In A Photoelectric Effect Experiment The Stopping Potential Is
Hey there, science curious folks! Ever stumbled upon something in physics that sounds super fancy, like, say, "stopping potential"? It pops up when we talk about this neat trick called the photoelectric effect. Sounds like something out of a sci-fi movie, right? But trust me, it's way cooler and more down-to-earth than you might think. So, what exactly is this "stopping potential" all about, and why should we even care? Let's dive in, no heavy textbooks required!
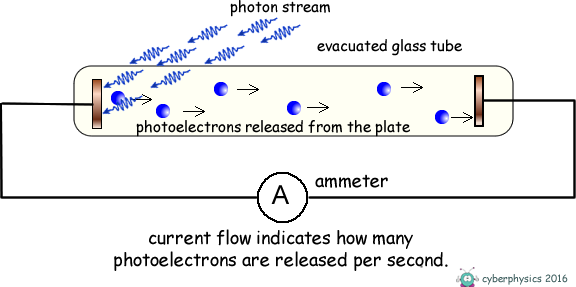
Imagine you're at a party, and someone's got a really cool light show going on. When the light hits certain shiny surfaces, poof! Electrons jump off. That's the basic idea of the photoelectric effect. Light, which we usually think of as just, well, light, can actually kick out tiny little particles called electrons from a material. Pretty wild, huh?
Now, the energy of the light matters. Think of it like throwing a ball. If you gently toss it, it might not go very far. But if you really chuck it, it flies! Different colors of light have different amounts of energy. Bluer light generally packs more of a punch than redder light. So, when the right kind of light hits a metal, it can transfer enough energy to an electron to break it free.
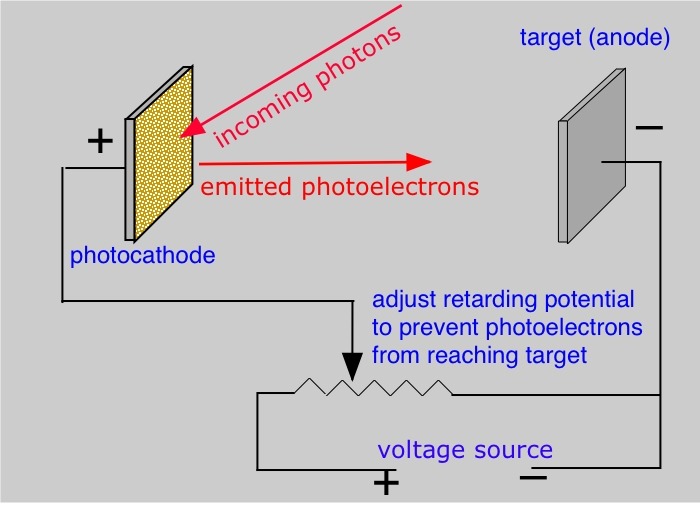
This is where our star, the stopping potential, enters the scene. So, we've got these electrons flying off a metal because light hit it. What if we wanted to stop them? Like, really put the brakes on these speedy little guys? That's exactly what stopping potential does!
Here's the cool part: we can set up an experiment where we use electricity to try and push these electrons back towards the metal they came from. We apply a voltage, which is like an electrical push or pull. If we make this push strong enough in the opposite direction of the electrons' flight, we can eventually stop them in their tracks. Not a single one gets through!

This specific voltage, the one that's just enough to halt all the outgoing electrons, is our stopping potential. It's like finding the exact amount of wind resistance needed to stop a feather from blowing away. You can't stop it with a tiny puff, but if the wind gets strong enough, eventually it'll hold still.
Why is this "stopping" so important?
