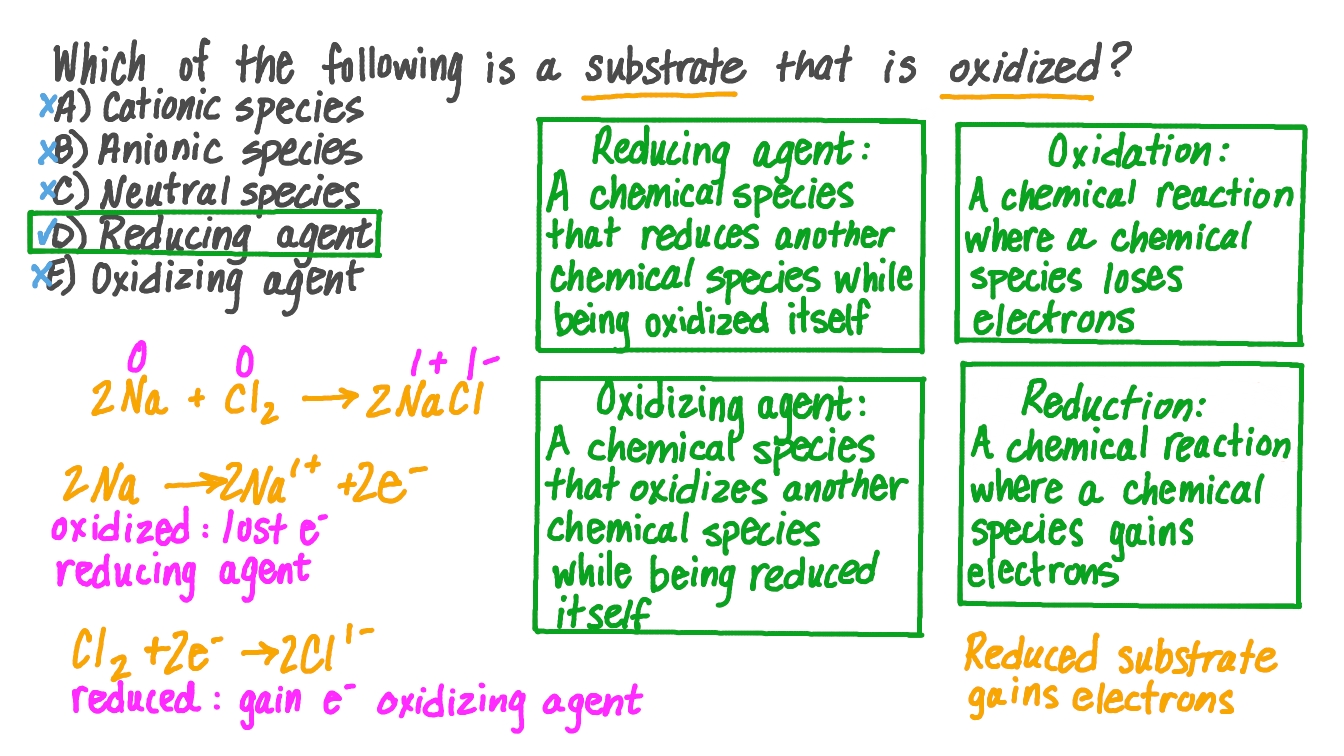
In The Following Reaction Which Species Is Oxidized

Hey everyone! Ever find yourself staring at a chemical reaction, maybe in a textbook or on a lab bench, and wonder, "What's actually going on here?" It’s like a tiny, microscopic dance, right? And sometimes, amidst all the swirling atoms and changing molecules, there’s a star of the show that sometimes gets a little overlooked. Today, we're going to zoom in on one of the most fundamental concepts in chemistry: oxidation. And we're going to tackle the question: "In the following reaction, which species is oxidized?" Don't worry, we're not going to drown you in jargon. Think of this more like a casual chat over coffee, exploring something pretty neat!
