In An Experiment A Scientist Makes A Radioactively Labeled
Okay, so picture this: I'm rummaging through my grandma's attic the other day, you know, the usual dust-bunny-filled adventure. I pull out this old, faded hatbox, expecting maybe some vintage scarves or a moth-eaten ball gown. Instead, I find… a ridiculously tiny, bright red rubber duck. Seriously, this thing was smaller than my thumb. It had this smug little painted-on smile, and I swear, it looked like it was judging my life choices. Like it knew I hadn't dusted the attic in a decade. It got me thinking, though. Even the smallest, silliest things can have a hidden purpose, right? Like this little duck, probably a novelty keyring or something from a forgotten vacation. It's not just there; it's a tiny artifact of someone's past. And that, my friends, is kinda how we're gonna dive into something a bit more… intense today.
We're talking about scientists and their toys. Specifically, when a scientist makes something radioactively labeled. Now, before you picture Indiana Jones running from rolling boulders of uranium, let's take a breath. It's not quite that dramatic, though it does involve things that can be, well, a little bit glowy. And it’s all about understanding the unseen. You know, like trying to figure out where your keys actually go when you swear you left them on the counter. Science has its own version of that, but with a lot more precision and a lot less frantic searching.
So, what does it mean to "radioactively label" something? Think of it like attaching a tiny, invisible GPS tracker to a molecule. Except this tracker doesn't just tell you where it is; it also tells you what it’s doing and how fast it's getting there. It's a way to make something incredibly specific and normally undetectable, utterly traceable.
Imagine you're trying to follow a single drop of ink in a vast ocean. Impossible, right? But what if that ink drop was flashing a tiny, silent, and unique signal? Suddenly, you can track it. That's the magic of radioisotopes.
In the lab, this involves taking a normal molecule – could be anything from a sugar molecule to a piece of DNA – and swapping out one of its atoms for a special, unstable version of that same atom. This unstable version is called a radioisotope. And these radioisotopes are, shall we say, a bit energetic. They tend to decay, and when they do, they release tiny particles or energy, which we can detect. It's like giving our molecule a secret handshake that only a special detector can pick up.
Why would anyone want to do this? Well, it opens up a whole universe of possibilities for understanding how things work. Biology, medicine, environmental science – they all benefit immensely from this technique. It’s like getting a backstage pass to the most intimate processes of life and nature.

Let's take a super common example: tracing how drugs work in the body. You develop a new pill, and you want to know: does it go where it's supposed to? Does it get broken down? How quickly? Instead of just seeing the results of the drug (like, "patient feels better"), you can actually see the drug itself moving through the system. You radioactively label the drug molecule, give it to a patient (in a controlled, safe way, of course!), and then use special scanners to track where the label goes. It’s like a treasure hunt, but the treasure is understanding how your body processes medicine.
This is crucial for understanding dosage, side effects, and how effective a drug is going to be for different people. It’s not just guesswork anymore; it's about precise, observable pathways. Imagine the difference between saying, "This medicine might work" versus "This medicine goes directly to the affected cells and has a predictable breakdown rate." Huge difference, right?
The Perks of Being Radioactive (for a Molecule)
So, what makes a radioisotope so darn useful? It's all about its "signature." Different radioisotopes have different decay rates (how long they take to break down) and emit different types of radiation. This allows scientists to choose the right label for the job. Some labels might be perfect for short-term tracking, while others are ideal for longer studies.
For instance, you might use a short-lived radioisotope to track a substance through a quick metabolic process. Or, for studying something that lingers in the environment for years, you’d pick a different, more persistent radioisotope. It's like picking the right tool for the job – you wouldn't use a sledgehammer to hang a picture, and you wouldn't use a tiny pin to move a mountain. It’s all about matching the label to the question you’re trying to answer.
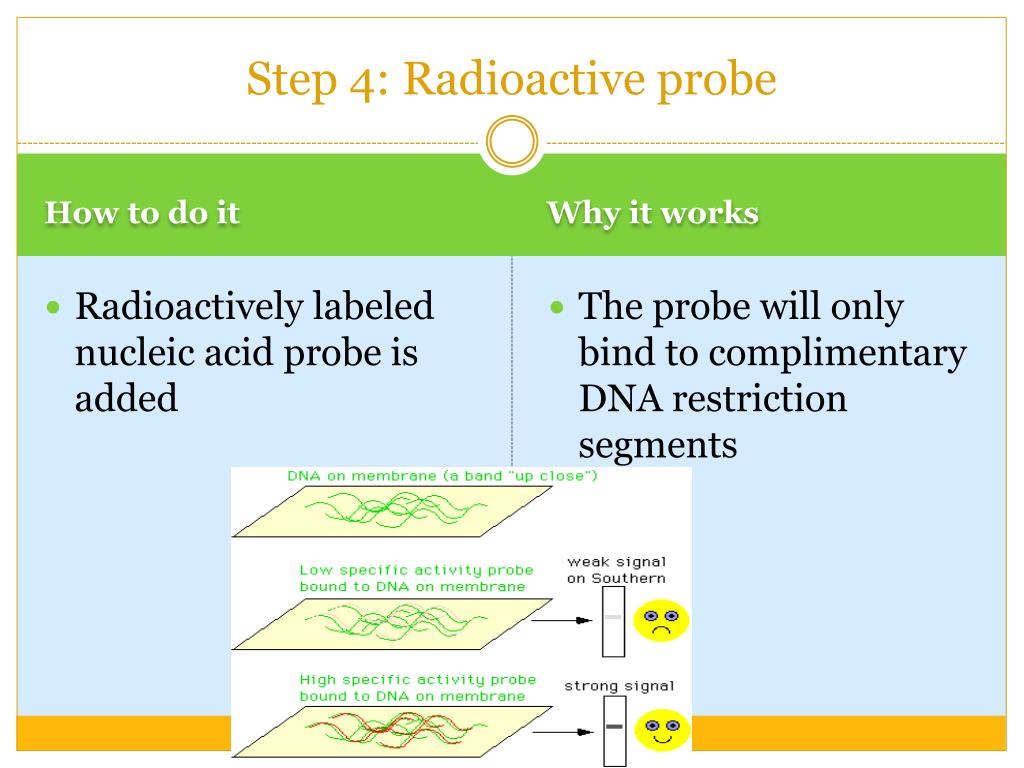
The detection methods are pretty fancy too. We’re talking about things like scintillation counters that "light up" when the radiation hits them, or imaging techniques that can create maps of where the labeled substance has gone. It’s not about seeing the molecule itself, but seeing the effect of its radioactivity. A ghost image, if you will, of our tagged molecule.
And the amount of substance you need? It can be incredibly small. This is a big deal, especially when you're studying delicate biological systems or trying to avoid overwhelming a natural process with too much of something. You can track femtograms – that’s a millionth of a billionth of a gram – of a substance. Pretty mind-blowing when you think about it. It's like finding a needle in a haystack, but the needle is constantly beeping.
Beyond the Lab: Real-World Applications
This isn't just some theoretical concept confined to sterile labs. Radioactively labeled compounds are everywhere. In medicine, they're used in diagnostic imaging like PET scans. You might have heard of them. A patient is given a small amount of a radioactively labeled glucose molecule. Cancer cells, being metabolically very active, gobble up this labeled glucose faster than normal cells. The scanner then detects where the radiation is concentrated, revealing the location and extent of the tumor. Pretty cool, huh? It's like giving doctors X-ray vision for disease.
Think about environmental science. How do pollutants move through the soil? How do nutrients get transported in water systems? Scientists can radioactively label specific chemicals or nutrients and then track their journey. This helps them understand how contamination spreads, how ecosystems function, and how to mitigate environmental damage. It's a way to understand the unseen flows of our planet.

