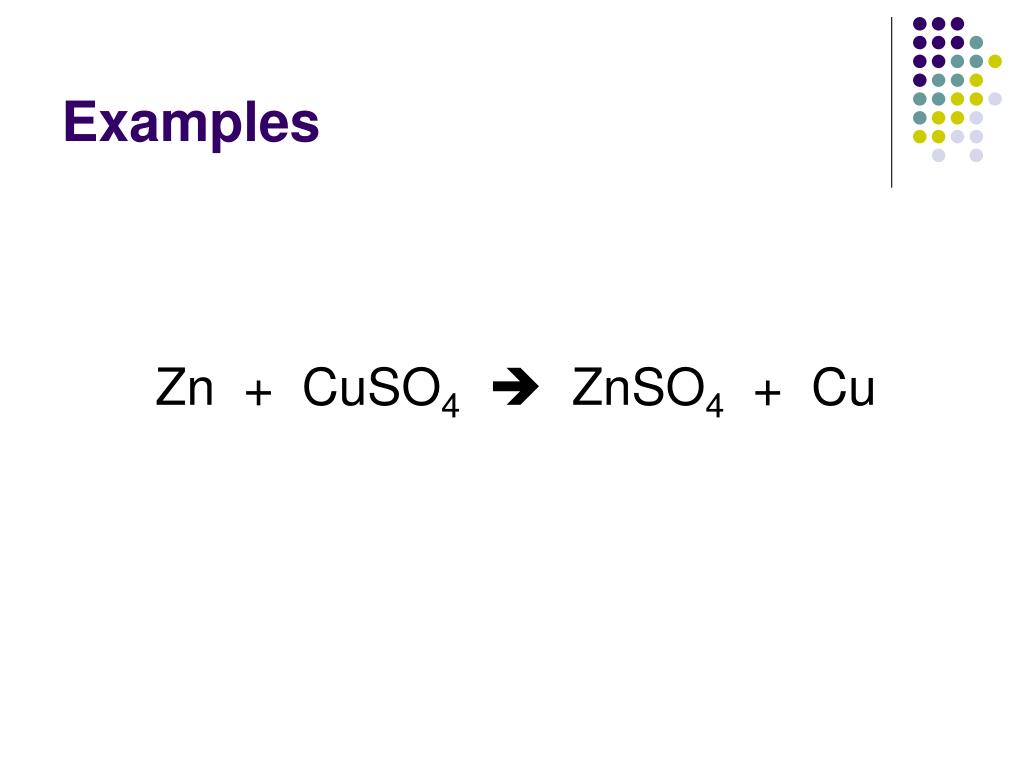
In The Reaction Cuso4+zn-znso4+cu The Oxidizing Agent Is
Hey there, science curious folks! Ever wondered if chemistry can be, well, a little bit dramatic? Like a tiny chemical play unfolding right before your eyes? Because sometimes, it really is! We’re talking about reactions that have a clear hero, a clear villain, and a whole lot of excitement. And today, we're spotlighting a real crowd-pleaser in the chemical world.
Let’s dive into a situation that’s as cool as it is colorful. Imagine you have a beaker of copper sulfate, that lovely blue stuff you sometimes see in science experiments. It looks pretty, right? Like a magical potion. Now, imagine you drop a piece of zinc metal into it. What happens next? Get ready for a bit of a show!
This isn’t just some boring mix-and-stir. Oh no. This is where the action heats up. The copper sulfate and the zinc get together, and things start to change. The vibrant blue of the copper sulfate solution begins to fade. And guess what starts to appear? Tiny, beautiful, reddish-brown specks. That’s the copper that used to be dissolved in the solution, deciding it’s had enough of being a liquid and wants to be a solid metal again. It’s like the copper is saying, "Okay, I'm out!"
But wait, there’s more! As the copper forms, something else is happening. The zinc, which was just a plain old gray metal rod or chunk, starts to disappear. It’s not vanishing into thin air, though. It’s transforming. It’s becoming part of a new partner: zinc sulfate. Think of it as the zinc getting a new, fashionable outfit made of sulfate. It’s a complete makeover!
So, we have copper sulfate + zinc giving us zinc sulfate + copper. Sounds simple, but the magic lies in the how. Who’s pulling the strings here? Who’s making all these dramatic transformations happen? This is where we introduce our star player, the oxidizing agent.

In this electrifying dance of elements, the oxidizing agent is the one that takes electrons. It’s like a gracious host at a party, accepting gifts from its guests. And in our copper sulfate and zinc show, that role is played by the copper sulfate. Specifically, it’s the copper ions within the copper sulfate that are the true MVPs.
Think of it this way: the zinc is feeling energetic. It’s ready to give away some of its electrons. It’s feeling generous. The copper ions, on the other hand, are a bit more… eager. They’re looking to grab some electrons. They want what the zinc has to offer. So, the copper ions are the ones doing the "oxidizing." They are oxidizing the zinc. They are the oxidizing agent!
It’s a bit like a popularity contest. The zinc is the popular kid who’s happy to share its toys (electrons), and the copper ions are the ones eagerly snatching them up. This electron-swapping is what drives the whole reaction. It’s the engine of this chemical adventure!

Why is this so special? Because it shows us that even in seemingly simple mixtures, there’s a constant, invisible give-and-take. It's a world of hidden energy and transformations. The fact that a dull piece of metal can cause a beautiful blue solution to change, and red metal to appear, is just mind-blowing when you stop to think about it. It’s nature showing off its creative side.
The copper sulfate, through its copper ions, is the oxidizing agent here. It's the electron-taker. It's the reason the zinc gets transformed into zinc sulfate and the copper decides to become solid metal. It’s the catalyst for change, the director of this miniature chemical drama.

Isn't it neat how understanding just one key player, the oxidizing agent, can unlock the whole story? It’s like having a secret decoder ring for chemical reactions. You see copper sulfate and zinc, and you know, "Aha! The copper sulfate is the oxidizing agent!" And suddenly, the whole process makes sense, and it’s even more fascinating.
So, the next time you hear about the reaction CuSO₄ + Zn → ZnSO₄ + Cu, remember the star of the show. It’s not just a bunch of letters and arrows. It’s a dynamic exchange, a transformation, and at the heart of it, is the oxidizing agent, the copper sulfate, orchestrating it all with its electron-grabbing prowess. It’s a tiny, yet powerful, demonstration of chemical power. Pretty cool, right?
