In A Neutral Solution The Concentration Of _____.

Hey there, curious minds! Ever find yourself staring into a glass of water and wondering what’s really going on in there? It’s easy to think of water as just… well, water. But when we start talking about solutions, things get a little more interesting. And today, we’re diving into a super fundamental idea: in a neutral solution, the concentration of _____. What do you think fills that blank? It’s a question that sounds a bit like a riddle, but the answer is actually pretty neat, and it has implications for everything from your morning coffee to the way our bodies work.
So, let’s unpack this. When we talk about a “solution,” we’re usually thinking about something dissolved in a liquid. Water is our go-to solvent for a lot of things, right? Think of sugar dissolving in your tea or salt in your pasta water. These are all examples of solutions. But what makes a solution “neutral”? That’s where the magic happens.
Think of it like a seesaw. On one side, we have things that make a solution more acidic. On the other side, we have things that make it more basic (or alkaline). A neutral solution is that perfect point where the seesaw is perfectly balanced. It’s not tipping one way or the other. It’s just… chill.
Now, what are these “sides” of the seesaw made of? In chemistry, we often talk about ions. Don’t let that word scare you! Ions are just atoms or molecules that have gained or lost electrons, giving them a positive or negative electrical charge. For our neutral solution puzzle, the two most important ions are hydrogen ions (often written as H⁺) and hydroxide ions (written as OH⁻).
These two ions are like dance partners. They’re constantly being created and interacting in water. In pure water, which is the epitome of a neutral solution, these two dance partners are always found in equal numbers. Isn't that cool? It's like a perfectly choreographed ballroom dance where everyone has a partner and the tempo is just right. No one is left out, and no one is doing too much.
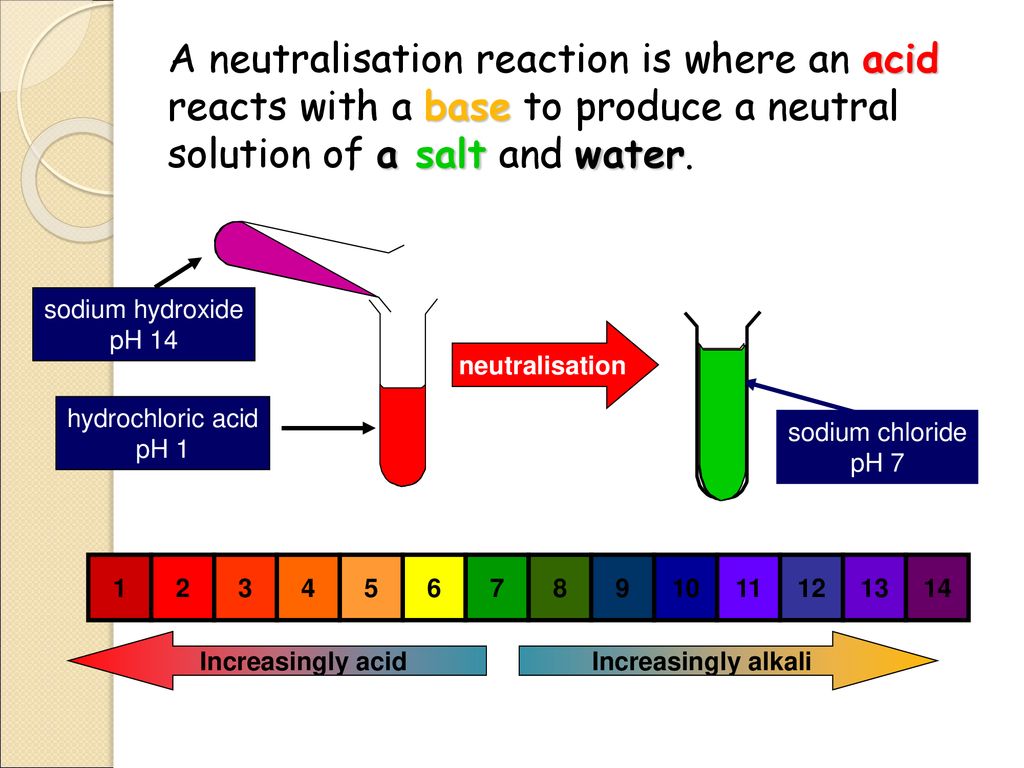
So, back to our riddle: In a neutral solution, the concentration of _____ is equal to the concentration of _____. You guessed it! It’s the concentration of hydrogen ions that is equal to the concentration of hydroxide ions. Ta-da! It’s that simple, and yet, it explains so much.
