In Fermentation _____ Is Reduced And _____ Is Oxidized.

Hey there! Grab a mug, pull up a chair. Let's chat about something kinda cool, maybe a little nerdy, but totally worth knowing. Ever wonder what's really going on when your sourdough starter bubbles, or when that kombucha gets fizzy? It's all about… fermentation! And in the wild, wonderful world of fermentation, there's a dynamic duo at play. We're talking about reduction and oxidation. Sounds fancy, right? But trust me, it’s not as intimidating as it sounds. Think of it like a dance, a chemical tango happening right under our noses, or more often, in a jar on the counter. So, what’s the big deal? Well, it boils down to this: in fermentation, something is getting reduced, and something else is getting oxidized. Mind-blowing stuff, I know!
So, let's break it down, like we're dissecting a particularly complex cookie recipe. Imagine you've got these molecules, right? They're just hanging out, minding their own business. Fermentation is like a party for these molecules, where they swap things around. And the star of this molecular swap meet? Electrons! It's all about those tiny, zippy things called electrons. Think of them like little energy packets. Who gets them, and who loses them? That's where our reduction and oxidation buddies come in.
Let's start with the one that's getting reduced. What does that even mean? It’s like getting a present, a really good present. In chemistry terms, when something is reduced, it's basically gaining electrons. Yep, it’s scooping up those little energy packets. It's like the molecule that was feeling a little… depleted, suddenly gets a boost. It’s gaining all this electron goodness. Think of it as going from a plain bagel to a cream cheese-slathered, everything-bagel masterpiece. That's a reduction!
And who’s usually the lucky recipient of this electron bounty in fermentation? Often, it’s something called an electron acceptor. This molecule is practically begging for those electrons. It's like that friend who’s always asking to borrow your charger – it needs that juice! In many fermentation processes, this electron acceptor is a molecule that’s going to become something else, something different, as it takes on those electrons. It's a fundamental transformation, and it's happening because it's getting something. It's gaining those precious electrons, becoming reduced.
Now, on the flip side, we have the molecule that's getting oxidized. This is the opposite of getting a gift. This is like… well, it’s losing something. It’s donating those precious electrons. Think of it as the generous friend, the one who always picks up the tab. They’re giving something away. It's a loss of electrons. And when a molecule loses electrons, we say it's been oxidized. It's like going from that glorious cream cheese bagel to just the plain bagel again. A bit less exciting, perhaps, but crucial for the whole process, wouldn't you agree?

So, who's playing the role of the electron donor, the one getting oxidized? It’s usually the fuel for the fermentation party! Think about the sugars you add to your starter or the grains in your beer. Those are the guys. They're the starting point, the energy source. They're the ones that will break down, and in the process, they give up their electrons. They’re the sacrificial lambs, in a way, but in a really productive, delicious way. They’re oxidized so that other things can be reduced. It’s a perfect balance, isn't it?
Think about that sourdough starter, that bubbly magic in a jar. What’s happening there? Yeast and bacteria are feasting on those lovely sugars in the flour. They’re breaking them down. And as they break them down, they’re oxidizing those sugar molecules. They're releasing energy, and those electrons are being passed around. It’s a whole cascade of events. The sugars are getting oxidized, losing their electrons, and other molecules are gratefully accepting them, getting reduced. This whole electron-swapping business is what powers the whole fermentation gig.
Let’s get a little more specific, shall we? In many common fermentations, like the ones that make your bread rise or your beer bubbly, you're dealing with glucose, right? That’s a sugar. And glucose is going to be the thing that gets oxidized. It’s going to lose its electrons. It's going to get broken down into simpler molecules. Think of it as the original energy bar being unzipped and its contents being shared. And what are those electrons going to? Well, that depends on the type of fermentation!

In alcoholic fermentation, which is what happens when yeast makes booze (hooray!), the oxidized glucose eventually helps to create ethanol and carbon dioxide. The electrons that were originally part of the glucose are ultimately accepted by a molecule that eventually forms part of the CO2. So, the glucose is oxidized, and indirectly, something related to the carbon in the glucose is reduced to form the carbon dioxide. It's a bit of a roundabout way of thinking about it, but the core idea is the electron transfer.
In lactic acid fermentation, which you see in yogurt, sauerkraut, and even in your muscles when you’re really working out (ouch!), it’s a similar story. Glucose gets oxidized. And those electrons end up being accepted by a molecule that forms lactic acid. So, again, the sugar is the thing giving up electrons (getting oxidized), and a form of carbon in that sugar is gaining electrons (getting reduced) to become lactic acid. It’s all about that electron trade!
So, to recap, in fermentation, the energy-rich molecule, often a sugar, is the one that gets oxidized. It's like the fuel that's burning, but in a controlled, biological way. It loses electrons. And those electrons have to go somewhere, right? They’re picked up by another molecule, which then gets reduced. This molecule that accepts the electrons is often called the terminal electron acceptor. It’s the final destination for those electron packets, and its transformation is a key part of what fermentation is. It’s the recipient of the electron bounty.

