Indicate Whether Each Structure Is Aromatic Nonaromatic Or Antiaromatic
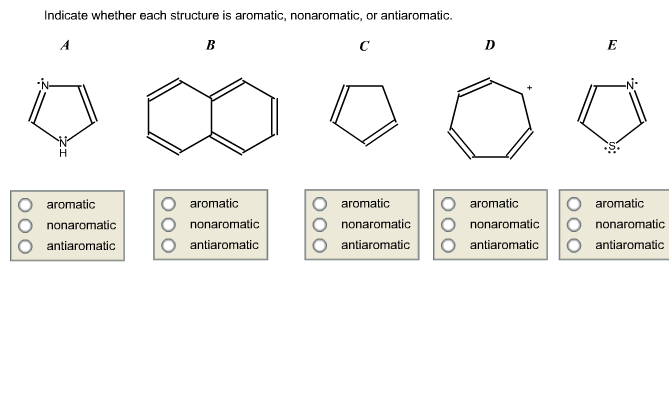
Hey there, curious minds! Ever looked at something and just felt a certain… vibe? Like how your favorite comfy t-shirt feels just right, or how that perfectly brewed cup of coffee hits all the right notes? Well, in the wacky world of chemistry, molecules have vibes too! And today, we're going to peek behind the curtain and talk about something called aromaticity. Don't let the fancy word scare you; it's actually pretty cool and, believe it or not, impacts your life in ways you might not expect.
Think of it like this: imagine a group of friends. Some friends are the super-organized, always-on-time types. They’ve got a plan for everything, and everything just… works. Then you have the free spirits, who are awesome but a little more unpredictable. And then, well, you have the folks who are just plain trouble, always stirring the pot and making things chaotic. In the molecular world, we have similar categories: aromatic, nonaromatic, and antiaromatic.
The "Chill and Stable" Gang: Aromatic Molecules
Let’s start with the MVPs, the cool cats of the chemical world: aromatic molecules. These guys are like that perfectly balanced recipe that always turns out delicious, or that timeless song that you can listen to on repeat. They’re incredibly stable and just… happy. They’ve got a special kind of ring structure, and within that ring, their electrons are doing a happy little dance.
Imagine a merry-go-round. The horses are all going in a circle, right? Aromatic molecules have a similar circular arrangement of electrons. But it's not just any circle; it's a specific kind of closed loop where a certain number of electrons are involved – usually 6. This electron-sharing party makes them incredibly stable, like a perfectly constructed domino chain that won't easily fall over.
Why should you care about these stable molecules? Because many of the things you encounter daily are aromatic! Think about the smell of your morning coffee. That delightful aroma? A lot of it comes from aromatic compounds like caffeine. Or how about the vibrant colors in a painting? Many pigments are also aromatic molecules, giving us the beautiful spectrum we see.
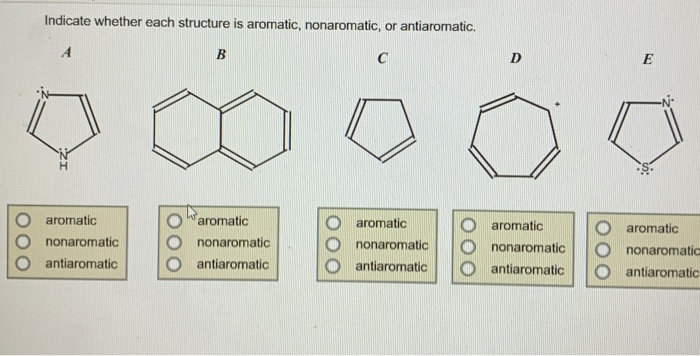
Take benzene, the poster child for aromaticity. It’s a simple ring of six carbon atoms with alternating double bonds. It’s not just some abstract chemical; it's a building block for countless other important molecules. It's like the foundational ingredient in a favorite family recipe – simple, but essential for the delicious end result.
The "Just Living Their Best Lives" Crowd: Nonaromatic Molecules
Now, let’s talk about the majority of molecules out there: the nonaromatic ones. These are like the friendly neighbors who are perfectly pleasant, but they don’t have any particularly outstanding qualities. They’re not super stable, but they’re not actively trying to cause a fuss either. They’re just… there, doing their thing.
These molecules might have rings, but they don't have that special, circular electron-sharing going on that aromatic compounds do. Some might have the right shape but are missing the right number of electrons, or vice-versa. It’s like a group of friends trying to play a board game but someone forgot the dice, or someone brought an extra player who doesn't know the rules. It just doesn't quite come together in that perfect, stable way.