Why is this balance so important?
This equal concentration is the very definition of neutrality. If there were more hydrogen ions, the solution would become acidic. Think of lemon juice – it’s acidic because it has a lot of H⁺ ions. If there were more hydroxide ions, the solution would become basic. Think of baking soda – it’s basic, meaning it has more OH⁻ ions.
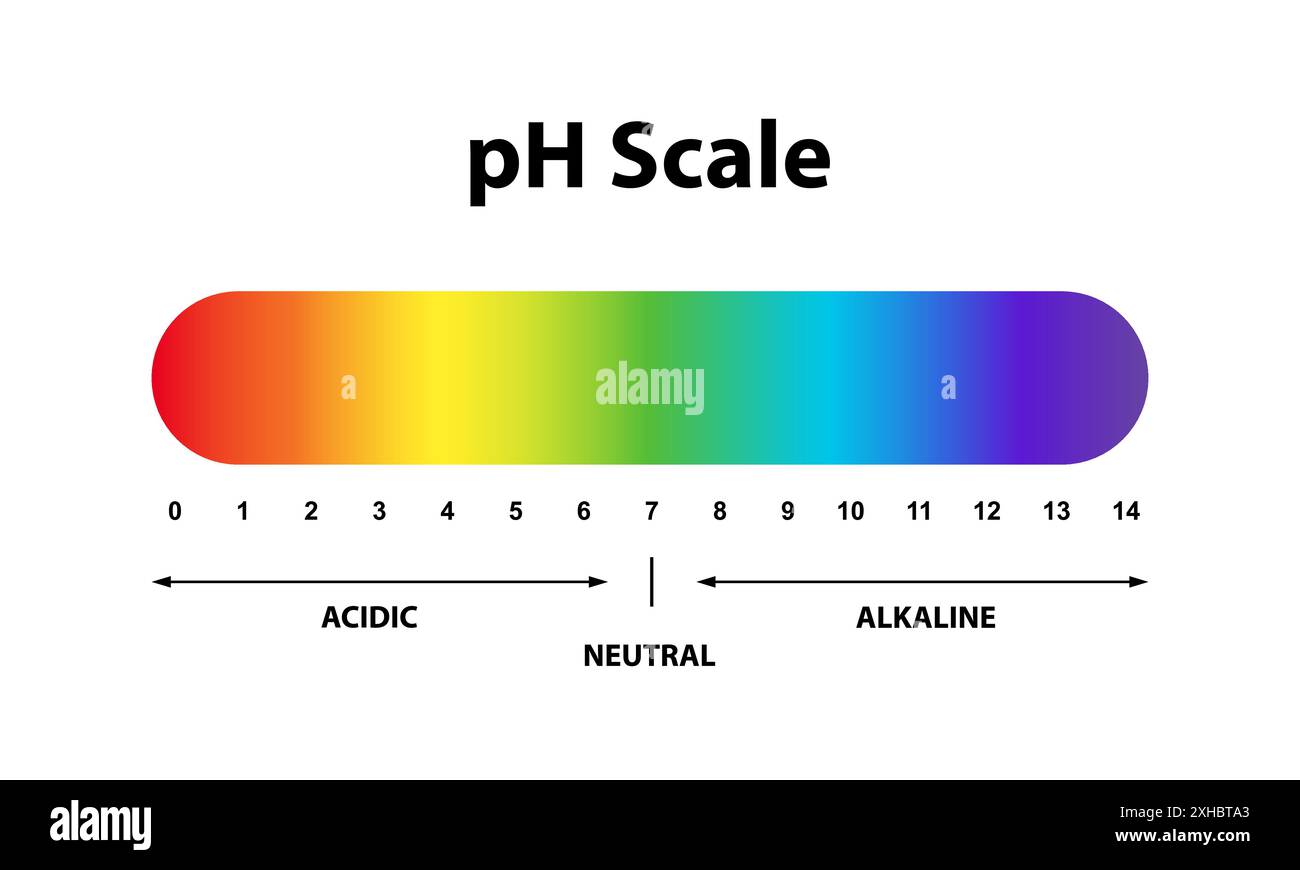
The pH scale, that familiar scale from 0 to 14, is actually a way of measuring the concentration of these hydrogen ions. A pH of 7 is neutral, meaning the H⁺ and OH⁻ ions are in perfect balance. Anything below 7 is acidic, and anything above 7 is basic. So, that neutral glass of water you’re picturing? It has a pH of 7. It’s hanging out right in the middle, totally serene.
Imagine a perfectly balanced scale. On one side, you have a single perfectly formed chocolate chip cookie. On the other side, you have another single perfectly formed chocolate chip cookie. That’s your neutral solution – a perfect 1:1 ratio. If you add more cookies to one side, the scale tips – that’s acidity or basicity kicking in.
Let’s get a little geeky (but still chill!)
In pure water at room temperature (around 25°C, which is pretty standard lab temperature), the concentration of both H⁺ and OH⁻ ions is about 1 x 10⁻⁷ moles per liter. Now, that 10⁻⁷ might look like a tiny, insignificant number, and in many contexts, it is. But in the world of chemistry, it’s the foundation of a whole lot of processes.

Think of it this way: if you had a million little boxes, and in each box, you put one grain of sand, that’s a pretty spread-out distribution. But in a tiny, confined space like a beaker of water, having 10⁻⁷ moles per liter means there are still a lot of these ions zooming around. It’s just that for every hydrogen ion, there’s a hydroxide ion right there with it.
