In Alcohol Fermentation Nad+ Is Regenerated From Nadh By

Ever wondered how your favorite sourdough bread gets its airy texture, or how that perfectly fizzy kombucha ends up tasting so delicious? It's all thanks to a fascinating biological process called alcohol fermentation, and at its heart lies a clever trick involving molecules that are a bit like tiny biological batteries. Today, we’re going to dive into the superhero role of a molecule named NAD+ and how it gets a crucial recharge from its tired counterpart, NADH, during this magical process.
Think of it this way: life, at its most basic, is about energy. Our cells are constantly working, whether it's to build new tissues, repair damage, or just keep us thinking. This energy isn't just plucked from thin air; it's carefully managed and transferred through a series of chemical reactions. In the world of metabolism, two key players in this energy transfer game are NAD+ and NADH. They're like a rechargeable battery pack for your cellular machinery.
NAD+, in its "charged" state, is ready to accept energy in the form of electrons and a hydrogen atom. When it does this, it becomes NADH, the "discharged" or "used" form. Now, this might sound a bit counterintuitive to call it "tired," but in the context of metabolic pathways, NADH has done its job of carrying energy. The problem is, if all our NAD+ gets converted to NADH and stays that way, our cellular energy factories would grind to a halt. We'd run out of charged batteries!
This is precisely where alcohol fermentation steps in, acting as a sort of pit stop to get those batteries recharged. It’s a pathway that organisms like yeast and some bacteria use to generate energy when oxygen isn't available. Without oxygen, they can't run the full, highly efficient energy production line we humans typically rely on (called cellular respiration). So, they need an alternative.
The Purpose: Keeping the Energy Flowing
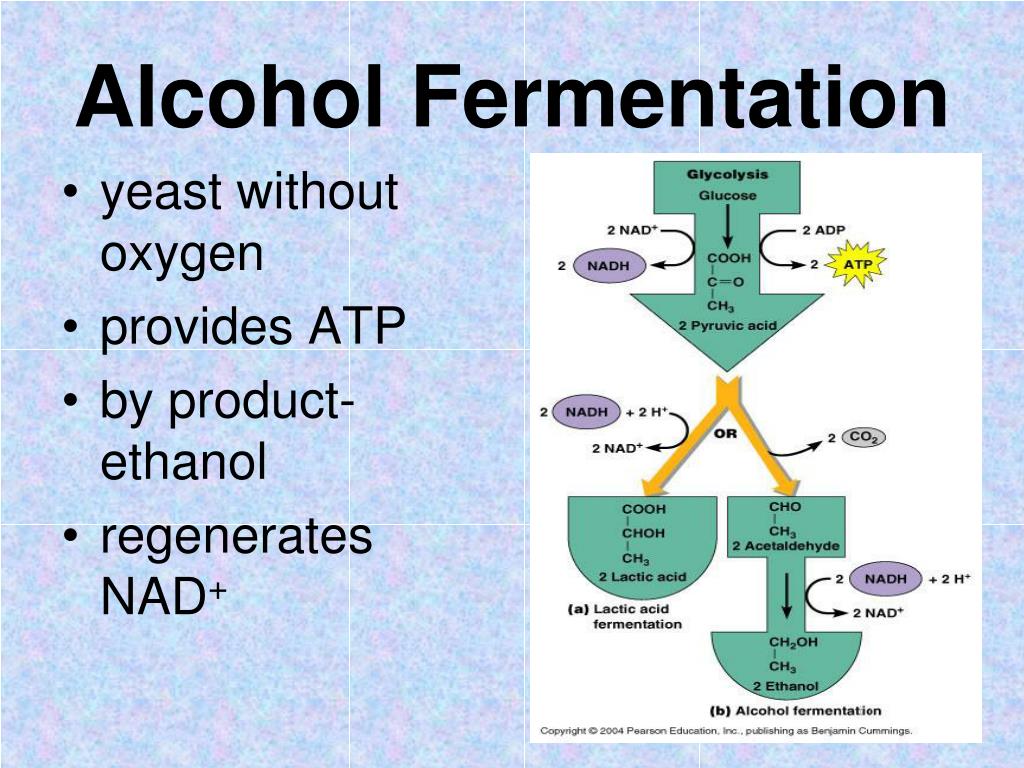
The main purpose of alcohol fermentation is to regenerate NAD+ from NADH. Why is this so important? Well, during an earlier stage of breaking down sugars (called glycolysis), cells produce a small amount of energy and also generate a lot of NADH. Glycolysis itself requires a constant supply of NAD+ to keep going. If the NADH isn't dealt with, glycolysis stops, and so does energy production. Alcohol fermentation is the ingenious solution to this bottleneck.
The benefits are enormous, both for the microorganisms themselves and for us! For yeast, it allows them to survive and thrive in oxygen-poor environments, which are common in many places they live. For us, it's the magic behind some of our most beloved foods and drinks:
- Bread: When yeast ferments sugars in dough, it produces carbon dioxide gas. This gas gets trapped in the dough, creating the airy, spongy texture we love in bread. The alcohol produced usually evaporates during baking.
- Beer and Wine: Yeast happily munches on sugars in grains or grapes, fermenting them into alcohol (ethanol) and carbon dioxide. This is the fundamental process that gives us these classic beverages.
- Kombucha: A SCOBY (Symbiotic Culture Of Bacteria and Yeast) works its magic through fermentation, breaking down sugars and producing organic acids, a touch of alcohol, and a delightful fizz.
- Other Fermented Foods: Many other fermented foods, from soy sauce to some cheeses, rely on various microbial processes that often involve regenerating cofactors like NAD+.
The Regeneration Trick: From Tired to Charged
So, how exactly does alcohol fermentation achieve this vital regeneration? It's a two-step process that happens after glycolysis:

- Decarboxylation of Pyruvate: In the first step, a molecule called pyruvate, which is the end product of glycolysis, loses a carbon atom in the form of carbon dioxide (CO2). This leaves behind a two-carbon molecule called acetaldehyde. This step is catalyzed by an enzyme called pyruvate decarboxylase. This is the step that releases the CO2 that makes bread rise and beer bubbly!
- Reduction of Acetaldehyde: This is the critical step for our battery recharge. The acetaldehyde that was just created is then converted into ethanol (the alcohol we drink). During this conversion, the NADH molecule donates its electrons and hydrogen atom to the acetaldehyde. This process effectively "takes back" the energy from NADH and uses it to build the ethanol molecule. Crucially, by giving up its electrons and hydrogen, NADH is oxidized back into its charged form: NAD+! The enzyme responsible here is alcohol dehydrogenase.
Essentially, the organism sacrifices a bit of its sugar to produce ethanol and CO2, but in doing so, it successfully converts a whole lot of spent NADH back into ready-to-use NAD+. This allows glycolysis to continue, producing more ATP (the cell's main energy currency) and keeping the whole metabolic engine running.
It’s a beautiful example of how life finds elegant solutions to its energy challenges. The next time you enjoy a slice of crusty bread or a refreshing fermented beverage, take a moment to appreciate the silent, microscopic world of alcohol fermentation and the unsung heroes, NAD+ and NADH, tirelessly working to keep those processes alive and kicking!
