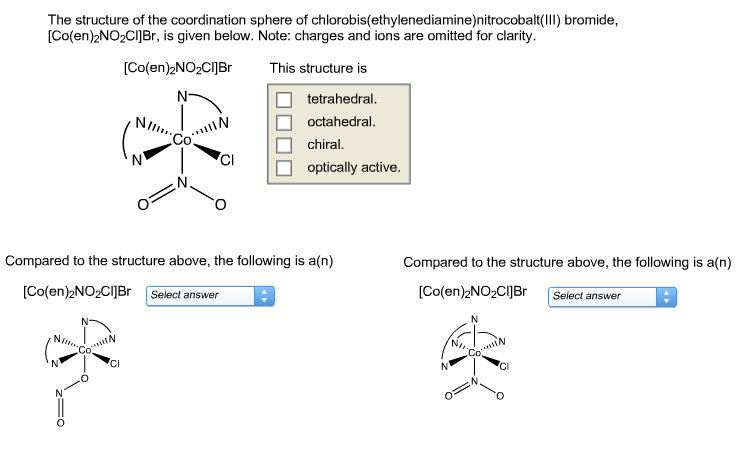
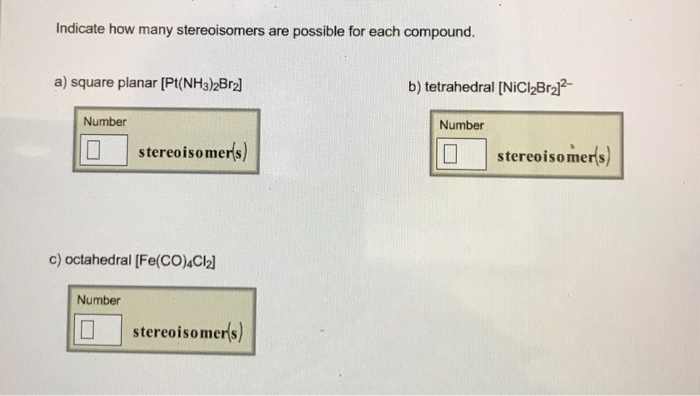
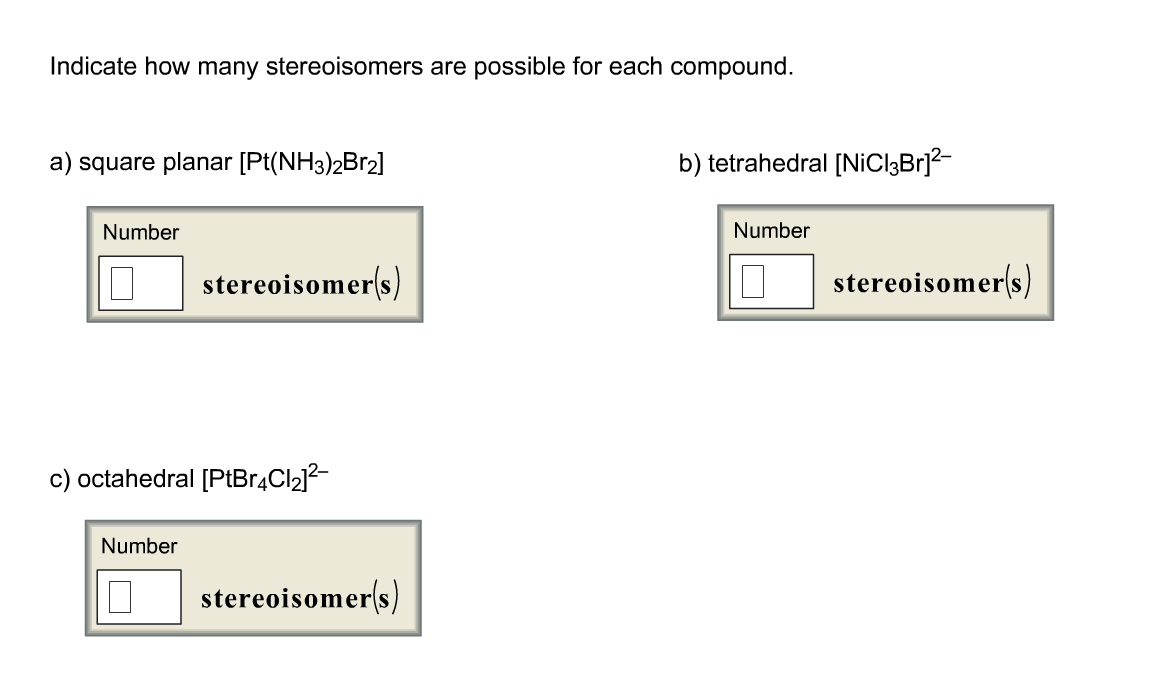
Indicate How Many Stereoisomers Are Possible For Each Compound
Hey there, curious minds! Ever looked at a molecule and thought, "Huh, that looks like it could be two things at once?" Well, buckle up, buttercups, because we're about to dive into a world where molecules can play dress-up and come in different, yet related, forms. We're talking about stereoisomers, and trust me, it's not as scary as it sounds. In fact, it's pretty darn cool and can make understanding chemistry a whole lot more fun!
