Intermolecular Forces Include All Of The Following Except

Ever wondered what makes things stick together? It's not magic, it's science! And today, we're diving into the super cool world of Intermolecular Forces. Think of them as the invisible glue that holds everything around you in place. From the water in your cup to the air you breathe, these forces are the unsung heroes of our everyday lives. It’s like a secret society of tiny particles, all holding hands!
Now, the really fun part? There's this fantastic game, a sort of quiz, that tests your knowledge about these forces. It's called "Intermolecular Forces Include All of the Following Except." Sounds a bit like a riddle, right? And that’s exactly what makes it so engaging. It's not just a dry list of facts; it’s a challenge that sparks your curiosity. You have to really think about what makes these forces tick. It’s like being a detective, piecing together clues to figure out what doesn't belong.
Imagine you're at a party, and everyone's mingling. Some people are in big, boisterous groups, while others are chatting one-on-one. Intermolecular forces are kind of like that, but on a molecular level. They describe how different molecules interact with each other. Some molecules are real social butterflies, attracted to each other strongly. Others are a bit more reserved. This quiz helps you identify the molecules that are not playing by these social rules. It's the outlier, the one that just doesn't fit into the picture of intermolecular interactions.
What’s so special about this particular quiz or concept? It’s the unexpected twists! You’ll learn about different types of forces, like the strong hugs of Hydrogen Bonds, the gentle nudges of Dipole-Dipole Interactions, and the fleeting whispers of London Dispersion Forces. Each has its own personality and its own way of making molecules stick. The "except" part of the question is the real kicker. It forces you to understand the definition of these forces so well that you can spot what's not a part of the club.
Think about it: you're presented with a list, and your mission, should you choose to accept it, is to find the imposter. It's like a molecular "Where's Waldo?" but instead of a striped shirt, you're looking for a scientific concept that doesn't belong in the family of intermolecular forces. This isn't just about memorizing definitions; it’s about developing an intuitive grasp of how molecules behave. It’s about seeing the connections and understanding the differences.
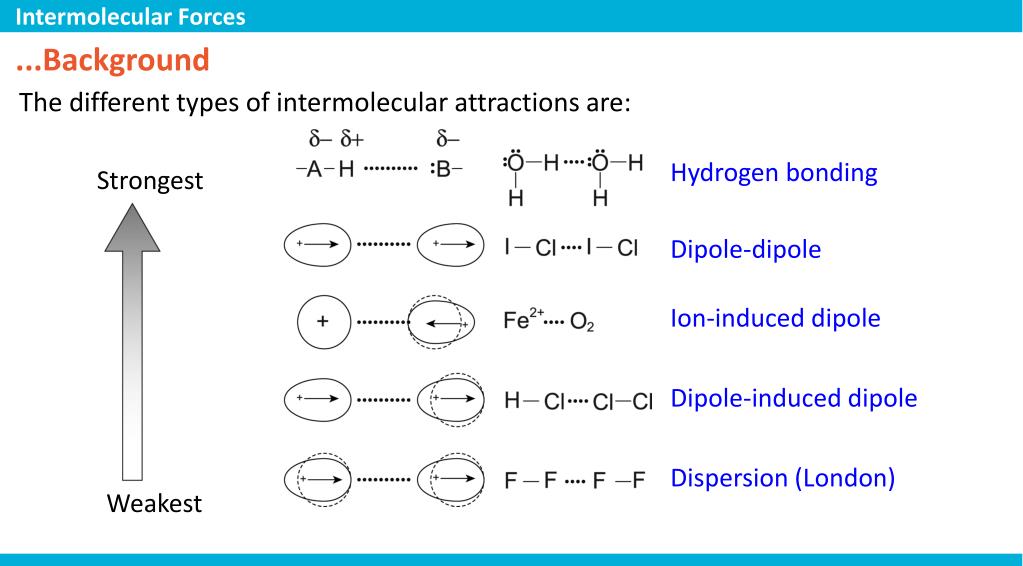
The beauty of it lies in its simplicity, yet its profound implications. We’re talking about everyday phenomena. Why does ice float on water? Intermolecular forces! Why do liquids have surface tension? Intermolecular forces! Why do you feel that slight stickiness when you touch something wet? You guessed it – intermolecular forces! This quiz, by focusing on what isn't an intermolecular force, helps you appreciate the ones that are by highlighting their unique characteristics.
It’s entertaining because it plays on our natural curiosity. We like puzzles, we like challenges, and we like to feel a little bit smarter when we figure something out. This quiz offers that satisfying "aha!" moment. You’re not just reading about science; you’re actively participating in it. You're using your brain to solve a molecular mystery.

