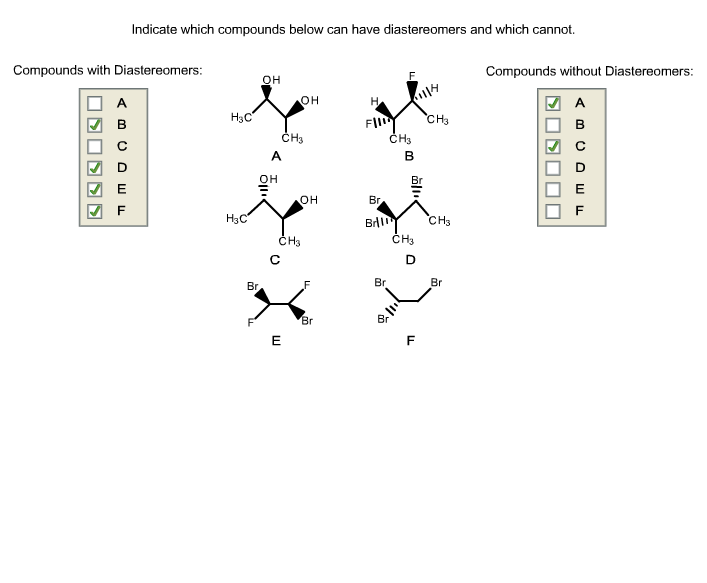
Indicate Which Compounds Below Can Have Diastereomers And Which Cannot
Hey there, fellow molecule enthusiasts! Grab your favorite mug, because we’re diving into a super fun, slightly mind-bending topic today: diastereomers. Ever feel like you're drowning in all these "-mers" and "-tops"? Yeah, me too. But this one's actually pretty cool, like finding a hidden twin in a crowd of strangers. We're gonna figure out which of these molecular kiddos can have those elusive diastereomers and which ones are just, you know, singular beings. No need to stress, this is just a chill chat, so settle in!
So, what’s the big deal with diastereomers anyway? Imagine you’ve got a molecule, right? And it’s got a few bits and bobs attached to it. Sometimes, you can arrange those bits and bobs in a few different ways. If two of those arrangements are not mirror images of each other, but they are different molecules… bingo! You’ve got yourselves some diastereomers. They're like siblings who look kinda alike, but not exactly. They're not identical twins (enantiomers, we'll chat about them another time!), and they're definitely not unrelated cousins. They're… well, diastereomers.
The key thing to remember, the golden rule, the secret handshake for spotting potential diastereomers, is the presence of multiple chiral centers. Think of a chiral center as a carbon atom that’s got four different things sticking off it. It’s like that one friend who can juggle four different balls at once – pretty impressive, right? These chiral centers are the dance floors where the stereochemistry happens. If you’ve only got one of these fancy juggling carbons, you might only get a pair of enantiomers (the mirror-image twins). But if you’ve got two or more, now things get interesting. It opens up a whole world of possibilities, like a choose-your-own-adventure book for molecules!
Let’s get down to business, shall we? We’ve got a list of compounds to suss out. Think of it as our molecule mystery challenge. Our mission, should we choose to accept it (and we totally do, because coffee!), is to identify which of these guys can play the diastereomer game and which are stuck in a solo act. It’s all about spotting those chiral centers, my friends. That’s our superpower for today.
The Compounds in Question!
Okay, so here’s the lineup. We’ll go through them one by one. Don’t be shy if you’re not immediately sure. That’s what we’re here for! We’re gonna break it down, no fancy jargon unless it’s absolutely necessary (and even then, we’ll explain it like we’re talking about pizza toppings).

First up, let’s consider something super simple. How about ethane? C2H6. Easy peasy, right? Two carbons, six hydrogens. Are there any carbons with four different things attached? Nope! Each carbon is only attached to one other carbon and three hydrogens. So, no chiral centers here. Therefore, can ethane have diastereomers? Absolutely not. It’s like trying to find a different flavor of plain water. It just doesn't exist!
