Introduction To Acids And Bases Webquest Answers

Ever feel like you're just… vaguely aware that there are "acids" and "bases" out there, maybe from a dusty high school science class? Like, you know vinegar is an acid, and baking soda is a base, and they do something when you mix them, but the rest is a bit fuzzy? Well, pull up a comfy chair and let's chat about them. Think of this as a little peek behind the curtain of everyday chemistry, no lab coat required!
You've probably stumbled across a "webquest" before, maybe for your kids or even for yourself. They're basically scavenger hunts on the internet, where you follow clues and find answers. And when it comes to acids and bases, a webquest can be a super fun way to unlock some cool secrets about the world around you. So, what are the answers to the big questions about these chemical critters?
Acids: The Zesty and The Zingy
Let's start with acids. Imagine your taste buds. Acids often have a sour or tart taste. Think of biting into a lemon. Oof! That's your sensory system telling you, "Hey, there's an acid here!" That sourness is a classic sign. It's like the citrus family's signature move.
But it's not just about taste! Acids are everywhere. Your stomach acid, for example, is a super strong acid (hydrochloric acid, if you're curious) that helps you digest your food. It's like a tiny, powerful cleaning crew inside you, breaking down all those yummy (and sometimes not-so-yummy) things you eat. Without it, we'd be in a bit of a pickle, literally!
Think about other everyday acids. Vinegar, of course, is a household staple for cleaning and cooking. That tangy flavor in your salad dressing? Yep, acid. Even some of the things you use to make your hair shiny or your skin feel smooth might contain mild acids.
So, what makes something an acid? In the simplest terms, many acids tend to release hydrogen ions (H+) when they're in water. Don't get too bogged down in the jargon, but think of it like these hydrogen ions being little explorers, ready to interact with other things. This "releasing" is a key part of their personality.
Why should you care about acids? Well, understanding them helps you understand why certain foods taste the way they do, how your body works, and even how to clean effectively. Plus, it’s kind of neat to know that these invisible forces are constantly at play!

Bases: The Smooth and The Slippery
Now, let's switch gears to bases. If acids are often sour, bases are typically bitter. Now, you probably aren't going around tasting bitter things unless you're, say, experimenting with some questionable berries. But that bitterness is a characteristic. Think of the slightly unpleasant taste of unsweetened baking chocolate – that's got some basic properties!
Another really noticeable trait of many bases is that they feel slippery. Like, when you accidentally get soap in your eyes (ouch!), that slick, sometimes slimy feeling? That's a base at work. Soaps and detergents are often basic. They're designed to break down grease and dirt, and that slipperiness is part of their cleaning power.
Remember baking soda? That's a classic base. It's often used in baking to react with acids (like buttermilk or vinegar) to make cakes and cookies rise. It’s like a little chemical party happening in your batter, creating tiny bubbles of gas that puff everything up. Who knew your favorite muffin owed its fluffiness to chemistry?
What makes a base a base? In a nutshell, they often accept hydrogen ions or release something called hydroxide ions (OH-). It's like they’re the opposite of acids in this regard, ready to balance things out. They are the Yin to the acid's Yang, if you will.