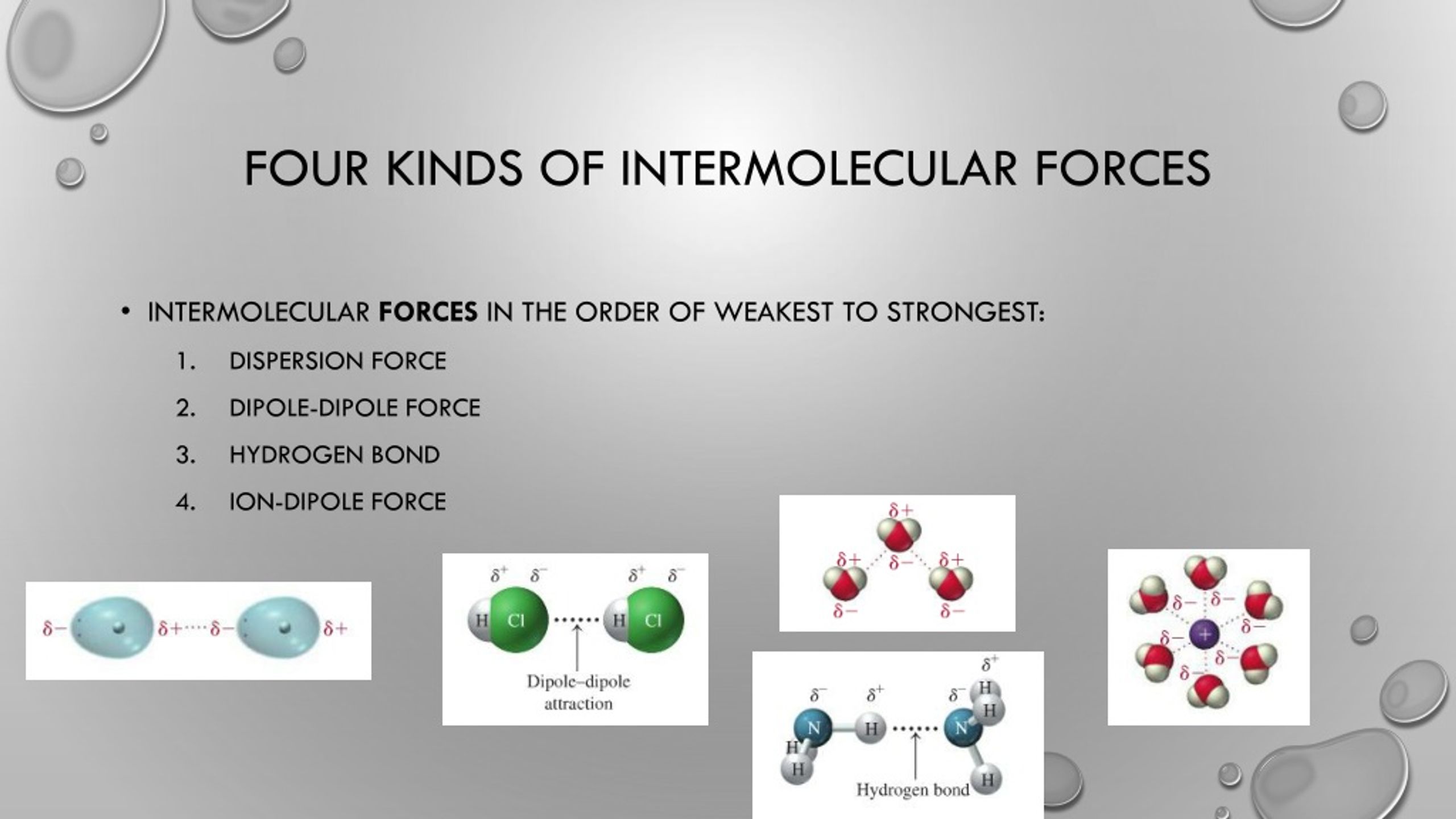
Let’s say you’re looking at a list of options for "Intermolecular Forces Include All of the Following Except." You might see things like Ionic Bonds or Covalent Bonds. Now, these are super important in chemistry, they’re the strong, unbreakable marriages between atoms within a molecule. But they’re not the intermolecular forces, the ones that happen between different molecules. It’s like the difference between the people inside a house and the people talking to each other across the street. Ionic and covalent bonds are like the walls and furniture inside the house; intermolecular forces are the conversations and relationships happening outside.
The quiz makes you think about these boundaries. It sharpens your understanding of what makes an interaction "intermolecular." It's a brilliant way to test comprehension without making it feel like a test. It’s more like a friendly game of "spot the difference," but the differences are fundamental to how matter behaves.
So, when you encounter the phrase "Intermolecular Forces Include All of the Following Except," don't just skim over it. Lean in! Get curious! It’s your invitation to explore the invisible forces that shape our world in the most delightful and surprising ways. It's a little bit of a brain teaser, a touch of a mystery, and a whole lot of fun science.
This isn’t just for chemistry buffs; it’s for anyone who’s ever looked at a dewdrop clinging to a leaf, or felt the steam rise from a hot drink and wondered, "How does that work?" The answer, time and time again, lies in these charming, intricate, and sometimes quirky intermolecular forces. And the "except" part? That’s your secret key to unlocking a deeper appreciation for them. So next time you see it, embrace the challenge. You might just find yourself totally captivated by the tiny, invisible dance of molecules!
It’s this very contrast – understanding what they are by identifying what they are not – that makes the concept so memorable and the quiz so effective. You learn about the strong attractions, the weaker ones, and then, the thing that fundamentally operates on a different level altogether. It’s like learning about different kinds of hugs and then being asked to identify a handshake. They’re both ways of connecting, but they’re distinct in their nature and strength. This "except" question is your chance to become a molecular matchmaker, discerning the true intermolecular connections from the imposters.
The excitement comes from that moment of realization. You’re presented with a question that initially seems complex, but as you break it down, the answer becomes clear, and you feel a genuine sense of accomplishment. It’s a small victory, but it’s a victory that deepens your understanding of the universe around you. So, keep an eye out for that phrase. It’s more than just a question; it’s a gateway to a fascinating scientific adventure!
