In Which Species Does Nitrogen Have The Highest Oxidation Number

Ever wondered about the secret lives of atoms? Specifically, nitrogen? This element is a real chameleon. It can be found in so many different forms, and its "oxidation number" can change like a celebrity's outfit.
Think of oxidation numbers like a score. It tells you how many electrons an atom has gained or lost. Nitrogen is a master of this game. It can be a giver, a receiver, or even play both sides!
But where does nitrogen truly shine? Where does it hit its highest possible oxidation score? It's a question that might sound a bit niche, but trust us, it leads to some seriously cool discoveries.
The answer isn't some obscure, hard-to-pronounce compound. It's actually found in a surprisingly common place. You've probably seen it, maybe even used it. It's a chemical that's crucial for many processes.
The champion of high oxidation numbers for nitrogen is in a molecule called nitric acid. Yep, that's right. This everyday acid is where nitrogen really shows off its electron-losing prowess.
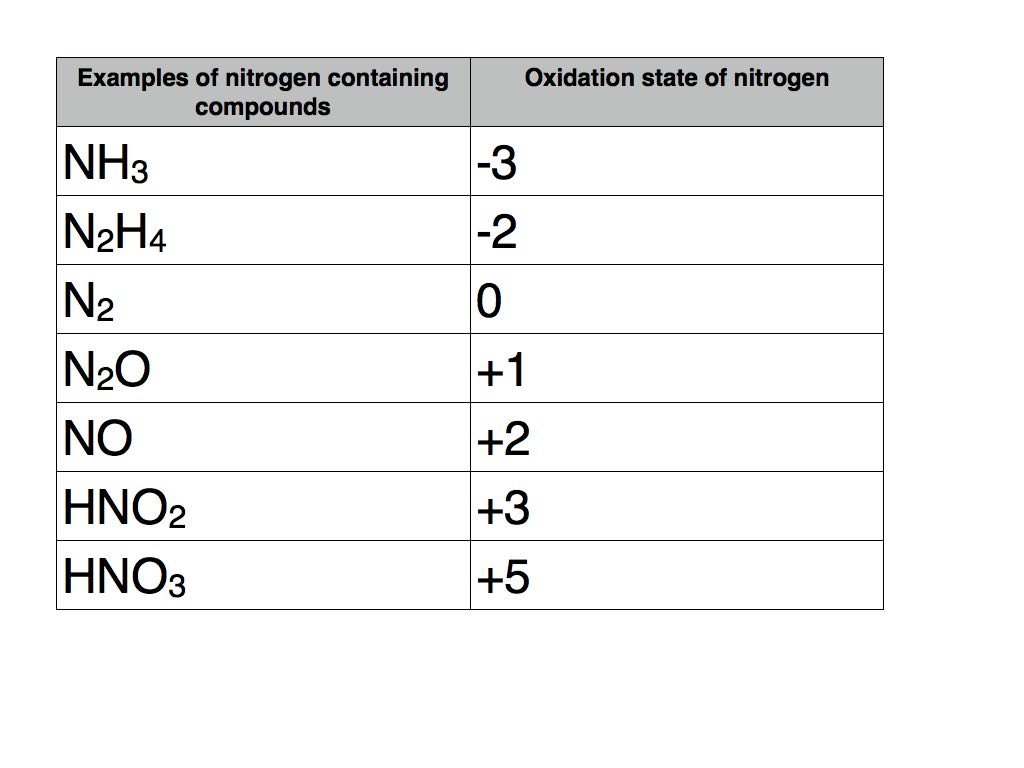
In nitric acid, nitrogen achieves an oxidation number of +5. That's a big number in the atomic world! It means nitrogen has pretty much given away all of its outer electrons. It's like it's completely "oxidized."
Why is this so exciting? Because it shows the incredible versatility of nitrogen. This element is everywhere. It's in the air we breathe, the food we eat, and even in our DNA.
And the fact that it can adopt such a high oxidation state in something like nitric acid means it plays a vital role in many chemical reactions. It's a key player in industry and nature.
Imagine nitrogen as a super-powered athlete. Sometimes it's sprinting (low oxidation numbers), sometimes it's lifting heavy weights (high oxidation numbers). Nitric acid is like its Olympic gold medal moment.

