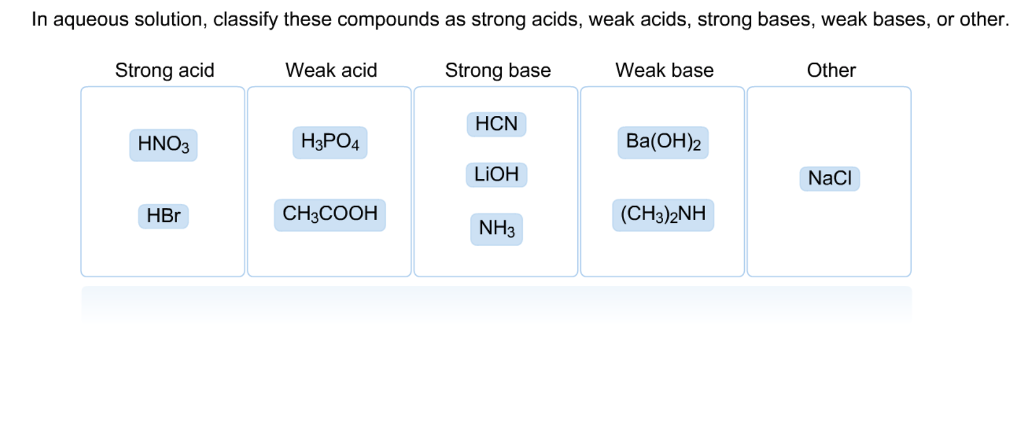
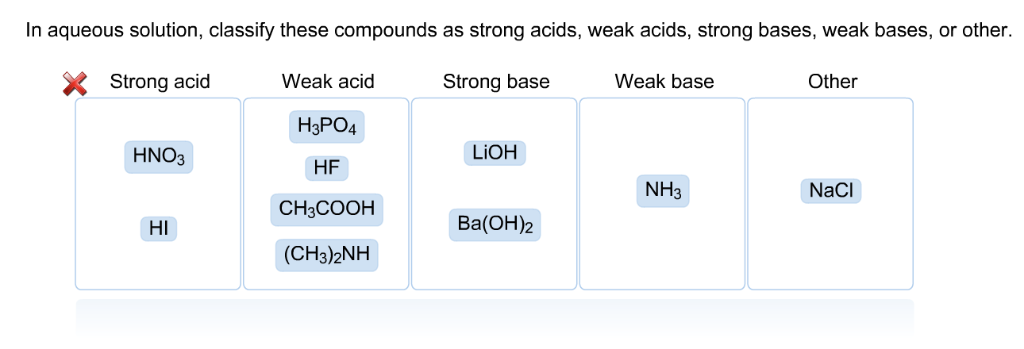
In Aqueous Solution Classify These Compounds As Strong
Hey there, curious minds! Ever looked at a bottle of something and wondered, "What exactly is going on in there when it hits the water?" Well, buckle up, because we're about to dive headfirst into the wonderfully weird world of aqueous solutions and how some compounds decide to be total rockstars in water!
We're talking about classifying compounds as strong in aqueous solution. Sounds a bit technical, right? But trust me, it's less about intimidating chemistry jargon and more about understanding how certain substances absolutely transform when they meet H2O. Think of it as a splashy party where some guests just explode with energy!
The Great Dissolving Adventure!
So, what's an "aqueous solution" anyway? Fancy words for stuff dissolved in water. And water, our amazing universal solvent (well, almost universal!), has this incredible ability to break things apart and let them mingle. It’s like a giant, bubbly dance floor!
Now, not all compounds play by the same rules when they hit this dance floor. Some are a bit shy and prefer to hang out in their original form. Others? They go wild! They break into their tiniest, most energetic components and zoom around, making everything around them, well, more… energetic!
Enter the "Strong" Ones: The Life of the Party!
This is where our "strong" compounds come in. When we say a compound is strong in an aqueous solution, we're essentially saying it's a super-enthusiastic participant in the water party. It completely breaks apart, or dissociates, into its constituent ions.
Imagine you have a bag of perfectly formed LEGO bricks. Some compounds, when you drop them in water, stay as those whole LEGO bricks. But our strong compounds? They’re like those LEGOs that spontaneously unclick into their individual studs and are now free to roam and interact with everything else. It’s a complete transformation!
Why Does This Matter? It's All About Conductivity!
You might be thinking, "Okay, so they break apart. Big deal." Ah, but this is where the magic truly happens, and it has some super practical (and fun!) implications. When a compound breaks into ions, these little charged particles are fantastic at conducting electricity. Think of it like adding tiny, mobile conductors to your water!
This is why solutions containing strong compounds are excellent electrical conductors. Batteries, for example, rely on these principles. That jolt of power? Often thanks to ions happily zipping around in solution. How cool is that? Your everyday gadgets are powered by chemistry happening in watery realms!
The Usual Suspects: Who Are These "Strong" Celebrities?
So, who are these life-of-the-party compounds? In the grand scheme of chemistry, the most common types of strong compounds you’ll encounter are strong acids and strong bases. They’re the undisputed champions of dissociation!