Think of it like a relay race. The sugar is the runner with the baton (the electrons), and they pass it off to the next runner (the electron acceptor). The first runner is giving up the baton, so they're in a way, "oxidized" from having it. The second runner is receiving the baton, so they're "reduced" in their state of not having it. It’s a bit of a simplification, but it gets the idea across! That electron transfer is the engine driving the whole operation.
And the beauty of it is, this happens in so many different ways! From the humble bacteria turning milk into tangy yogurt to the yeasts creating the complex flavors in wine, the fundamental principle remains: something is reduced, and something is oxidized. It's the fundamental dance of life at a molecular level, making our food tastier, our drinks more interesting, and our bodies function. It’s not just chemistry; it’s culinary magic powered by electron exchange!
So, next time you enjoy a crusty loaf of bread, a sharp pickle, or a frothy pint, you can wink at it and say, "I know what you did there! You were busy with some serious reduction and oxidation, weren't you?" It’s pretty cool to think about the intricate processes happening to create all the delicious things we take for granted. It’s all about these tiny, invisible electron transfers, making the world a better, tastier place. Who knew something so small could have such a huge impact? It’s a testament to the power of chemistry, and a good reminder that sometimes, the most amazing transformations come from giving and receiving.
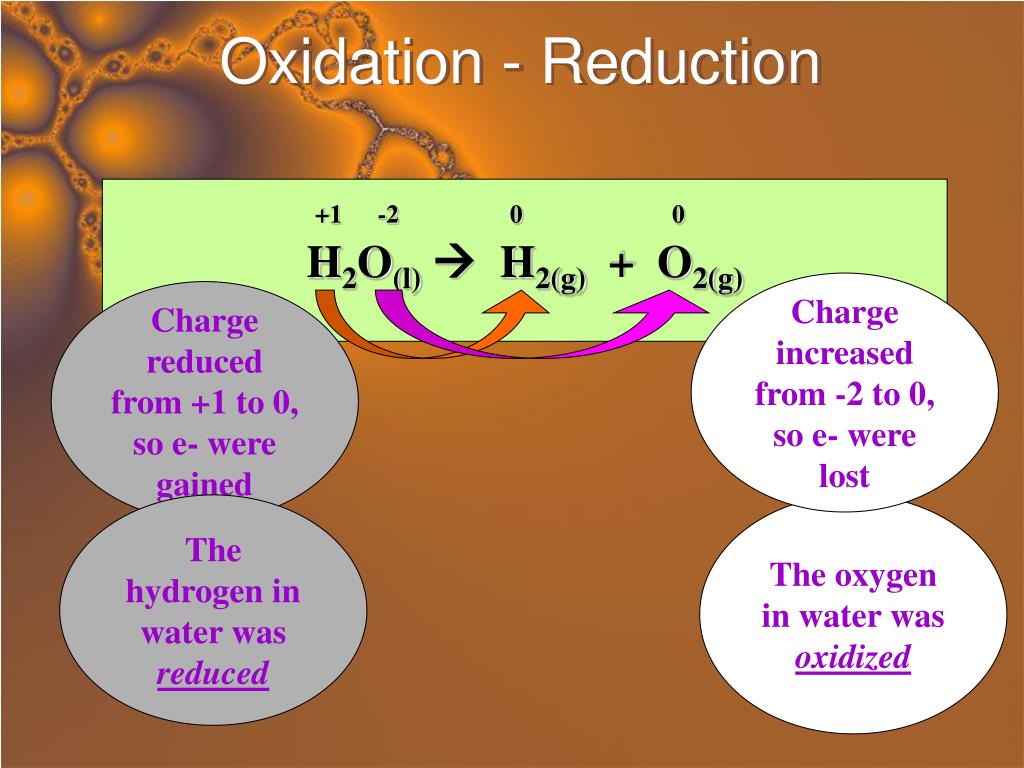
It’s really about energy, at its core. The molecules that get oxidized release energy as they lose those electrons. And that released energy is what powers the whole fermentation process, allowing the organisms doing the fermenting to live and thrive. And in turn, they break down compounds for us, creating all sorts of delicious and beneficial byproducts. It’s a win-win situation, really. The microbes get fed, and we get fermented goodies. All thanks to the give-and-take of electrons.
So, let’s just say it one more time for good measure, because it’s that important and that central to the whole fermentation fiesta. In Fermentation, a molecule (typically a sugar or other energy source) is reduced, and another molecule (an electron acceptor) is oxidized. Wait, did I get that right? Let me check my notes… squints at imaginary notepad… Oh, hang on. I might have flipped it. Let me rephrase that, because even over coffee, I want to get this right for you! It’s actually the other way around. So, take two, and try to keep up! In Fermentation, a molecule (typically a sugar or other energy source) is oxidized, meaning it loses electrons. And another molecule (an electron acceptor) is reduced, meaning it gains electrons. Phew! See? It’s easy to get mixed up, but the core concept is that electron transfer. The sugar is the one breaking down and giving up electrons, and something else is happily taking them. That's the heart of it all!
Think of it like this: the sugar is the donor, and the electron acceptor is the receiver. The donor gives away electrons, becoming oxidized. The receiver takes those electrons, becoming reduced. It’s the most fundamental principle at play, powering everything from your gut health to your favorite craft beer. It’s a constant exchange, a chemical conversation that leads to delicious results. So, embrace the redox reactions, my friends! They’re the unsung heroes of the fermented food world!
