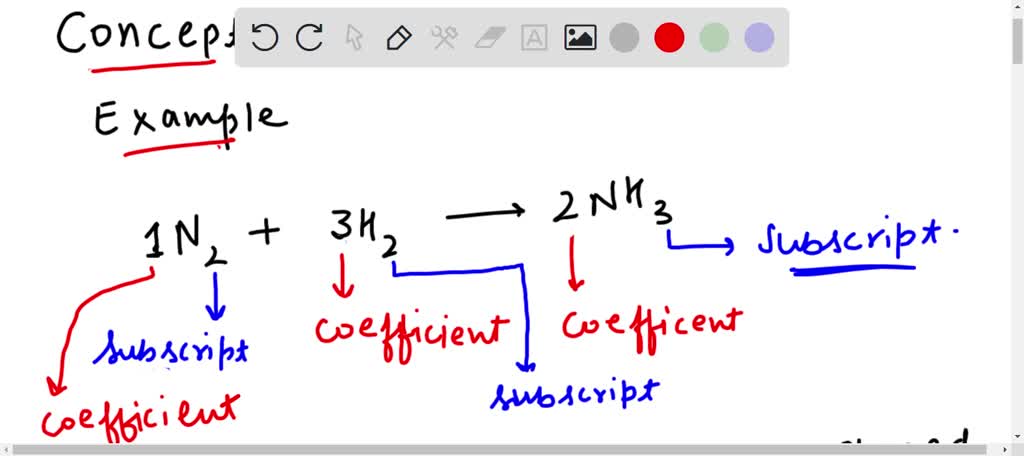
In The Reaction Above The Coefficient Of Cl2 Is
Hey there, trendsetters and life enthusiasts! Ever find yourself scrolling through your feed, seeing all these beautifully curated lives, and wondering, "How do they do it?" Well, let's talk about that feeling, but not in the way you might expect. Forget the unattainable perfection for a moment. Today, we're diving into something a little different, something that might seem purely academic at first glance, but trust me, it's got more life lessons than your average self-help guru's manifesto. We're talking about coefficients. Yeah, I know, sounds like it belongs in a dusty textbook, right? But bear with me, because understanding a simple concept like a coefficient in a chemical reaction can actually unlock a surprisingly chill perspective on how we approach our own busy lives. It’s all about balance, intention, and making sure everything is just right – sound familiar?
So, let’s get this party started with a little something that might have popped up on your screen recently. Maybe you were doing some late-night online learning, prepping for a quiz, or perhaps you just have that insatiable curiosity for, well, everything. You might have encountered a reaction. Think of it like a recipe, but for molecules. And in this recipe, there’s often a bunch of numbers hanging out in front of the chemical symbols. These are our coefficients. They’re the silent directors, the conductors of the molecular orchestra, telling us how much of each ingredient we need to make the magic happen. It’s like a peek behind the scenes of the universe’s ultimate cooking show.
Now, let’s hone in on a specific query that might have landed you here: "In the reaction above, the coefficient of Cl2 is..." This is like asking, "In this particular dish, how much of the specific type of onion did the chef use?" Cl2, for the uninitiated (and no judgment here!), is chlorine gas. It’s a pretty reactive element, kind of like that one friend who’s always the life of the party, but sometimes needs a little reigning in. And the coefficient? It’s the number that tells us how many of those Cl2 molecules are involved in the reaction.
Let's imagine a common scenario where chlorine gas is involved. Think about the creation of table salt, sodium chloride (NaCl). It’s a classic reaction. You’ve got your sodium (Na), that silvery metal that’s super eager to react, and your chlorine gas (Cl2), that greenish-yellow gas with a bit of a punch. When they get together, they form the stuff we sprinkle on our fries. Now, if you were to write out the balanced chemical equation for this, you'd typically see something like this: 2Na + Cl2 → 2NaCl.
See that "2" in front of the Na? That means we need two atoms of sodium. And that "2" in front of the NaCl? That means we get two molecules of sodium chloride. But what about the Cl2? In this particular balanced equation, there's no number explicitly written in front of Cl2. This is where a little bit of scientific etiquette comes in. In chemistry, just like in polite society, if there’s no number, it’s understood to be a "1". It’s the unspoken, yet perfectly understood, agreement. So, in the reaction 2Na + Cl2 → 2NaCl, the coefficient of Cl2 is 1.
Why is this "invisible 1" so important? Because it’s all about balance. Chemical reactions follow the Law of Conservation of Mass, meaning atoms aren't created or destroyed, just rearranged. The coefficients are the key to ensuring that the number of atoms of each element is the same on both sides of the equation. It's like a perfectly calibrated scale; everything has to add up. This principle of balance is actually a fantastic metaphor for our own lives. Are you ever feeling overwhelmed, like there’s just too much going on? It’s often a sign that your personal "coefficients" are out of whack.

