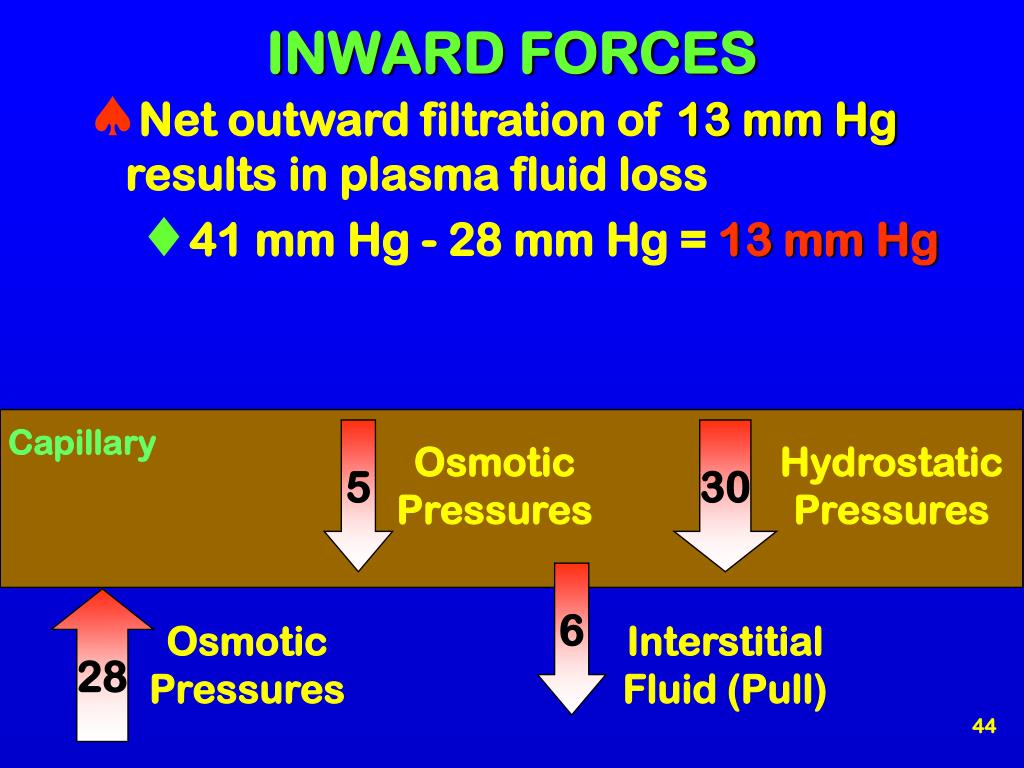
Inward-pulling Force Of Particles In The Vascular Fluid
Imagine your body is a bustling city, and your blood vessels are the superhighways. Everything zips around, from tiny delivery trucks carrying oxygen to speedy little messengers delivering nutrients. But have you ever thought about what keeps all these vehicles from just flying off the roads?
There's a secret force at play, a gentle but persistent tug-of-war happening constantly within your vascular fluid. It’s like a group of friendly but determined bouncers, making sure everyone stays in their lane and the whole city runs smoothly.
This invisible force is called the osmotic pressure. Don't let the fancy name scare you! Think of it as your body's clever way of saying, "Hey, let's keep things balanced!"
It all starts with the tiny building blocks of life: molecules. These little guys love to move around, and they have a special preference for hanging out where there are more of their friends. It’s like a party – molecules tend to gather where the crowd is thickest!
In your blood, you have all sorts of molecules floating around: water, salts, proteins, and sugars. Some of these molecules are really big and bulky, like giant lumbering buses – these are your proteins. Others are tiny and nimble, like speedy motorcycles – these are your salts and small sugars.
Now, here’s where the magic happens. The big protein buses can't easily slip through the walls of your blood vessels. They're likeVIP guests who have special access but can't just wander into any room.
But the tiny salt and sugar motorcycles? They can zip in and out of those walls like they own the place! This difference is key to our inward-pulling force.
Because the big protein buses are stuck inside the blood vessel, they create a sort of "molecule party" inside. There’s a higher concentration of "partygoers" inside the vessel than outside, in the surrounding tissues.
And guess what? The water molecules, being the friendly social butterflies they are, want to join the party! They are attracted to areas where there are more dissolved particles, like the salts and sugars that are hanging out with the proteins.

