate (SO42-), and carbonate (CO32-). Notice how they all have a charge at the end? That's crucial.

The big challenge with these is remembering them. There’s no simple periodic table trick for polyatomic ions; you just have to memorize them. I know, I know, “memorize” is a scary word. But trust me, once you get a few under your belt, they start to feel familiar. It’s like learning song lyrics; the first few times are rough, but then they just flow.
Let’s break down how they fit into ionic compounds. When a polyatomic ion forms an ionic compound with a metal, it still follows the rules of charge balance. So, if you have sodium (Na+) and you want to combine it with nitrate (NO3-), it's a straightforward 1:1 ratio because the charges cancel out. Sodium nitrate is NaNO3. Easy peasy!
But what happens when you have a polyatomic ion with a different charge, or you're combining it with a metal that can have multiple charges? That’s where things get a little more interesting. Take magnesium (Mg2+) and sulfate (SO42-). The charges are both 2+, so they combine in a 1:1 ratio to form magnesium sulfate (MgSO4). Still pretty clean.
Now, imagine you have aluminum (Al3+) and you want to combine it with sulfate (SO42-). Here’s where you need to find the least common multiple of the charges. You need enough sulfates to balance out the three positive charges of aluminum. So, you need two aluminum ions (total charge +6) and three sulfate ions (total charge -6). This gives you aluminum sulfate, Al2(SO4)3.
And here's a super important detail for the worksheets: when you need more than one of a polyatomic ion, you have to put it in parentheses! See that (SO4)3? That little set of parentheses tells us we have three whole sulfate units. If you forget the parentheses, like Al2SO43, it would imply you have 43 oxygen atoms, which is NOT what you want. It's like saying you have three apples and saying you have 'applesss'. It changes the meaning entirely!
Decoding the Worksheet Answers: Common Pitfalls and Aha! Moments
Okay, so let's get to the juicy part: looking at some hypothetical worksheet answers and figuring out what went wrong (or right!). Imagine a problem asks you to name the ionic compound formed from calcium (Ca) and hydroxide (OH-).
Calcium is in Group 2, so it forms a Ca2+ ion. Hydroxide is a common polyatomic ion with a charge of -1 (OH-). To balance the charges, you need two hydroxide ions for every one calcium ion. So, the formula is Ca(OH)2. The name? You say the cation first, then the anion. Calcium hydroxide. If your answer was just CaOH, you'd be missing a hydroxide. If you wrote CaOH2 without parentheses, it would imply you have two hydrogen atoms and one oxygen atom stuck together in a weird way, not two separate hydroxide ions. The parentheses are your best friend here!
Another common scenario: naming a compound with a transition metal. Transition metals, like iron (Fe) or copper (Cu), can often form ions with different charges. This is where Roman numerals come in handy for naming. Let's say you have a compound that's FeCl2.
We know chlorine (Cl) typically forms a -1 ion (Cl-). Since there are two chloride ions, the total negative charge is -2. To balance this, the iron ion must have a charge of +2. So, the name is iron(II) chloride. The (II) in Roman numerals tells you the charge of the iron. If you just wrote "iron chloride," your teacher (or the answer key) would have no idea if you meant iron(I) chloride (FeCl), iron(II) chloride (FeCl2), or even iron(III) chloride (FeCl3). Specificity is key, my friends!

What if the worksheet answer says something like "sulfur trioxide" for an ionic compound? Hmm, that’s a red flag! Sulfur and oxygen are both nonmetals. Ionic compounds form between metals and nonmetals. "Sulfur trioxide" (SO3) is a covalent compound, typically formed between two nonmetals. Ionic compounds are named differently. For example, if you were looking at the sulfate ion (SO42-) and it formed a compound with potassium (K+), the formula would be K2SO4, and the name would be potassium sulfate. Notice the "ate" ending on sulfate. That's a hallmark of many polyatomic ions.
Let's consider another one. If you saw the answer "sodium hydroxide" and you wrote the formula as NaH(OH), that would be incorrect. Sodium is Na+. Hydroxide is OH-. They combine 1:1. So, NaOH. The NaH(OH) suggests you're trying to combine a sodium ion with a hydrogen ion and then another hydroxide. It's overcomplicating things and likely a misinterpretation of how the ions bond.
Sometimes, people get confused with the "per-" and "hypo-" prefixes. For example, with chlorine, you have hypochlorite (ClO-), chlorite (ClO2-), chlorate (ClO3-), and perchlorate (ClO4-). Each step down in oxygen atoms (or up in the case of perchlorate) has a specific name. If the answer key for a problem involving ClO2- says "chlorite," and you’ve written "chlorate," you've probably mixed up your oxygen count. It’s a common slip-up!
And what about those tricky polyatomic ions that have the same name but different charges? Like sulfite (SO32-) and sulfate (SO42-). They look so similar! If you're asked to name the compound formed from iron(III) and sulfite, and you write Fe(SO4)3, that's not quite right. Iron(III) is Fe3+. Sulfite is SO32-. To balance these, you need two Fe3+ ions (total +6) and three SO32- ions (total -6). So the formula is Fe2(SO3)3. The name is iron(III) sulfite. If you accidentally used sulfate, you'd get Fe2(SO4)3, which is iron(III) sulfate. Two different compounds, two different names, two different formulas. It’s these little details that matter!

Tips and Tricks for Nailing Your Own Answers
So, how can you avoid the confusion I’m painting here? Well, first off, get a good polyatomic ion cheat sheet. Print one out, stick it on your wall, tattoo it on your arm (okay, maybe not that last one). Having the formulas and charges readily available is half the battle.
Second, practice, practice, practice. It sounds cliché, but it’s true. The more you do these problems, the more natural the naming and formula writing will become. You'll start to recognize the patterns and the common polyatomic ions.
Third, slow down. When you’re doing a worksheet, take your time. Read the question carefully. Identify the metal and the nonmetal (or polyatomic ion). Determine their charges. Then, figure out the ratio needed to balance those charges. And don't forget those parentheses for polyatomic ions!
Finally, don't be afraid to check your answers. Looking at the answers isn't cheating; it's learning. When you get something wrong, try to understand why it was wrong. Was it a forgotten Roman numeral? A missing set of parentheses? A mistaken polyatomic ion formula? Pinpointing the error is the fastest way to improve.
Ionic compounds and polyatomic ions can seem daunting at first, but they're really about understanding a set of rules and a vocabulary. Once you get the hang of it, you'll find a certain satisfaction in being able to decode these chemical formulas and names. It's like learning a new language, and a pretty useful one at that! So, next time you’re staring down a worksheet, remember this chat. And who knows, maybe your binder will be a source of pride rather than a dusty reminder of chemistry woes. Happy bonding!