Think about it. If you’re constantly saying "yes" to every invitation, every request, every new project, your "coefficient" for external demands is way too high, and your "coefficient" for rest and self-care is a lonely, neglected "1" (or even zero!). In the same way that chemists meticulously balance equations, we need to consciously balance the different aspects of our lives. It's not about doing less, it's about doing the right things in the right proportions. It’s about making sure that for every intense burst of activity, there's a corresponding period of recharge. That's the essence of a truly easy-going, yet productive, lifestyle.
Let’s delve a bit deeper into the world of these coefficients. They’re not just arbitrary numbers; they represent ratios. In our salt example, the ratio of sodium atoms to chlorine molecules reacting is 2:1. For every two sodium atoms, one chlorine molecule is consumed. This precise stoichiometry is crucial for chemists to predict yields, design experiments, and understand how matter transforms. It’s the underlying precision that makes scientific progress possible.
And this precision is echoed in the art of living well. Consider the concept of mindfulness. It’s essentially about paying attention to the present moment with intention. It’s about consciously applying a "coefficient" of awareness to your daily activities. When you're eating, you're not just shoveling food in; you're paying attention to the taste, texture, and smell. Your "eating coefficient" becomes more focused. When you're talking to a friend, you're not just waiting for your turn to speak; you're actively listening, giving your "listening coefficient" a prominent place. This intentionality, this mindful allocation of your focus, is what transforms mundane activities into richer experiences.

Now, you might be thinking, "Okay, I get the balance thing, but how does this relate to real life, beyond just mentally adjusting my to-do list?" Let’s sprinkle in some fun facts and cultural references to liven things up. Did you know that chlorine gas, while crucial in many industrial processes (like making PVC pipes, which are in so many things around your house!), was also infamously used as a chemical weapon during World War I? It’s a stark reminder that powerful forces, like even the "invisible 1" coefficient, need to be understood and managed with care. The same energy that can build can also destroy, depending on how it's directed.
Think of it like your social media feed. You have a certain "scrolling coefficient" for mindless browsing, and a "meaningful engagement coefficient" for connecting with friends or learning something new. If the mindless scrolling coefficient is set too high, you can end up feeling drained and unproductive, much like an unbalanced chemical reaction can lead to unwanted byproducts. The trick is to consciously adjust those coefficients. Perhaps setting a timer for your scrolling, or actively seeking out content that sparks joy or knowledge. It’s about being the scientist of your own digital life, meticulously balancing the inputs and outputs.
Let’s consider another angle: cooking. When you’re following a recipe, the coefficients are the measurements. Too much salt? Your dish is ruined. Too little baking soda? Your cake won’t rise. The recipe itself is a balanced equation for deliciousness. But here’s where the easy-going lifestyle comes in: sometimes, you have to trust your instincts and adjust those coefficients. Maybe you like a little more spice, or a bit less sugar. That’s you, the home chef, fine-tuning the reaction to your personal preference. It’s about understanding the fundamental principles (the balanced equation) and then applying your own creative flair, your own personal "coefficients" for taste and enjoyment.

This idea of a "1" coefficient is also incredibly freeing. It means that not every element in your life needs to be an overwhelming force. Sometimes, the most impactful contributions are the subtle ones, the quiet presences that hold things together without demanding the spotlight. Think about a good supporting actor in a film. They might not have the most lines, but their presence is essential to the narrative. Or consider the quiet hum of your refrigerator – it’s constantly working, maintaining a crucial balance for your food, but you rarely give it a second thought. Your own quiet acts of kindness, your consistent efforts towards a goal, your patient listening – these are your "1" coefficients, and they are incredibly powerful.
So, when you see that Cl2 with no number in front of it, don't just dismiss it as "nothing." Recognize it for the deliberate and significant "1" that it is. It’s a testament to balance, to the fundamental ratios that govern how things work. And in applying this to our lives, it’s a reminder that not everything needs to be a dramatic, headline-grabbing event. The most sustainable, enjoyable lifestyles are often built on a foundation of mindful balance, intentional choices, and the quiet power of the "invisible 1."
Think about your morning routine. What’s your "coffee coefficient"? Is it a single, strong shot to kickstart your day, or a more diluted, leisurely brew? What’s your "exercise coefficient"? Maybe it’s a full hour at the gym, or perhaps it’s just a brisk 15-minute walk around the block. Both can be perfectly balanced and effective, depending on your overall lifestyle equation. The key is intentionality. It’s about consciously deciding what proportion of your energy and time to allocate to each activity, just like a chemist chooses the right coefficients to ensure a successful reaction.
This is also where the beauty of adaptation comes into play. Just as a chemist might adjust coefficients to optimize a reaction for different conditions or desired outcomes, we too can adapt our "coefficients" as life throws us curveballs. Had a particularly stressful day? Maybe your "relaxation coefficient" needs to be bumped up, and your "socializing coefficient" temporarily reduced. Facing a big deadline? You might increase your "focus coefficient" for work and temporarily decrease your "leisure coefficient." It’s about fluidly recalibrating your internal recipe for well-being.
The takeaway here is that understanding these seemingly small details, like the coefficient of Cl2 being a "1," can offer a profound shift in perspective. It’s about appreciating the elegance of balance, the power of intention, and the often-underestimated impact of consistent, quiet contributions. It’s about being the architect of your own life’s equation, ensuring that all the elements are present in the right proportions for a happy, healthy, and fulfilling existence.
So, the next time you encounter a chemical equation, or even just feel overwhelmed by the sheer pace of modern life, take a moment to reflect on the coefficients. Remember that the "invisible 1" is just as important as the numbers you can see. It's about the underlying structure, the essential balance that makes everything work. And in that understanding, you might just find a little more ease, a little more control, and a whole lot more joy in your everyday adventures. Embrace the balance, folks. It’s the secret ingredient to an effortlessly chic and deeply satisfying life.