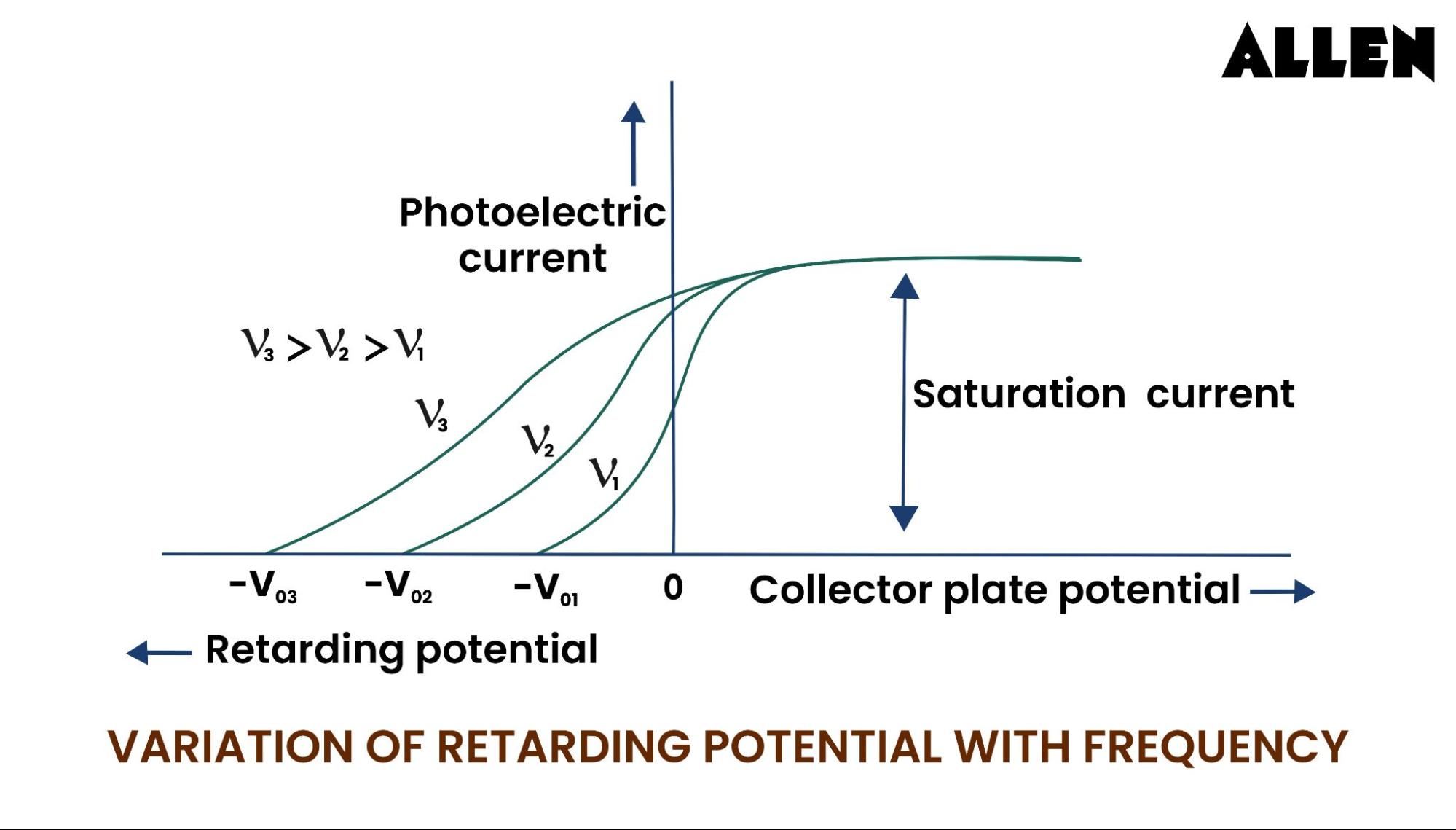
Well, it tells us a lot about the energy of those ejected electrons. Remember how different colors of light have different energies? The brighter the light (more intensity), the more electrons get kicked out. But the color of the light, or its frequency, determines the energy of each individual electron that escapes.
So, if you shine a blue light (higher energy) on the metal, the electrons will zoom off with more oomph. To stop them, you'll need a bigger electrical push – a higher stopping potential. If you use a red light (lower energy), the electrons won't be flying as fast, and you'll need less of a push to stop them.
It's like this: imagine you have a bunch of energetic toddlers. If they're just bouncing around, it’s easy to catch them. But if they’ve had a sugar rush and are sprinting, you need to be quicker and stronger to grab them. The stopping potential is like measuring how fast you need to be to catch all the sprinting toddlers.
Einstein and the Light Quanta
This whole photoelectric effect thing was a big deal, so big that it helped Albert Einstein win his Nobel Prize! Before this, people thought light was just a wave, like ripples on a pond. But the photoelectric effect showed that light also behaves like tiny packets of energy, which we now call photons.
These photons are like little energy bullets. When a photon hits an electron, it transfers its energy. If that energy is enough to overcome the forces holding the electron in the metal, zap, the electron is free! The stopping potential experiment was crucial because it allowed scientists to measure the energy of these photons and, in turn, figure out the energy needed to free an electron from the metal (which is called the work function, another cool term!).

Think of it like trying to guess how much effort it takes to open a stubborn jar. You can try twisting the lid gently, but it won't budge. You need to apply a certain amount of force. The stopping potential is like measuring that minimum force required to get the lid off, but for electrons escaping a metal.
So, when we talk about the stopping potential in a photoelectric effect experiment, we're really talking about a way to measure the maximum kinetic energy of the electrons that are being kicked out by light. Kinetic energy is just the energy of motion, so it's how fast they're zipping around.
The higher the stopping potential, the faster those electrons were moving, and the more energy they had when they left the metal. And that, in turn, tells us about the energy of the light that hit the metal in the first place.
It's a bit like being a detective, isn't it? The light is the clue, the ejected electrons are the suspects, and the stopping potential is the tool that helps us understand how much energy was involved in the "crime" of freeing those electrons.
This seemingly simple measurement – finding the voltage that just stops all the electrons – unlocked a huge understanding of how light and matter interact. It helped pave the way for quantum mechanics, the mind-bending field that explains how the universe works at its tiniest scales.
So, the next time you hear about the "stopping potential," don't let it scare you. It's just a clever way of saying: "How much electrical push do we need to apply to completely stop these electrons that light has just kicked out?" It’s a fundamental tool in understanding the energetic dance between light and matter, and a key piece of the puzzle that led us to a deeper understanding of our universe. Pretty neat, right?
