In Which State Of Matter Are Molecules Moving The Slowest

You know, I had this brilliant idea the other day while I was trying to make my morning coffee. I was watching the little sugar crystals tumble into the hot water, and for a split second, they seemed to just… linger. Like they were having a little existential crisis before they decided to fully dissolve. It got me thinking about movement, about how things, even tiny things like molecules, are always on the go. And then I thought, okay, if they're always moving, what about when they're moving the slowest?
It's a weird question, right? We usually think about things either being still or being pretty darn active. Like my dog, Buster. He's either sprinting around the park like a furry bullet or he's deeply, profoundly asleep, twitching his paws as if he's chasing squirrels in his dreams. There's not much in between. But with molecules? Oh boy, that's where things get really interesting.
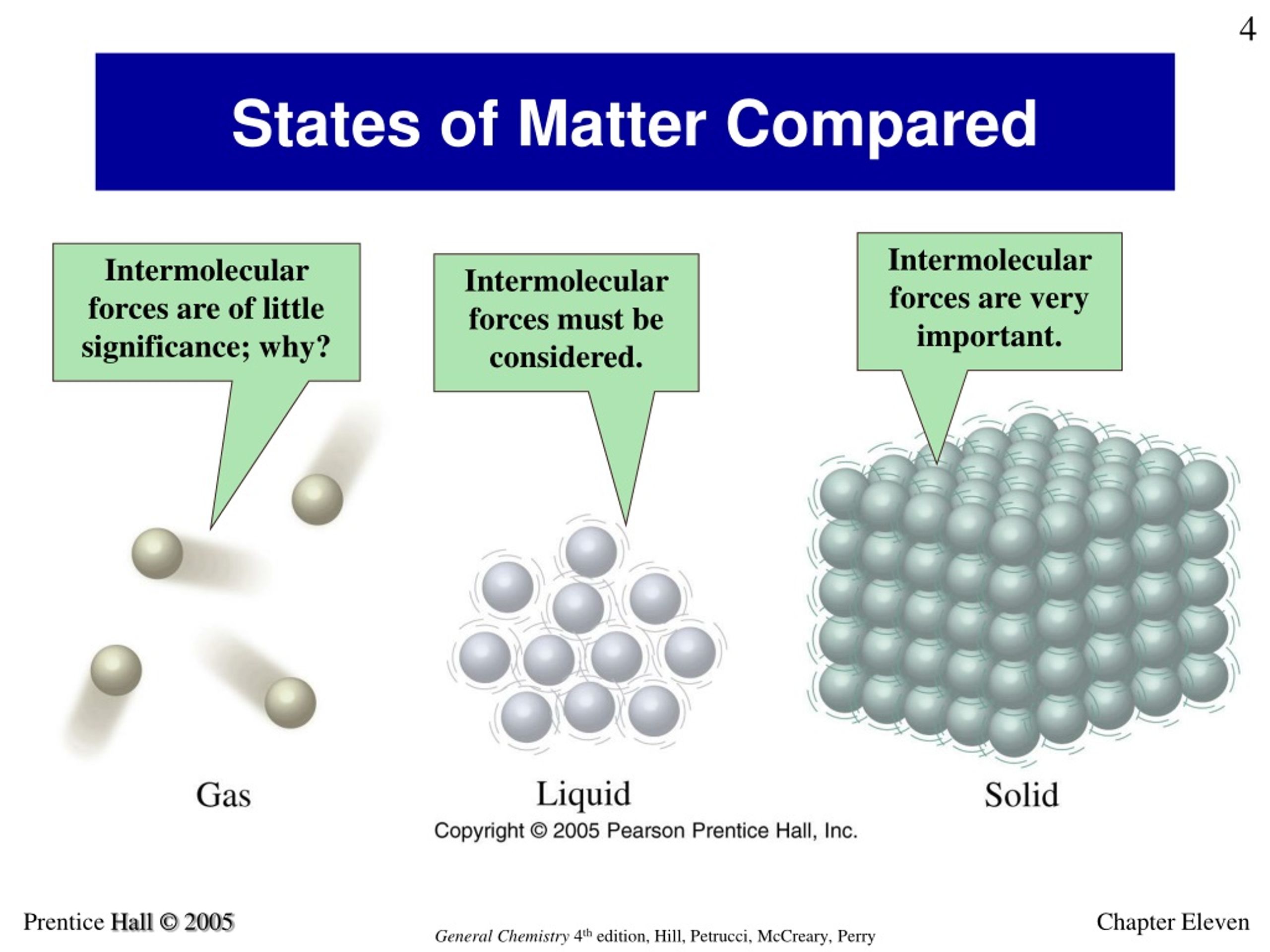
So, let's dive into this whole molecular movement thing. We're going to be talking about states of matter, which I'm sure you remember from science class. You know, solid, liquid, gas? Those trusty old friends. They’re the big categories that help us understand the world around us, from the ice in our drinks to the air we breathe.
