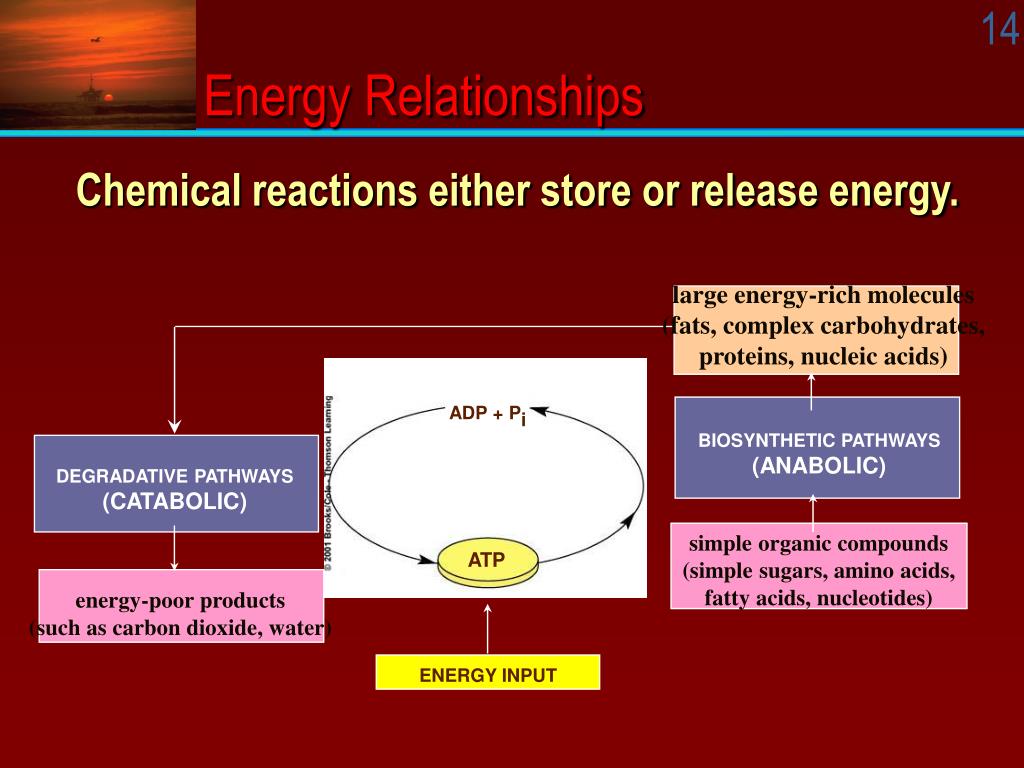
Investigating Energy Relationships For Various Reactions
Hey there, fellow humans! Ever stopped to think about the magic that happens when things change? You know, like when you bake a cake, or when your phone battery finally gives up the ghost? It's all about energy, baby! And today, we're going to dive into the wonderfully weird world of how energy plays with different reactions. Don't worry, it's not going to be like a boring chemistry lecture. Think of it more like a friendly chat over a cuppa, exploring the hidden powers behind everyday stuff.
So, what exactly are we talking about when we say "energy relationships for various reactions"? Imagine energy as this invisible force, like the feeling you get when you're super excited about something, or the slump you feel after a long day. Reactions are just things changing – molecules bumping into each other, breaking apart, or sticking together to make something new. And in all these changes, energy is either being borrowed, lent, or just shuffled around.
Let's kick things off with something we all know and love: burning stuff. Think about lighting a campfire. Poof! Fire. That fire gives off heat and light, right? That's energy being released. This kind of reaction is called exothermic. "Exo" like "exit," so energy is exiting the reaction and heading out into the world for us to feel as warmth or see as light. It's like when you open a really good bottle of fizzy pop, and all that trapped bubbly energy just bursts out!
Now, picture this: you're trying to make a particularly stubborn jelly set. You add all the ingredients, stir and stir, but it's just not jelling. You need to give it a bit of a nudge, right? Maybe a bit more heat, or a bit more time. Some reactions are like that – they need a little input of energy to get going. These are called endothermic reactions. "Endo" like "enter," so energy is entering the reaction. Think about melting ice. You have to add heat (energy) to turn that solid ice into liquid water. It’s like your body needing food (energy) to function and stay warm. If you don't eat, you feel sluggish and cold, much like an endothermic reaction that hasn't received enough energy.
Why Should You Even Care About This Energy Shenanigans?
Honestly? Because it explains so much about the world around you! It’s the secret sauce behind how our bodies work, how our cars run, and even why your ice cream melts on a hot day.

Let's talk about our own bodies. We're essentially a giant, incredibly complex series of reactions. When we eat food, our bodies break it down through a whole bunch of exothermic reactions. That's where we get the energy to run, jump, think, and even just blink! If our bodies weren't good at releasing energy from food, we'd be pretty much just… lumps. Kind of like a dead battery in a remote control. No juice, no action!
And think about your car. When you put gas in it, that gasoline is full of stored energy. The engine then sparks a controlled explosion – a super-fast exothermic reaction – that pushes the pistons and makes your car zoom down the road. Without that energy release, your car would just sit there, looking pretty but going nowhere. It’s like trying to play your favorite video game without plugging in the console – pretty useless, right?

The Little Stories of Energy
Let's get a bit more granular. Imagine two molecules are best friends, and they're super happy together. They're holding hands, just chilling. This is a low-energy state. Now, something comes along and breaks them apart. If it takes a lot of effort (energy) to pull them apart, then when they were together, they were holding onto a lot of that energy. When they break apart, that energy might be released, or it might be absorbed to make the separation happen. It’s like trying to separate two people who are really in love. You’d have to expend a lot of energy to get them to go their separate ways, and maybe they'd even feel a bit drained afterward.
Conversely, sometimes two molecules are just sort of… meh. They’re not that happy together, and they’re not really doing much. Then, something comes along and makes them stick together. This might happen because the new bond they form is more stable, meaning it has a lower energy. When they form this new, happier bond, they often release some extra energy. Think of it like finding your perfect soulmate after a string of awkward dates. When you finally connect, there’s this surge of positive energy, right? That’s sort of what happens in some chemical reactions when new, more stable bonds are formed.

So, the relationship between energy and a reaction is all about whether the starting materials are happier (lower energy) and the products are less happy (higher energy), or vice versa. If the products are happier (lower energy) than the starting materials, energy is usually released (exothermic). If the products are less happy (higher energy) than the starting materials, energy needs to be absorbed to make it happen (endothermic).
The Coolest Part: Making Things Happen!
Understanding these energy relationships is how scientists and engineers design all sorts of amazing things. They can figure out how to create fuels that release lots of energy (like gasoline or even rocket fuel!), or how to design processes that need very little energy input (saving us money and resources!).

Take photosynthesis in plants, for example. Plants take sunlight (energy!), carbon dioxide, and water, and they use it to make their own food (sugars) and release oxygen. This is an endothermic process, powered by the sun's energy. Without it, we wouldn't have the oxygen we breathe or the plants that form the base of so many food chains. It’s like a solar-powered snack factory for the planet!
And then there are batteries. When you charge your phone, you're essentially forcing an endothermic reaction to happen, storing energy. When you use your phone, an exothermic reaction releases that stored energy, powering your screen and your apps. It’s a constant dance of energy storage and release, all thanks to understanding these fundamental relationships.
So, the next time you feel the warmth of a fire, watch a plant grow, or even just notice your phone battery draining, remember the incredible dance of energy happening behind the scenes. It’s not just abstract science; it’s the very essence of how our world works, one reaction at a time. Pretty neat, huh?