So, what is oxidation, anyway? It's a word we hear a lot, but what does it really mean in the chemical world? For a long time, it used to just mean reacting with oxygen. Like when iron rusts, it's getting oxidized by the air. Pretty straightforward, huh? But chemists, being the curious bunch they are, realized that this "oxygen" part wasn't always the whole story. They discovered that other things could also cause a similar kind of change, a change in the "oxidation state" of a substance. It's a bit like how a person can "lose" something valuable, not just to a thief (like oxygen), but maybe they misplaced it, or gave it away – the end result is they have less of it.
In modern chemistry terms, oxidation is all about the loss of electrons. Yep, those tiny, negatively charged particles that whiz around atoms. When a chemical species, be it an atom or a molecule, loses electrons, it's said to be oxidized. Think of it like this: electrons are like little pieces of currency. If you’re holding onto your money, you're stable. But if you start giving your money away, you're essentially being "oxidized" in a financial sense – you have less of that valuable currency.
Now, the flip side of this coin is equally important, and it’s called reduction. Oxidation and reduction always happen together. They are like two sides of the same coin, or like a handshake. You can't have one without the other. While one species is losing electrons (getting oxidized), another species has to be gaining those electrons (getting reduced). It's a constant give and take. So, if oxidation is losing electrons, reduction is the gain of electrons. Back to our money analogy, if you're giving money away, someone else is receiving it, right? That person is getting "reduced" in terms of their available cash, in a way, because they've just acquired some!
So, when we ask "Which species is oxidized in the following reaction?", we're essentially asking: Which species lost electrons? It's like being a detective, looking for clues about who gave away the goods.
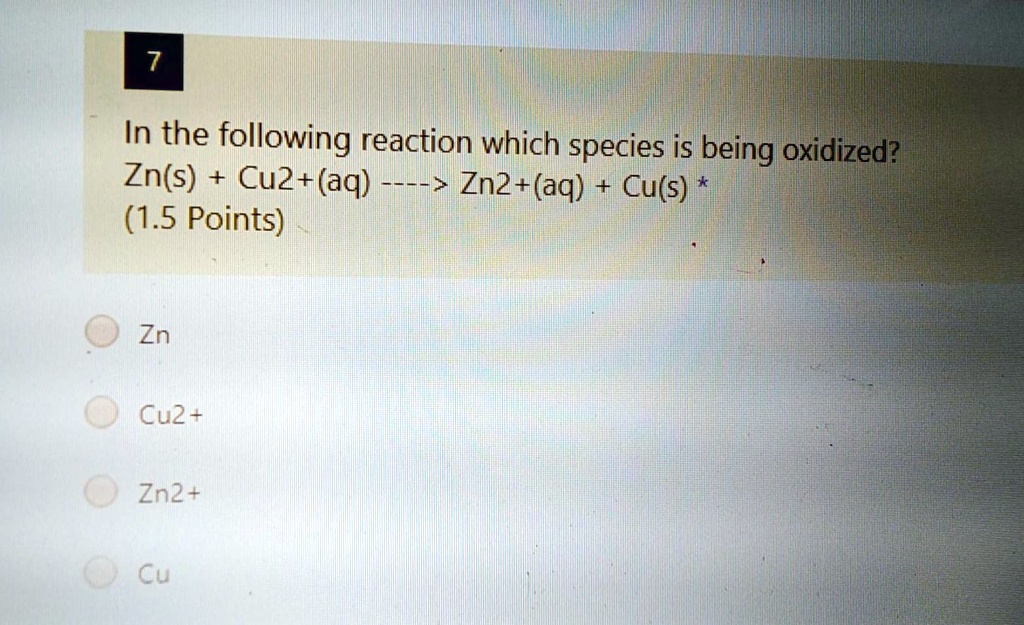
How do we spot the culprit?