Why should you care about bases? They are essential for keeping things clean, for making your bread rise, and even for controlling the acidity in certain environments, like swimming pools. They’re the unsung heroes of many a household task!
When Acids and Bases Meet: The Grand Neutralization
Here's where the real magic happens! What do you think occurs when you mix an acid and a base? Do they fight? Do they run away from each other? Nope! They neutralize each other. It’s like a friendly handshake and a hug.
Remember that volcano science project you might have done as a kid? That involved mixing baking soda (a base) and vinegar (an acid). The fizzy eruption you saw was the result of this neutralization reaction, where they cancel each other out and produce water and a salt. It’s a spectacular demonstration of them finding a happy medium.
This neutralization is super important in many applications. If you get heartburn, which is caused by too much acid in your stomach, you might take an antacid. Antacids are basically mild bases that help to neutralize that excess stomach acid, bringing you some relief. It's like a tiny chemical peace treaty happening in your gut!
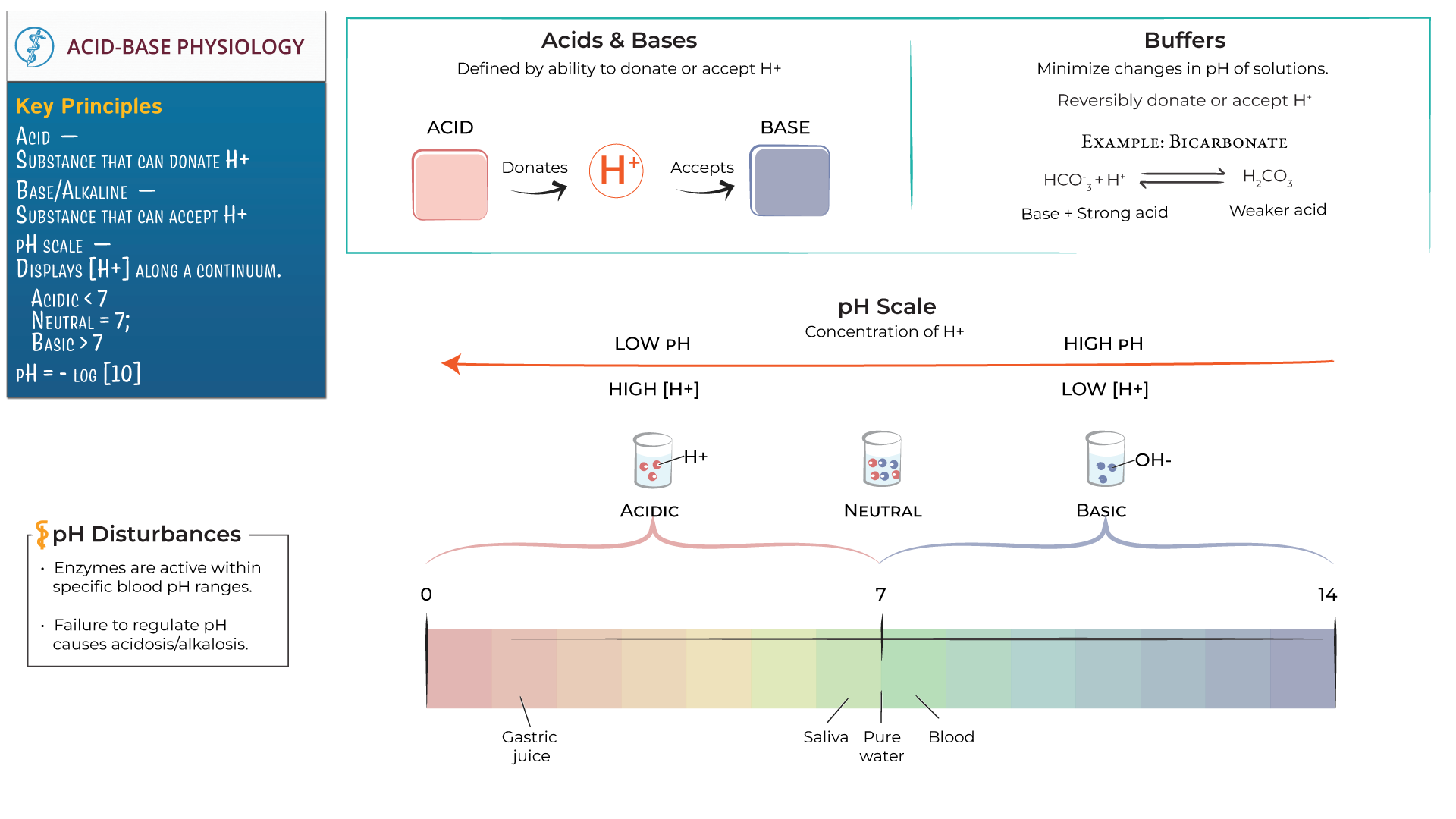
This balancing act is a fundamental concept in chemistry and in life. It's all about finding equilibrium, about things working together to create something new and often, something calmer.
The pH Scale: The Acid-Base Thermometer
So, how do we know if something is strongly acidic, weakly acidic, strongly basic, or weakly basic? Enter the pH scale. Think of it like a thermometer, but instead of measuring temperature, it measures how acidic or basic something is. It ranges from 0 to 14.
Things with a pH below 7 are acidic. The lower the number, the stronger the acid. So, stomach acid (around pH 2) is much more acidic than lemon juice (around pH 2.3). Pure water sits right in the middle at pH 7, which is neutral.
Things with a pH above 7 are basic. The higher the number, the stronger the base. So, ammonia (around pH 11) is a much stronger base than baking soda (around pH 8.3).

Why does this matter? The pH of something can affect how it behaves and how it interacts with its surroundings. For example, many plants need soil with a specific pH range to grow properly. Fish need water with a certain pH to survive. Our own bodies have tightly controlled pH levels to keep us healthy.
Understanding the pH scale helps us understand why things happen the way they do, from why your laundry detergent works to why certain natural environments are healthy or not.
Putting It All Together: Why Bother?
So, after this little dive into acids and bases, are you any less fuzzy? Hopefully, a little bit! You've learned that acids are often sour and reactive, bases are often bitter and slippery, and they love to neutralize each other. You've also met the pH scale, your guide to their strength.
Why should you, an everyday reader, care about these chemical concepts? Because they are the invisible threads that weave through so much of our daily lives. They explain the taste of our food, the effectiveness of our cleaning supplies, the health of our bodies, and the balance of our planet. They are not just abstract ideas in a textbook; they are the building blocks of our reality.
Next time you squeeze a lemon, use soap, or bake a cake, you'll have a little more appreciation for the incredible chemistry happening right under your nose. It’s a small peek into a vast, fascinating world, and knowing a bit about it can make the ordinary just a little bit more extraordinary.