This balance is so delicate. Even a tiny change can shift things. For example, adding just a little bit of acid to water will release extra H⁺ ions, making the concentration of H⁺ higher than OH⁻, and thus, the solution becomes acidic.
This isn’t just a theoretical concept confined to a lab. This principle is vital for life itself! Your blood, for instance, needs to be kept at a very specific pH, just slightly basic (around 7.35-7.45). If it drifts too far from that neutral-ish zone, even by a little bit, serious problems can arise. Your body has amazing buffering systems that work to keep those H⁺ and OH⁻ concentrations in check, ensuring everything runs smoothly.

Think about cooking. When you bake bread, the yeast thrives in a slightly acidic environment. But if it gets too acidic, the yeast won't work. The right balance of ions is crucial. Or consider how we clean things. Many cleaning products are basic because bases are good at breaking down grease and oils, which are acidic in nature. It’s all about that chemical tug-of-war, and neutrality is the peaceful coexistence.
So, what’s the takeaway?
The next time you take a sip of water, or pour yourself a cup of tea, remember the incredible molecular dance happening within. In that seemingly simple liquid, a constant equilibrium exists. In a neutral solution, the concentration of hydrogen ions is perfectly matched by the concentration of hydroxide ions. This fundamental balance is the bedrock of the pH scale, the key to understanding acidity and basicity, and absolutely essential for countless biological and chemical processes around us.
It’s a reminder that even in the most ordinary things, there’s a hidden world of fascinating chemistry at play. So keep asking those questions, keep being curious, because understanding these basic principles can really unlock a deeper appreciation for the world we live in!