So, what makes this particular species, nitric acid, so special? It's all about the dance of electrons. In nitric acid, nitrogen is bonded to three oxygen atoms.
Oxygen is a very greedy element when it comes to electrons. It pulls them towards itself. This strong pull from the oxygen atoms forces nitrogen to give up its electrons.
The more oxygen atoms around, the more nitrogen has to "donate." In nitric acid, there are enough oxygen atoms to convince nitrogen to hand over almost all its spare electrons.
This results in that impressive +5 oxidation state. It’s a testament to the power of chemical bonding and the influence of neighboring atoms.
Now, you might be thinking, "Is this something I need to worry about?" Not really! While nitric acid can be corrosive, it's also incredibly useful.
It's used in making fertilizers, which help grow our food. It's also used in making explosives, though hopefully you won't encounter those very often! And it's involved in processes that create dyes and medicines.
The fact that nitrogen can reach such a high oxidation number in nitric acid highlights its importance in chemistry. It's a fundamental property that drives many important chemical transformations.
Think about it: the same element that makes up a huge part of our atmosphere also plays a crucial role in industrial processes and even in the creation of life-saving drugs. It's a true multitasker.
Other nitrogen-containing compounds have different oxidation numbers. For example, in ammonia (NH₃), nitrogen has an oxidation number of -3. This is the opposite extreme!
In compounds like nitrous oxide (N₂O), nitrogen might have an oxidation number of +1. In nitrogen dioxide (NO₂), it could be +4. There's a whole spectrum of possibilities.
But the pinnacle, the absolute highest score, belongs to nitric acid. It's like nitrogen is saying, "Look at what I can do!" It’s a statement of its chemical flexibility.
So, next time you hear about nitric acid, remember the nitrogen inside. Remember its incredible journey to achieve that +5 oxidation number. It’s a tiny detail, but it’s part of a grander chemical story.
It's this variability that makes studying elements like nitrogen so endlessly fascinating. They are not static; they are dynamic. They interact and change based on their surroundings.

And the species where nitrogen reaches its highest oxidation number, nitric acid, is a perfect example of this chemical dynamism. It’s a molecule that’s both common and incredibly powerful.
Understanding these oxidation states helps scientists predict how chemicals will react. It's like having a secret code to the chemical world. And the highest oxidation state for nitrogen is a particularly exciting clue.
It’s a reminder that even in the seemingly simple world of atoms, there are complex and captivating stories unfolding. Nitrogen’s journey to +5 is one of them.
So, the next time you encounter nitric acid, give a little nod to the nitrogen within. It’s a small atom that achieves big things, reaching its maximum potential in this powerful compound. It’s a true chemical rockstar.
Isn't it amazing that such a fundamental concept in chemistry can be found in something so tangible? It connects the microscopic world of atoms to the macroscopic world we live in.
The quest for understanding oxidation numbers is ongoing. Scientists are always discovering new compounds and new reactions. But for now, nitric acid holds the title for the highest nitrogen oxidation number.

It’s a small victory for nitrogen, perhaps. But it’s a huge insight for chemistry. It shows us the incredible range of behavior that a single element can exhibit.
This little tidbit about nitric acid is a great starting point for anyone curious about chemistry. It’s a simple question with a surprisingly interesting answer that leads to even more questions.
So, feel inspired! Look up more about nitric acid. Explore other nitrogen compounds. You might find yourself captivated by the endless possibilities of the atomic world.
It’s a journey that’s both educational and surprisingly entertaining. And it all starts with a humble element and its quest for the highest oxidation number.
The species where nitrogen achieves its highest oxidation number is indeed nitric acid. It's a testament to nitrogen's versatility and its ability to engage in powerful chemical interactions.
The story of nitrogen's oxidation numbers is like a soap opera, full of drama and unexpected twists. Each compound is a new episode, showcasing nitrogen in a different role.
From the humble ammonia to the potent nitric acid, nitrogen transforms. It’s a chemical actor with an impressive range. And its highest oxidation state is a standing ovation moment.
So, if you ever want a little spark of chemical curiosity, think about nitrogen. Think about its journey to +5. It’s a small piece of a huge and wonderful puzzle.