So, what exactly are these sneaky stereoisomers? Imagine you have a Lego creation. Now, imagine you can take that same set of Legos and build a slightly different version that's a mirror image of the first, or maybe it's just twisted in a different way. That, my friends, is the essence of stereoisomers. They're compounds that have the same molecular formula and the same connectivity (meaning the atoms are connected in the same order), but they differ in their three-dimensional arrangement in space. Mind. Blown. Right?
Why should you care about molecules playing peek-a-boo in 3D? Well, it turns out that these subtle differences in how atoms are arranged can have a HUGE impact. Think about your own hands. They're mirror images of each other, right? But you can't exactly wear your left glove on your right hand, can you? Molecules can be like that too! Some stereoisomers might be super useful, like medicines that fit perfectly into a receptor in your body, while others might be… well, not so helpful. It’s like having the right key for the lock – and the wrong one just won’t do!
Now, let's get to the nitty-gritty: how do we figure out how many of these isomer buddies a molecule can have? This is where the magic number comes in. For each compound, there's a specific way to calculate the maximum possible number of stereoisomers. Don't worry, we're not going to get bogged down in complex calculations here, but the underlying principle is fascinating.
The Chirality Capers!
The main troublemaker, or rather, the main creator of stereoisomers, is something called chirality. A chiral molecule is like a hand – it's not superimposable on its mirror image. Think of it as having a "handedness." The most common way to create a chiral center in a molecule is to have a carbon atom that's bonded to four different groups. We call this a chiral center or a stereocenter.

If a molecule has one chiral center, it's guaranteed to have two stereoisomers. These are called enantiomers. They are like your left and right hands – non-superimposable mirror images. Pretty neat, huh? It's like having a twin, but a reflection twin!
What if our molecule gets a little more complicated and decides to have two chiral centers? Now things get even more interesting! The maximum number of stereoisomers can go up to four. So, we might have a pair of enantiomers, and then another pair of enantiomers. But wait, there's a catch! Sometimes, due to symmetry within the molecule, one of these potential stereoisomers might actually be identical to its mirror image. This is called a meso compound, and it's like a molecule that's its own reflection, even though it has chiral centers. It's a clever little shortcut the molecule takes!
A Few Fun Examples!
Let’s spice things up with some examples, shall we? We won't draw them out, but we'll talk about them. Imagine a simple molecule with a single chiral center. Let's say it's like a little carbon atom with a hydrogen, a chlorine, a bromine, and a methyl group attached. Because that carbon has four different things stuck to it, it's a chiral center. And guess what? This molecule can exist as two stereoisomers – an enantiomeric pair!
Now, picture a slightly bigger molecule, maybe something with two chiral centers. For instance, consider a molecule like tartaric acid. It can have up to four stereoisomers! Two of these are enantiomers (like R,R and S,S configurations), and then there's a meso compound (R,S configuration that’s superimposable on its mirror image, S,R). It's like a party with a few different dancing partners!
The general rule of thumb for the maximum number of stereoisomers for a compound is 2n, where 'n' is the number of chiral centers. So, if a molecule has 3 chiral centers, it could potentially have 23 = 8 stereoisomers! Isn't that a wild thought? A relatively small molecule could have eight different three-dimensional arrangements.
When Less is More (or Just Different!)
But remember that symmetry thing we talked about? Sometimes, a molecule with chiral centers might not reach the full 2n count. If a molecule has a plane of symmetry that goes right through it, even with chiral centers, it might be a meso compound. This means it's achiral, and its mirror image is identical. It's like finding a perfectly balanced seesaw. So, the actual number of distinct stereoisomers can sometimes be less than the theoretical maximum.

This is where chemistry gets really intriguing! It’s not just about the atoms and how they’re connected, but also about their precise spatial orientation. It’s like art, but with atoms! Understanding stereoisomers helps us appreciate the incredible diversity and complexity that can arise from seemingly simple building blocks.
So, why is this so inspiring? Because it shows us that even small changes can lead to big differences. It’s a reminder that in nature, and in science, there’s always more to explore, more to understand. The world of molecules is a vast, intricate, and beautiful place, full of possibilities. Learning about stereoisomers isn't just memorizing rules; it's about unlocking a deeper appreciation for the elegance and complexity of the universe around us.
So, the next time you encounter a chemical structure, take a moment to consider its potential for stereoisomers. Think about the different ways it could twist and turn in 3D space. It’s a fantastic way to train your brain to think spatially and to truly appreciate the art of chemistry. Keep exploring, keep questioning, and keep finding the wonder in the molecules!