And at the heart of it all are these microscopic little dudes called molecules. They're the building blocks of everything. Seriously, everything. That coffee mug you’re holding? Molecules. Your phone? Molecules. Even that slightly embarrassing photo of you from last year? Yep, molecules. And these molecules are constantly doing a little jig, a waltz, or a full-on rave, depending on their situation.
The key difference between these states of matter, and the reason our sugar crystals seemed to hesitate, lies in how much energy these molecules have. Think of energy like, well, energy. When they have a lot of energy, they're bouncing off the walls, practically vibrating with excitement. When they have very little energy, they're… well, they're a bit more chill. A lot more chill, actually.
The Case of the Lively Gas
Let's start with what you might think of as the most energetic. We’ve got gases. Imagine a room full of hyperactive toddlers who’ve just discovered a bag of marshmallows. That’s kind of what molecules are like in a gaseous state. They’ve got tons of energy, they’re zooming around in all directions, bumping into each other, and basically spreading out to fill whatever container they're in. They have no definite shape and no definite volume, because they’re too busy having their own little molecular party.
Think about steam rising from a boiling kettle. Those water molecules are absolutely flying. They’ve absorbed so much heat energy that they’ve broken free from their liquid bonds and are now off exploring the vastness of the kitchen. They're so spread out and moving so fast that you can't even see them individually. It’s a beautiful, chaotic dance of pure kinetic energy.

And the speed? We’re talking hundreds of meters per second. It’s mind-bogglingly fast. If you were a gas molecule, you could probably get from one end of your room to the other in the blink of an eye. Pretty wild, huh?
It's this high-energy movement that allows gases to compress and expand so easily. They’re not packed tightly together, so there’s plenty of room for them to get squished or to spread out. It’s why balloons inflate and why you can smell your neighbor’s questionable cooking from across the street.
The Fluidity of Liquids
Next up, we have liquids. These guys are a bit more restrained than gases, but they’re still far from being sedentary. Think of a busy sidewalk on a Saturday afternoon. People are moving, they’re interacting, they’re bumping shoulders occasionally, but they’re generally staying within the confines of the pavement. They’re not running wildly in every direction like in the gas phase.
Liquid molecules have enough energy to slide past each other, but not enough to completely escape each other’s influence. They have a definite volume, but they take the shape of their container. That’s why water fills your glass but doesn't suddenly decide to become a sphere. The molecules are still interacting, holding onto each other a bit, but they have enough freedom to flow.
The speed of liquid molecules is significantly less than that of gas molecules. They’re still moving, don’t get me wrong, but it’s more of a flowing, tumbling motion rather than a free-for-all sprint. Imagine a crowd of people milling around in a room. They’re moving, but they’re not exactly breaking speed records. They’re interacting and flowing, but with a bit more cohesion.

This is why liquids are great for things like carrying substances or transferring heat. The molecules can move around and interact with whatever they’re carrying or heating up. It’s a nice balance between order and movement. Not too wild, not too still. Just right, like Goldilocks’ porridge.
The Surprisingly Still World of Solids
Now, this is where we start to get to the heart of our question. We’re talking about solids. Think of a perfectly organized bookshelf. Each book is in its place, not moving from its position on the shelf. Sure, there might be a slight tremor if you bump the shelf, but the books themselves aren't wandering around.