Next on our list, let’s think about 2-butanol. C4H10O. This one’s a bit more interesting. Let’s draw it out, or at least picture it in our heads. We’ve got a four-carbon chain. The hydroxyl group (-OH) is on the second carbon. Now, let’s scrutinize that second carbon. What’s attached to it? We’ve got a methyl group (-CH3) pointing one way. We’ve got a hydrogen atom. And then we’ve got the rest of the carbon chain (a propyl group, -CH2CH3) going the other way. And, of course, the hydroxyl group itself! So, that second carbon has four different things attached: -CH3, -H, -CH2CH3, and -OH. Ding, ding, ding! We’ve found a chiral center! Now, does 2-butanol have any other chiral centers? Nope, the other carbons are either attached to two hydrogens, or a carbon and two hydrogens, or a carbon and three hydrogens. So, we have one chiral center. With just one chiral center, what can we have? We can have a pair of enantiomers (mirror images). But can we have diastereomers? Not really. You need at least two chiral centers for true diastereomers to pop up. Think of it as needing two distinct dance floors to have dancers doing different steps simultaneously. So, 2-butanol, while stereochemically interesting with its enantiomers, cannot have diastereomers in the traditional sense of having non-mirror image stereoisomers other than its enantiomer.
Moving on to something a bit more complex, let’s ponder 2,3-dibromobutane. This sounds like it might have some interesting possibilities. Let's break down the name. "Butane" means a four-carbon chain. "Di-bromo" means two bromine atoms. And "2,3-" tells us those bromines are on the second and third carbons. So, we've got carbons numbered 1, 2, 3, and 4. On carbon 2, we have a bromine. On carbon 3, we have another bromine. What else is attached to these carbons? Each carbon in the main chain also has hydrogens attached. Let's look at carbon 2. It's attached to carbon 1 (a methyl group, -CH3), a bromine atom, a hydrogen atom, and carbon 3. Now, carbon 3 is attached to carbon 2, a bromine atom, a hydrogen atom, and carbon 4 (another methyl group, -CH3). So, is carbon 2 chiral? Yes! It has a methyl group, a bromine, a hydrogen, and the rest of the chain (including carbon 3). Is carbon 3 chiral? Yes! It has a methyl group, a bromine, a hydrogen, and the rest of the chain (including carbon 2). Aha! We have two chiral centers here! When you have two or more chiral centers, that's when the diastereomer party can really start. With two chiral centers, you can have up to 2n stereoisomers, where 'n' is the number of chiral centers. So for 2,3-dibromobutane, we could potentially have 22 = 4 stereoisomers. These would include pairs of enantiomers, but crucially, they can also be diastereomers of each other. So, yes, 2,3-dibromobutane can have diastereomers! Exciting stuff, right?
Now, let’s throw a curveball. What about meso compounds? Ever heard of them? They’re like the sneaky twins of the stereochemistry world. A meso compound has chiral centers, but it’s not chiral overall. It has a plane of symmetry. Think of it as a molecule that’s its own mirror image. For example, meso-tartaric acid has two chiral centers, but it’s super symmetrical, so it doesn’t have enantiomers. Can a meso compound have diastereomers? This is where it gets a little tricky, but generally, if a compound is a meso compound, it means it has internal symmetry that prevents it from forming non-mirror image stereoisomers with itself. However, if you have a related compound that isn't meso and has the same number of chiral centers, then yes, that non-meso compound can have diastereomers with the meso compound. But for the purpose of asking if this specific compound (the meso one) can have diastereomers, it's a bit of a nuanced "no" in the sense that it won't have different diastereomers arising from its own internal symmetry in the same way a non-meso compound with multiple chiral centers will. It's like asking if a perfectly balanced seesaw can have a lopsided counterpart – it’s designed to be balanced. So, while it might be part of a diastereomeric relationship with a different molecule, the meso compound itself doesn't generate its own unique set of diastereomers in the typical way. Let's park this for a sec and focus on simpler cases first, but keep that symmetry in mind!
Okay, let’s look at 1,2-dichloropropane. C3H6Cl2. Three carbons. Chlorines on the first and second carbons. Let's examine carbon 1. It's attached to a chlorine, two hydrogens, and carbon 2. Not chiral. Now carbon 2. It's attached to a chlorine, a hydrogen, a methyl group (carbon 3), and carbon 1. Aha! Carbon 2 is attached to four different things: Cl, H, CH3, and the rest of the chain (including carbon 1). So, carbon 2 is a chiral center! What about carbon 3? It’s a methyl group, so it's attached to three hydrogens and carbon 2. Not chiral. So, we have one chiral center here. Just like 2-butanol, having only one chiral center means we can have a pair of enantiomers. But can we have diastereomers? Nope. For diastereomers, we need at least two chiral centers. So, 1,2-dichloropropane cannot have diastereomers.