Strong Acids: The Zesty Zingers!
Acids, in general, are known for their sour taste (though please, never taste unknown chemicals!) and their ability to donate a proton (a positively charged hydrogen ion, H+). But strong acids are the ones that do this with gusto. They don't hold back. When you put them in water, they release all their available protons.
Think of hydrochloric acid (HCl). In water, it's like, "Here you go, water! Have all my H+ ions!" HCl completely splits into H+ and Cl- ions. No hesitation, no half-measures. It's 100% dissociation. Other famous strong acids include sulfuric acid (H2SO4) and nitric acid (HNO3). These are the acids that mean business when it comes to donating those protons.
Strong Bases: The Slippery Smooth Operators!
On the flip side, we have strong bases. Bases are generally known for their bitter taste (again, don't taste them!) and their ability to accept protons or produce hydroxide ions (OH-). Strong bases, however, are incredibly efficient at releasing those hydroxide ions. They're like fountains of OH-!
When you dissolve sodium hydroxide (NaOH) in water, it’s another complete transformation. It’s not just a little bit of dissociation; it's a full-blown ion party! NaOH breaks apart entirely into Na+ and OH- ions. They’re ready to mingle and neutralize acids with impressive speed. Other popular strong bases include potassium hydroxide (KOH) and calcium hydroxide (Ca(OH)2).

What About the "Weak" Ones?
Now, it's only fair to mention their less exuberant cousins: the weak compounds. These guys are more reserved. When you put a weak acid or a weak base in water, only a small fraction of them will dissociate. Most of them will stay intact, like shy guests politely observing the dance from the sidelines. This is why weak acids and bases are not as good at conducting electricity – there are fewer free-roaming ions!
Understanding the difference between strong and weak is key to appreciating the nuances of chemistry. It’s like knowing the difference between a full-on rave and a quiet, intimate gathering. Both have their purpose and their unique energy!
Making Life More Fun with Strong Solutions!
So, how does knowing about strong acids and bases make your life more fun? Well, for starters, it’s about understanding the world around you! That fizzy drink you enjoy? The cleaning products you use? Even the biological processes happening inside your body? Chemistry is at play!

Think about how strong acids are used in industrial processes, like pickling metals or in batteries. Or how strong bases are used to make soap or in water treatment. These aren't just abstract concepts; they are the building blocks of so many things we rely on and use every day. It’s like having a secret code to understanding how the world works!
And for those of you who enjoy a good DIY project or a science experiment at home (with proper supervision, of course!), understanding these concepts opens up a whole new level of understanding and creativity. You can start to predict what might happen when you mix certain things, or why one solution behaves differently from another. It’s like unlocking superpowers for your brain!
The Journey of Discovery Awaits!
This is just the tip of the iceberg, my friends! The world of aqueous solutions and compound classification is vast, fascinating, and surprisingly accessible. Every time you encounter a chemical reaction or a substance in solution, remember the strong ones – the compounds that go all-in, fully dissociating and electrifying their surroundings.
Don't be intimidated by the terms. Embrace the curiosity! The more you learn, the more you'll see the elegant simplicity and the incredible complexity of the universe at a molecular level. So, go forth, ask questions, and let your own journey of discovery in the wonderful world of chemistry begin. You might be surprised at how much fun you have learning about how things really get down in water!