In a solid, molecules are packed very tightly together in a fixed, ordered structure. They don't have the energy to move past each other or to break free from their bonds. Instead, they are mostly just vibrating in place. They're jiggling, oscillating, and doing a little shimmy, but they're essentially stuck in their spots.
Imagine a tightly packed mosh pit where everyone is just jumping up and down in their own little space, but nobody is actually moving forward or backward. That’s a pretty good analogy for the molecular motion in a solid. The molecules are full of energy, but that energy is primarily expressed as vibration, not as translation (moving from one place to another).
The speed of these vibrations is much, much slower than the speeds seen in liquids and gases. They are essentially at their slowest in terms of overall positional movement. They’re not going anywhere, they’re just… jiggling. It’s a surprisingly quiet kind of motion.
This is why solids have a definite shape and a definite volume. The molecules are held in rigid positions, and they can’t easily be compressed or expanded because there's no space between them for movement. Think of a block of ice – it keeps its shape and doesn't just spread out. The water molecules are locked in place.
The Ultimate Chill: Absolute Zero
But wait, can it get even slower? Oh, absolutely! We’re talking about the ultimate state of molecular chill: absolute zero. This is the theoretical point at which all molecular motion would cease. No vibration, no movement, nothing. It's the ultimate stillness.
Now, here's a fun fact: we can get really, really close to absolute zero, but we can never actually reach it. It's like trying to catch your own shadow – you can get close, but you can never quite grab it. Scientists have managed to cool things down to incredibly low temperatures, just fractions of a degree above absolute zero. At these temperatures, molecular motion is so minuscule it’s practically undetectable.
At these near-absolute-zero temperatures, strange and wonderful things start to happen. We get phenomena like superconductivity (where electricity flows with zero resistance) and superfluidity (where liquids flow without any friction). It’s like the molecules, in their extreme stillness, start behaving in completely unexpected and amazing ways.
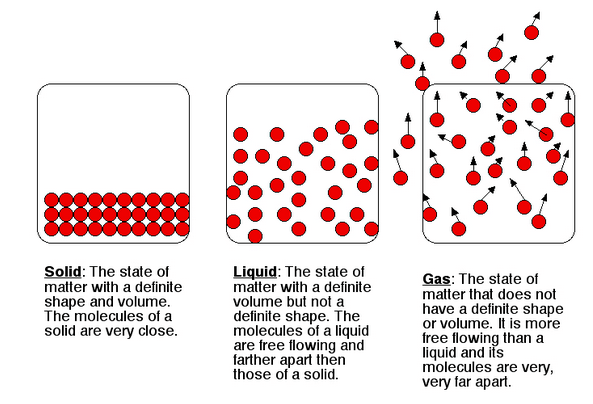
So, to directly answer the burning question: in which state of matter are molecules moving the slowest? It's the solid state. And if we're talking about the theoretical absolute slowest movement, it's at absolute zero, where motion would ideally stop altogether.
Think back to that sugar crystal in my coffee. When it's a solid chunk, those water molecules within it are just vibrating. Once it hits the hot water and starts to dissolve, it becomes a liquid, and those molecules get a little more freedom to move. But the solid form, the un-dissolved crystal? That’s where the real stillness resides.
It’s fascinating, isn’t it? How a simple change in temperature, a little bit of energy added or removed, can completely transform the behavior of matter. From the frantic dance of gases to the flowing currents of liquids, and finally to the quiet, contained jiggles of solids. It all comes down to the energy of those tiny, invisible molecules.
So next time you’re holding a piece of ice, or watching steam rise, or even just looking at a solid object, take a moment to appreciate the incredible, silent dance of its molecules. They’re always moving, but in solids, they’re doing it at the slowest, most serene pace.
And who knows, maybe if you concentrate really hard, you can feel the faint vibrations of your own molecules. Probably not, but it's a fun thought experiment, right? Just remember, the slower the movement, the more structured and stable the matter. It’s a fundamental principle of the universe, happening all around us, all the time.
