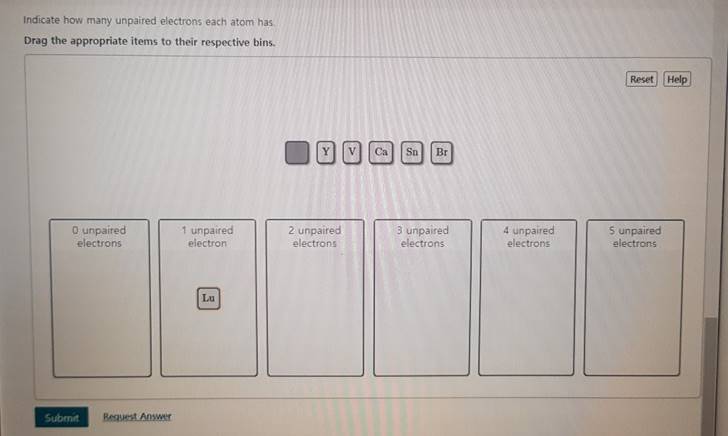
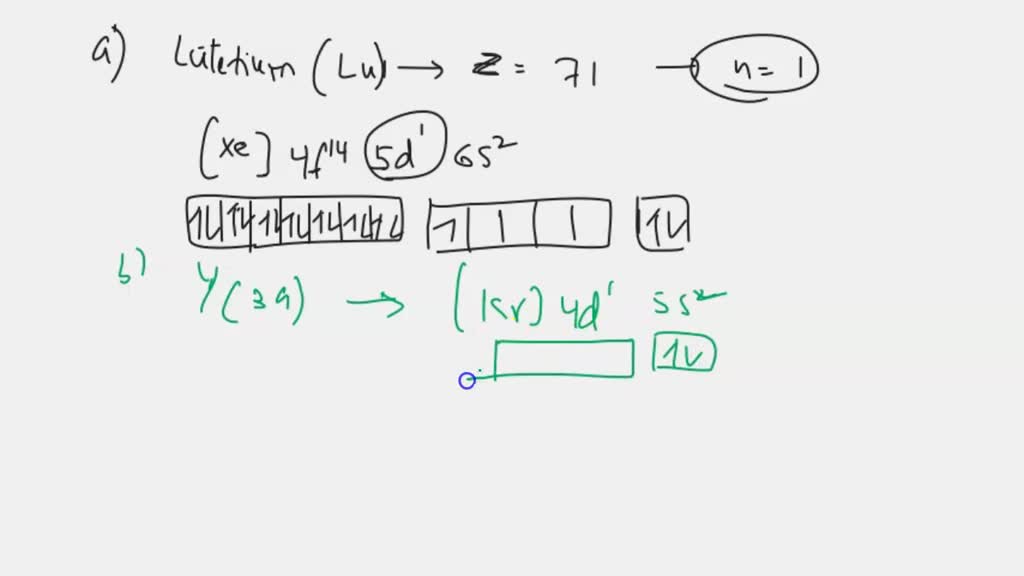
Indicate How Many Unpaired Electrons Each Atom Has.
Okay, let's talk about something you probably haven't thought about since that one chemistry class you barely passed. We're diving into the wonderful, wild world of unpaired electrons. Think of them as the lone wolves of the atom world. The rebels. The ones who haven't quite found their dance partner. And honestly, I think they get a bad rap.
So, how many of these mischievous little guys does each atom have hanging out? It's not as scary as it sounds. Imagine a bunch of shy people at a party. Some are already in cozy pairs, chatting away. Others are standing by the snack table, awkwardly looking around. Those are our unpaired electrons. They're available, ready for… well, whatever atoms do when they're available.
Let's start with the most basic. Hydrogen. This little guy is practically the poster child for being a lone wolf. It's got one electron, and it's all by itself. Zero pairs. One unpaired electron. It's like the person who shows up to the party solo, looking for someone to talk to. Poor Hydrogen. Always looking for a friend.
Then we have Helium. Now, Helium is the opposite. It's the life of the party, all snuggled up. It has two electrons, and they're happily paired. Zero unpaired electrons. Helium is all about chilling. It's the atom that's already found its soulmate. It doesn't need any drama. It's done.
Moving on to Lithium. This one's got a bit of a split personality. It's got three electrons. Two of them are like a bonded couple, all cozy. But the third one? Yep, it's out there, looking for a connection. One unpaired electron. Lithium is like, "I'm comfortable, but I'm also open to possibilities." It's the sophisticated single at the party, enjoying its own company but not ruling out a good conversation.
Carbon is where things get really interesting. Carbon is the ultimate social butterfly, or maybe the ultimate indecisive one. It has four electrons in its outer shell. It's got two pairs already chilling. But then it has two more electrons that are just… there. Waiting. Two unpaired electrons. Carbon is like, "I'm good, but I could be better. I'm open to multiple relationships, you know?" It’s the atom that can form all sorts of bonds, which makes sense, given its electron situation. It’s the Swiss Army knife of the periodic table.
And then there’s Nitrogen. Nitrogen is a bit more reserved, but still has some spice. It has five electrons in its outer shell. It manages to pair up two of them. But that leaves three electrons all by themselves. Three unpaired electrons. Nitrogen is the one with a strong inner circle, but also three potential plus-ones. It's got a whole vibe going on. It’s like, "I have my crew, but I'm definitely bringing backups."
Now, let's talk about Oxygen. Oxygen is a bit more eager to mingle than Nitrogen. It has six electrons in its outer shell. It forms two pairs, leaving two electrons to wander. Two unpaired electrons. Oxygen is like, "Alright, I've got my comfort zone, but I’m definitely ready for more action. Two is a good number for opportunities." It’s the friend who’s ready for a double date.

And finally, Fluorine. Fluorine is the one who's almost there. It has seven electrons. It pairs up three of them. But that leaves one lonely electron. One unpaired electron. Fluorine is so close to being perfectly content. It's like the person who’s almost finished their networking event, but there’s still that one last person they want to meet. It’s got that intense focus, that one goal in mind. It’s the atom that’s famously reactive, and you can see why!
Honestly, I think we should all be a little more like these atoms with unpaired electrons. Be open to new connections. Don't be afraid to be a little bit "available." Life's too short to be perfectly paired up all the time. Sometimes, the most interesting things happen when you're the lone wolf, looking for a spark.
Solved Indicate how many unpaired electrons each atom has. | Chegg.com
Think about it. Neon? Another one of those chill, happy-with-itself atoms. Eight electrons, all perfectly paired. Zero unpaired electrons. Neon is just vibing. It’s the atom that’s truly content. It doesn’t need to prove anything. It's just… there. And that's okay!
What about the heavier elements? They're just playing the same game, but with more players. Sodium, for example, is like a Lithium situation. One unpaired electron. It's ready to shed that one extra to feel complete. It's the atom that's eager to share. And Chlorine? It's the fluorine friend, looking for that one last partner to make a perfect pair. One unpaired electron.
It’s a whole ecosystem of electron relationships out there. Some are perfectly content in their pairs, like Helium and Neon. Others are the ultimate networkers, like Carbon, ready to make all sorts of connections. And some are just one shy of perfection, like Fluorine and Chlorine, desperately seeking that last partner. And I, for one, find it all rather… charming. These unpaired electrons, they’re not a sign of being incomplete. They’re a sign of potential. Of possibility. Of being ready for whatever comes next. So, next time you see a molecule, maybe give a little nod to the unpaired electrons. They’re the unsung heroes of chemical bonding, and, dare I say, a little bit of life.

