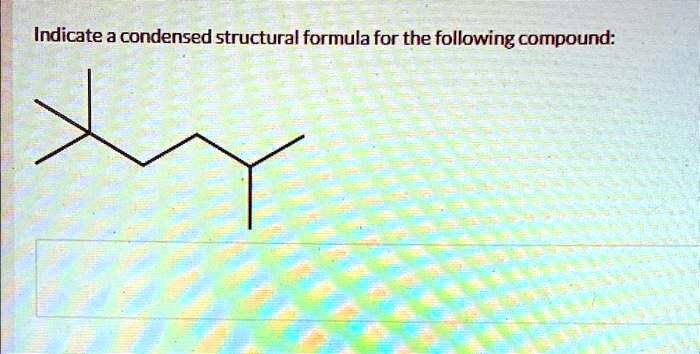
Indicate A Condensed Structural Formula For The Following Compound:
Ever glanced at a complex chemical name and felt a little overwhelmed? You're not alone! But what if I told you there's a way to represent these intricate molecules in a surprisingly neat and tidy fashion? Today, we're diving into the world of condensed structural formulas, a clever shortcut that makes chemistry a whole lot more approachable and, dare I say, even fun.
Think of it like this: instead of drawing out every single atom and bond like a meticulous architect, a condensed structural formula is more like a shorthand note. It strips away some of the visual clutter while still giving you all the essential information about how atoms are connected. Its purpose is simple: to make it easier and quicker to write and understand the structure of organic molecules, which are the building blocks of life and countless materials we use every day.
The benefits are pretty significant. For starters, it saves a ton of space on paper or a screen. Imagine trying to draw the structure of a long, complex organic molecule with every single bond clearly shown. It would be enormous! Condensed formulas allow us to represent these same molecules in a much more compact way. This makes them ideal for textbooks, scientific papers, and even quick notes between chemists. Beyond just saving space, they help us focus on the functional groups – the specific arrangements of atoms that determine a molecule's properties and reactivity. Recognizing these groups is a key skill in chemistry, and condensed formulas highlight them beautifully.
You'll encounter condensed structural formulas all over the place once you know what to look for. In your high school or college chemistry classes, they're a standard way to represent organic compounds. But their influence extends beyond academia. Think about the medicines you take – their active ingredients are complex organic molecules, and their structures are often communicated using condensed formulas. Even in the food industry, understanding the structure of flavor compounds or preservatives can involve these handy representations.
So, how can you start exploring this yourself? It's easier than you might think! The best way to get a feel for condensed structural formulas is to start with simple examples and gradually work your way up. Look at the structure of methane (CH4) or ethane (C2H6). Then, try to find the condensed formula for propane (CH3CH2CH3). You'll notice a pattern emerge: the central carbon atoms are shown, and the hydrogen atoms attached to them are grouped together. For more complex molecules, you might see parentheses used to indicate branched groups. For example, an isopropyl group might be shown as -CH(CH3)2. Don't be afraid to look up common organic molecules and see how their condensed formulas are written. Websites dedicated to chemistry often have extensive databases. With a little practice, you'll find yourself intuitively understanding these representations and appreciating the elegance of chemical shorthand!