Think about a simple chain of carbon atoms, like in a saturated fat. It’s stable enough, but it doesn’t have that extra flair. Or consider a molecule that has a ring but one of the atoms in the ring is sp3 hybridized (meaning it's more like a tetrahedral structure, not flat enough for that perfect circle). It breaks the flow, and the electrons can't get into that harmonious dance.
So, why are these nonaromatic molecules important? Because they’re everywhere! They make up a huge chunk of the stuff around us. Your plastic water bottle? Likely contains many nonaromatic components. The fuel in your car? Nonaromatic hydrocarbons are a big part of that. They’re the workhorses, the reliable, everyday materials that make our modern lives possible.
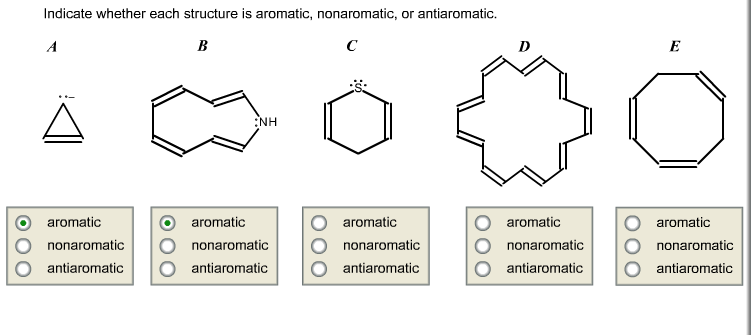
The "Drama Magnets": Antiaromatic Molecules
Finally, we come to the molecules that are the chemical equivalent of a gossip session or a reality TV show – the antiaromatic ones! These guys are the opposite of stable. They are incredibly unstable and just hate being in their ring formation. They're like that one friend who always seems to be on the verge of a meltdown, or a house of cards just waiting for a slight breeze to topple it.
These molecules have the right ring structure and the right number of electrons, but there’s a catch: they have too many electrons for that specific setup. Instead of a harmonious dance, it's more like a mosh pit. These extra electrons are forced into higher energy levels, making the molecule very reactive and eager to change into something else. They’re constantly looking for a way out of their unhappy situation.
Imagine a tiny room packed with way too many people. Everyone is uncomfortable, bumping into each other, and just wanting to get out. That's an antiaromatic molecule. They have a specific number of electrons in their ring – usually 4 – that makes them highly unstable. They’re like a tightly wound spring, ready to release energy at the slightest provocation.
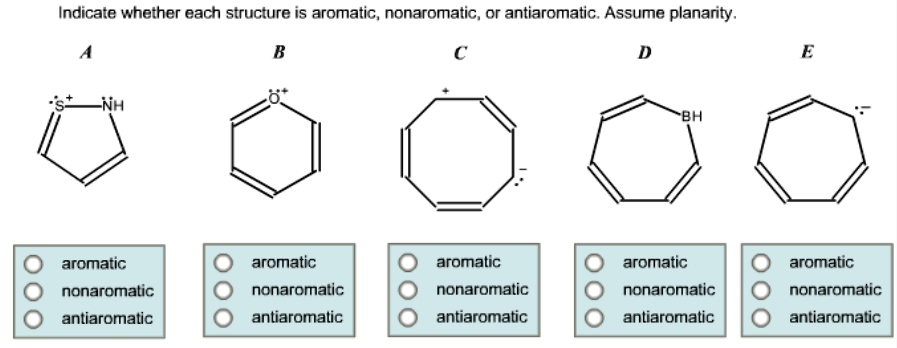
While you won’t typically find these unstable guys hanging around for long in your everyday life, their tendency to react is actually super important in chemical reactions. They’re the ones that might get ‘used up’ to form more stable compounds. Think of them as the volatile ingredients that, when handled carefully, can lead to something useful, but left to their own devices, can be a bit of a mess.
Putting It All Together
So, we've got our aromatic friends: stable, harmonious, and found in things like coffee and dyes. We have our nonaromatic crowd: the everyday workhorses of the molecular world. And then there are the dramatic, unstable antiaromatic molecules, who are usually eager to change.
Why should you remember this little bit of molecular gossip? Because understanding these differences helps chemists design new materials, create better medicines, and understand how the world around us works at its most fundamental level. It’s about recognizing the "vibe" of a molecule, and that vibe tells us a lot about its personality and how it will behave. So next time you’re enjoying that fragrant flower or marveling at a sturdy building, remember the hidden world of aromaticity, nonaromaticity, and antiaromaticity, all playing their part!