Even in agriculture, this technique plays a role. Researchers can use radioactively labeled fertilizers to see how efficiently plants absorb nutrients. This helps in developing more effective fertilizers and reducing waste, which is good for both farmers and the environment. It's about making sure that precious resources aren't going to waste, just floating around doing nothing useful.
A Word on Safety (Because, You Know, Radiation)
Now, I know what some of you might be thinking. "Radiation? Isn't that dangerous?" And you'd be absolutely right to ask! This is where the "controlled" part of "controlled experiments" really comes into play. Scientists who work with radioisotopes are highly trained, and the materials are handled under very strict safety protocols.
The key is that they use very small amounts of radioactive material, and often, the radioisotopes they use have very short half-lives. A half-life is the time it takes for half of the radioactive atoms in a sample to decay. Some isotopes decay in minutes or hours, meaning their radioactivity disappears pretty quickly, leaving behind stable, harmless atoms. It's like a party that's over before it really gets out of hand.
So, while the concept of radioactivity might sound scary, in practice, it's a carefully managed tool used to gain incredible insights. It’s about harnessing a natural phenomenon for understanding, not for destruction or widespread harm. It’s like using fire to cook your dinner – it can be dangerous if misused, but controlled, it's incredibly useful.

The process of creating these labeled compounds itself is also a science. It requires specialized equipment and expertise. They can't just sprinkle some radioactive dust on a molecule. It involves sophisticated chemical reactions to ensure the radioisotope is incorporated precisely where it's needed in the molecule. It’s a bit like molecular surgery, really.
And the choice of radioisotope is critical. For medical applications, for instance, they often choose isotopes that emit gamma rays, which can penetrate tissues and be detected by external scanners, and which have relatively short half-lives so the radiation doesn't stay in the body for too long. For tracing reactions in a test tube, they might use different types of radiation that are easier to detect in a lab setting.
So, the next time you hear about a scientific experiment involving a "radioactively labeled" substance, don't picture a glowing, pulsating blob of doom. Picture a tiny, invisible tracker, a molecular detective, meticulously revealing the secrets of the universe, one decay at a time. It’s a testament to human ingenuity, our insatiable curiosity, and our ability to turn even potentially hazardous phenomena into powerful tools for discovery. Pretty neat, when you think about it. It’s like finding a hidden message in a bottle, but the message tells you how a cell divides or how a river flows.
It all goes back to that tiny rubber duck, doesn’t it? Seemingly insignificant, but a carrier of a story, a hint of purpose. And these radioactively labeled molecules, whether they’re helping us understand cancer, pollution, or how our bodies function, are the ultimate storytellers. They’re the tiny, glowing messengers of the scientific world, and we’re lucky to have them.
