Ionic Compounds Are Poor Conductors Of Electricity In Which State

Hey there, science explorers! Ever wondered about the secret lives of everyday stuff? Like, what makes salt, well, salt? We're about to dive into a super cool, slightly surprising world of ionic compounds. Think of them as tiny, charged teams, playing a game of give-and-take with electrons. It's all about attraction and making sweet, sweet bonds. And while they can be pretty darn useful in their solid form, there’s a particular situation where they decide to take a break from their electrifying duties. And honestly? It’s kind of a drama!
Now, these ionic compounds, when they’re all snug and solid, are like a tightly packed ballroom. Imagine all the dancers holding hands really, really tightly. They're all in place, looking super neat and orderly. This is where the magic of their structure really shines. They form these amazing, repeating patterns called crystal lattices. It's like building with perfectly shaped LEGO bricks, but instead of plastic, we've got positively charged bits and negatively charged bits, all holding onto each other with a powerful grip. This grip, this strong attraction, is what makes them solid and stable. They're happy in their positions, not really wanting to move around too much. And that’s key to our story!
So, when we’re talking about conducting electricity, we need something to move. Think of electricity as a parade of tiny, energetic characters. For them to march, they need space to wiggle and jiggle and march along. In the solid state, our ionic compounds are like a super disciplined army. Everyone is locked in step. There are these charged particles, right? We call them ions. They're the charged players in our ionic team. But in a solid ionic compound, these ions are stuck! They’re like VIPs at a very exclusive party, absolutely fixed in their spots. They can jiggle a little in place, like doing a tiny jig, but they can’t actually move from one spot to another to carry that electric parade. It’s like trying to have a conga line where everyone is glued to the floor. No conga line, no electricity!
It’s this immobility that makes them such poor conductors when they're just chilling as a solid. They're not performing their electrical duties. They're taking a well-deserved nap, essentially. And it’s this very characteristic that makes their behavior in other states so utterly fascinating. It’s like watching a shy performer suddenly take center stage – the transformation is what makes you lean in and pay attention!
So, what state are we talking about where they don't conduct electricity? It’s the state where they're all neat, tidy, and holding onto each other with all their might. That’s right, it’s the solid state. They are poor conductors of electricity when they are in their solid, crystalline form. They are perfectly happy being solid, and in that state, they are not going to be carrying any electrical current. They're just too… well, solid! Imagine trying to get a crowd of people who are all linked arm-in-arm to suddenly form a moving line. It's just not going to happen easily!

The solid state is where ionic compounds really put on their "closed for business" sign for electricity.
It’s almost like they’re saying, “Electricity? Not today, thank you. We’re quite comfortable right here, being a solid.” And honestly, it’s a perfectly reasonable stance to take. They are built to be stable and strong in their solid form. This stability is what makes them useful for so many things – from the salt on your table to the minerals in the Earth’s crust. They are the silent, steady anchors of our world. But when it comes to electricity, their solid nature means they’re not participating in the electric rave. They’re more like the spectators, observing from the sidelines.
Now, here’s where the plot thickens, and it’s super cool! If you were to take that same ionic compound and, say, melt it down (which requires a lot of heat, by the way!), or dissolve it in water, something amazing happens. Suddenly, those ions, which were previously stuck in their crystal lattice, are now free to roam! They’re like kids let out of school, ready to move and groove. And when they can move, they can carry that electrical parade. It’s this dramatic shift from being a non-conductor to becoming a conductor that makes ionic compounds so endlessly interesting.
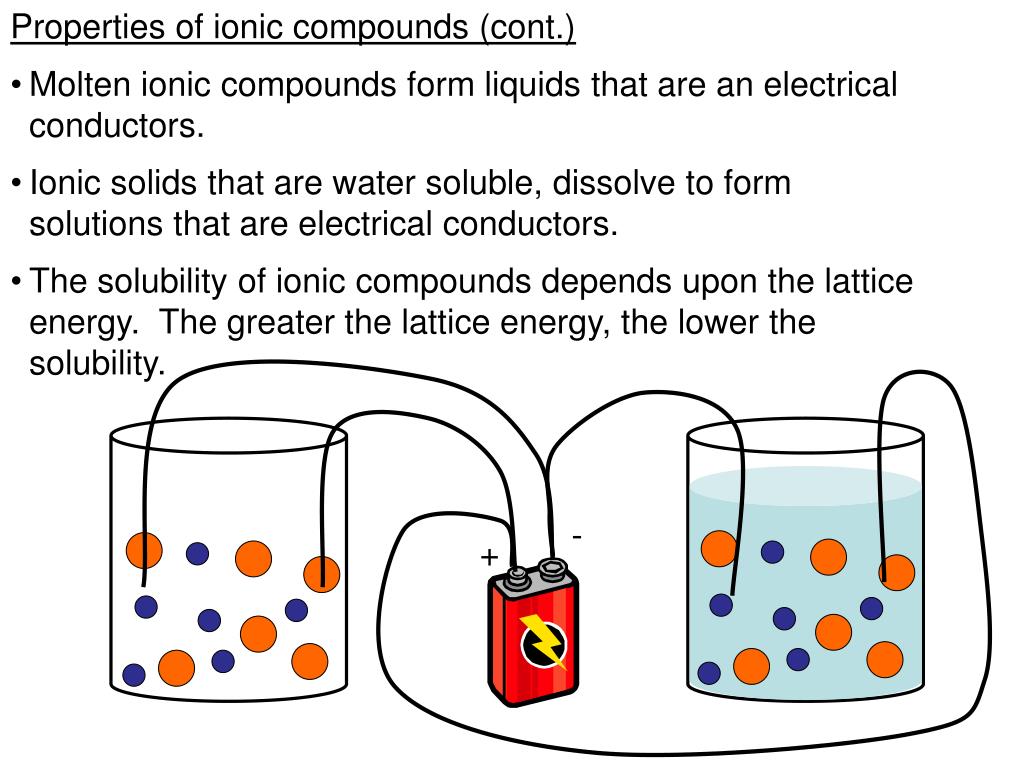
The fact that they are poor conductors only in their solid state is a bit of a signature move. It’s their way of saying, “You can’t just expect me to conduct electricity all the time! My conditions need to be just right.” It’s like a diva with very specific requirements for a performance. And that specificity is part of the charm. It makes you want to understand the ‘why’ behind it. What makes them change their tune so drastically? It’s this contrast, this ability to transform, that makes studying them so rewarding and, dare I say, even a little bit fun. You're not just learning facts; you're witnessing a chemical personality!
So, the next time you’re looking at a solid piece of salt, remember its secret. It’s a master of stillness when it comes to electricity, a true solid citizen. And that’s not a boring fact; it’s a clue to a much bigger, more exciting story about how matter behaves. It’s a reminder that even the most common things have hidden depths and surprising quirks. And who knows? This little tidbit might just spark your curiosity to explore more of the fascinating, electrifying (or not!) world of chemistry. It’s a journey worth taking!