So, the water molecules from the surrounding tissues get gently coaxed, or pulled, towards the inside of the blood vessel. It’s like a silent invitation: "Come on in, the water's fine and there's lots of stuff to hang out with here!"
This inward pull is incredibly important. It’s constantly working to keep fluid inside your blood vessels, preventing your tissues from swelling up like overfilled balloons.
Think about when you get a cut and see a bit of clear fluid oozing out. That’s your body’s emergency response, but even in those situations, the osmotic pressure is trying to pull fluid back in and seal the deal.
This force, often called oncotic pressure when referring specifically to the pull of proteins, is like a silent guardian of your vascular system. It ensures that your blood stays in its designated highways and byways.
It’s a beautiful example of how even at the microscopic level, there are these incredible, coordinated efforts keeping you alive and functioning. No one’s giving orders, no one’s shouting commands, just these tiny particles doing their thing.
Imagine a team of tiny, invisible gardeners constantly tending to your internal landscape. They are carefully watering the plants (cells) without letting the soil (tissues) become waterlogged. The osmotic pressure is their gentle watering can.
It’s also why when you eat salty snacks, you might feel a bit thirsty. Your body is trying to dilute that extra salt in your bloodstream, and the osmotic pressure plays a role in drawing water to help out!
This is a natural process, a beautiful dance of molecules. It’s not a forceful shove, but rather a soft, insistent whisper, guiding the water where it needs to be.
This inward pull is a constant reminder that your body is a marvel of engineering. It’s a living, breathing system where every little component has a crucial role.
So next time you feel a pulse in your wrist or see the blush of blood in your cheek, remember the unsung heroes: the molecules and their osmotic pressure. They are the gentle tug that keeps your internal rivers flowing correctly.
It's like a secret handshake between water and the larger molecules. They say, "You stay here, and I'll bring the water to you." And it works, beautifully and tirelessly.
This is why hydration is so vital. When you're well-hydrated, your body has plenty of water molecules to go around, and the osmotic pressure can work its magic efficiently.
Think of it as the ultimate team-building exercise. The proteins are the anchors, and the water is the supportive crew, making sure everything stays stable.

It's a humbling thought, isn't it? That such a profound process, essential for life, is happening silently within us all the time.
This inward pull isn’t just about preventing swelling; it’s also about ensuring that your cells get the right amount of fluid they need to function. Too little water, and they'd shrivel; too much, and they might burst.
The osmotic pressure is like a perfectly calibrated thermostat for your cellular fluid levels. It’s always adjusting, always maintaining that delicate balance.
It’s a testament to the elegance of nature. Simple principles, when applied on a grand scale, create the complex and wonderful reality of a living organism.
So, while doctors might use big words like colloid osmotic pressure, remember it’s just your body’s incredibly smart way of managing water using the power of its tiny inhabitants.
It’s the quiet hum of life, a gentle tug that keeps everything in its place, ensuring your body's internal city thrives.

This is the same force that helps your kidneys filter waste and your intestines absorb nutrients. It’s a fundamental player in so many vital bodily functions.
It's like having a team of tiny, invisible doormen at the entrance of your blood vessels, politely but firmly encouraging the water to stay inside.
The plasma proteins, particularly albumin, are the stars of this show. They are the magnets that draw the water in.
It's a silent agreement, a constant negotiation happening at the molecular level. A beautiful, life-sustaining ballet.
So, the next time you feel a gentle pull or a sense of fullness, you can smile and think of the invisible forces at work, keeping your body perfectly balanced, one molecule at a time.
It's a heartwarming reminder that even the smallest things can have the biggest impact.
And that, in essence, is the delightful and surprising story of the inward-pulling force of particles in your vascular fluid!