This is where things get a bit more interesting, and a little bit like solving a puzzle. We often look at the oxidation numbers (or oxidation states) of the atoms involved. These numbers are like a scorecard, assigning a hypothetical charge to an atom in a molecule or ion, based on a set of rules. They tell us how "electron-rich" or "electron-poor" an atom is considered to be.
Here's a simplified way to think about it:
- An element in its pure form (like a chunk of iron, or a molecule of oxygen gas O2) has an oxidation number of zero. It's just chilling, unattached.
- In compounds, elements usually get assigned numbers based on electronegativity (how much they "want" electrons). Oxygen is often given a -2 charge (except in peroxides). Hydrogen is usually +1 when bonded to nonmetals and -1 when bonded to metals.
- The sum of the oxidation numbers in a neutral compound must equal zero.
- The sum of the oxidation numbers in a polyatomic ion must equal the charge of the ion.
Now, for the big reveal! To find out which species is oxidized, we compare the oxidation numbers of an element before and after the reaction. If the oxidation number of an element increases, it means it has lost electrons, and therefore, it has been oxidized. If the oxidation number decreases, it means it has gained electrons, and it has been reduced.
Let’s look at an example (without getting too complicated!)
Imagine a simple reaction like the formation of rust: iron reacting with oxygen.
Fe + O2 → Fe2O3

Let's break down the oxidation numbers:
- On the left side (before the reaction):
- Fe is in its elemental form, so its oxidation number is 0.
- O in O2 is also in its elemental form, so its oxidation number is 0.
- On the right side (after the reaction):
- In Fe2O3 (iron(III) oxide), oxygen usually has an oxidation number of -2.
- To make the whole compound neutral (sum of oxidation numbers = 0), we have 2 iron atoms. So, 2 * (oxidation number of Fe) + 3 * (-2) = 0. This means 2 * (oxidation number of Fe) = +6, and the oxidation number of Fe is +3.
Now, let's compare:
- Iron (Fe): Went from 0 to +3. Its oxidation number increased. This means iron lost electrons. So, Fe is oxidized.
- Oxygen (O): Went from 0 to -2. Its oxidation number decreased. This means oxygen gained electrons. So, O2 is reduced.
See? It’s like watching a trade happen. Iron gave away some of its "electron currency" to oxygen. It's a fundamental process that powers everything from how our bodies digest food to how batteries work.
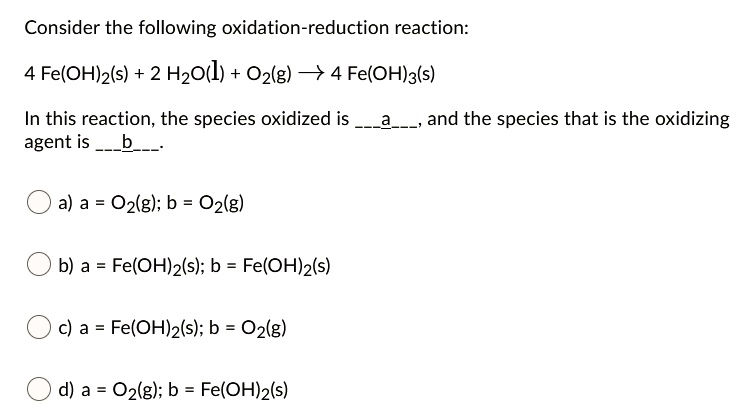
Sometimes, the reactions can look more complex, with whole molecules participating. But the core principle remains the same: find the species whose atoms have a higher oxidation number after the reaction than before. That's your oxidized species! It’s the one that’s undergone a loss of electrons.
It’s pretty cool when you think about it, isn't it? This constant dance of electrons, driving chemical changes all around us. So next time you see a reaction, don't just see a jumble of symbols. Try to spot the electron losers and the electron gainers. It’s a whole miniature drama unfolding!
Keep that curiosity buzzing, and happy observing!