Let’s try 1,3-dimethylcyclohexane. Now this is where things get really interesting. Cyclohexanes are rings, which can add a whole new layer of complexity. We’ve got a six-membered ring. Two methyl groups attached. Let’s say one methyl is on carbon 1 and the other is on carbon 3. Now, let’s think about the carbons that have the methyl groups attached. Carbon 1 is attached to a methyl group, a hydrogen, and two other carbons in the ring. These ring carbons are themselves attached to other ring carbons and hydrogens. The crucial thing is, are these ring carbons always chiral? It depends on the substituents! If the two methyl groups are on the same side of the ring (cis) or on opposite sides (trans), that matters. But let’s focus on the chiral centers first. For carbon 1, it's attached to the methyl group, a hydrogen, and two different paths around the ring. If the ring itself is substituted such that these paths are different, then carbon 1 could be chiral. Similarly for carbon 3. When you have substituted cyclic systems like this, you often end up with multiple chiral centers. For 1,3-dimethylcyclohexane, both carbon 1 and carbon 3 are chiral centers, provided the ring has the right symmetry or lack thereof. And indeed, they are. So, we have two chiral centers here! With two chiral centers, we can definitely have diastereomers. We can have cis and trans isomers, and then within those, enantiomers. So, yes, 1,3-dimethylcyclohexane can have diastereomers!

What about something like dichloromethane (methylene chloride)? CH2Cl2. This is a classic. A central carbon with two hydrogens and two chlorines. Are any of those groups different? Nope! We have two identical hydrogens and two identical chlorines attached to the central carbon. Therefore, no chiral centers. So, can it have diastereomers? No way. It's just… dichloromethane. No room for variations there!
Let's consider 2,3-butanediol. This is similar to 2,3-dibromobutane, but with hydroxyl groups instead of bromines. We have a four-carbon chain. Hydroxyl groups on carbons 2 and 3. Let's look at carbon 2. It's attached to a hydroxyl group (-OH), a hydrogen atom, a methyl group (-CH3), and carbon 3 (which is part of the rest of the chain). These are four different things! So, carbon 2 is chiral. Now, let's look at carbon 3. It's attached to a hydroxyl group (-OH), a hydrogen atom, a methyl group (-CH3), and carbon 2 (which is part of the rest of the chain). These are also four different things! So, carbon 3 is also chiral. We have two chiral centers! This means, just like 2,3-dibromobutane, 2,3-butanediol can have diastereomers. In fact, it has a fascinating array of stereoisomers, including a meso compound and a pair of enantiomers. So, definitely a yes!
One more for the road! Let’s think about glycine. This is the simplest amino acid. Its structure is H2N-CH2-COOH. The central carbon atom (the alpha-carbon) is attached to an amino group (-NH2), a carboxyl group (-COOH), two hydrogen atoms, and a hydrogen atom. Wait, no, that’s not right! The alpha-carbon is attached to an amino group, a carboxyl group, and two identical hydrogen atoms. Since two of the groups attached to the alpha-carbon are identical (the hydrogens), it is not a chiral center. Therefore, glycine has no chiral centers. Can it have diastereomers? You guessed it: no. It’s too simple to have that kind of stereochemical complexity!

The Takeaway - It’s All About Those Chiral Centers!
So, what have we learned from our little coffee chat about molecules? It’s pretty straightforward when you get down to it. The absolute key to a molecule being able to have diastereomers is the presence of two or more chiral centers. If a molecule only has one chiral center, it can only have enantiomers (mirror image pairs). If it has no chiral centers at all, well, then it’s just… itself. No stereoisomers to worry about, which can be a relief sometimes!
Think of it like this: chiral centers are the Lego bricks that allow you to build different 3D structures. One brick can only give you so many different arrangements. But two, three, or more bricks? Now you've got a whole construction site of possibilities! And when those arrangements aren't mirror images, you've hit the jackpot – diastereomers!
So next time you’re looking at a molecule and wondering about its stereochemical potential, just do a quick scan for those chiral centers. Count them up. If you see two or more, start thinking about diastereomers! If you see one, you’re looking at enantiomers. And if you see none, well, enjoy the simplicity. It’s all part of the beautiful, intricate world of chemistry. Keep those molecular minds buzzing!
