Indicate Whether The Following Balanced Equations Involve Oxidation Reduction
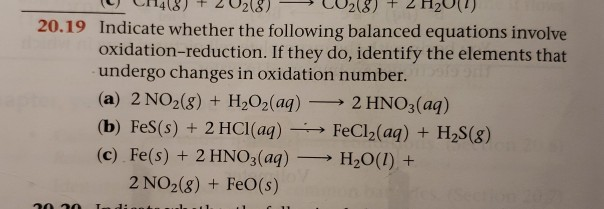
Ever found yourself mesmerized by the intricate beauty of stained glass windows, the vibrant hues of a meticulously painted ceramic, or the protective sheen on a newly forged metal sculpture? There's a subtle yet powerful force at play, a chemical ballet that transforms raw materials into lasting art. We're talking about the fascinating world of oxidation-reduction reactions, or redox for short. While it might sound like something confined to a science lab, understanding redox is like unlocking a secret superpower for anyone with a creative spark or a curious mind.
For artists and hobbyists, a grasp of redox can elevate their craft to new levels. Imagine a potter understanding how certain glazes react with oxygen at high temperatures to create stunning, iridescent finishes, or a metalsmith appreciating how controlled oxidation can produce the rich patina on bronze. Even for the casual learner, it’s a chance to see the chemistry of everyday objects and appreciate the science behind their creation and longevity. It demystifies processes, turning seemingly magical transformations into understandable, repeatable techniques. It fosters a deeper connection with the materials we use, leading to more informed and intentional artistic choices.
The applications of redox are as diverse as art itself. Think of the iconic colors in stained glass, often achieved through the addition of metal oxides that undergo redox during firing. Ceramicists utilize redox to control the color of their clay bodies and glazes, ranging from earthy reds and browns to deep blues and greens. Metalworkers employ redox for patination, creating everything from the verdigris on copper to the antique look of silver. Even the preservation of historical artifacts relies on understanding these reactions to prevent further degradation. It's a versatile principle that underpins a vast spectrum of creative endeavors.
Ready to dip your toes into the world of redox? You don't need a full laboratory! A simple way to observe redox is through rusting. Leave a piece of iron or steel outdoors, perhaps a nail or a small decorative item, and watch as it slowly transforms. The iron reacts with oxygen and water, forming iron oxides – that familiar reddish-brown rust. For a more controlled artistic experiment, consider simple metal etching using household acids (with adult supervision, of course!). Observing how the metal surface changes, often developing a different color or texture, is a direct encounter with redox. Another accessible experiment is creating your own "patina" on copper using vinegar and salt, which speeds up the natural oxidation process, creating a lovely green layer.
Ultimately, exploring redox is about appreciating the dynamic nature of matter and the beauty that can arise from chemical change. It's a journey of discovery, where understanding the 'why' behind a color, a texture, or a transformation enriches the creative process. It's the quiet magic that makes the world around us, and the art we create, so endlessly fascinating. It’s not just science; it’s the art of transformation, and it’s incredibly enjoyable to witness and understand.
