If You Were To Isolate A Molecule Containing Uracil
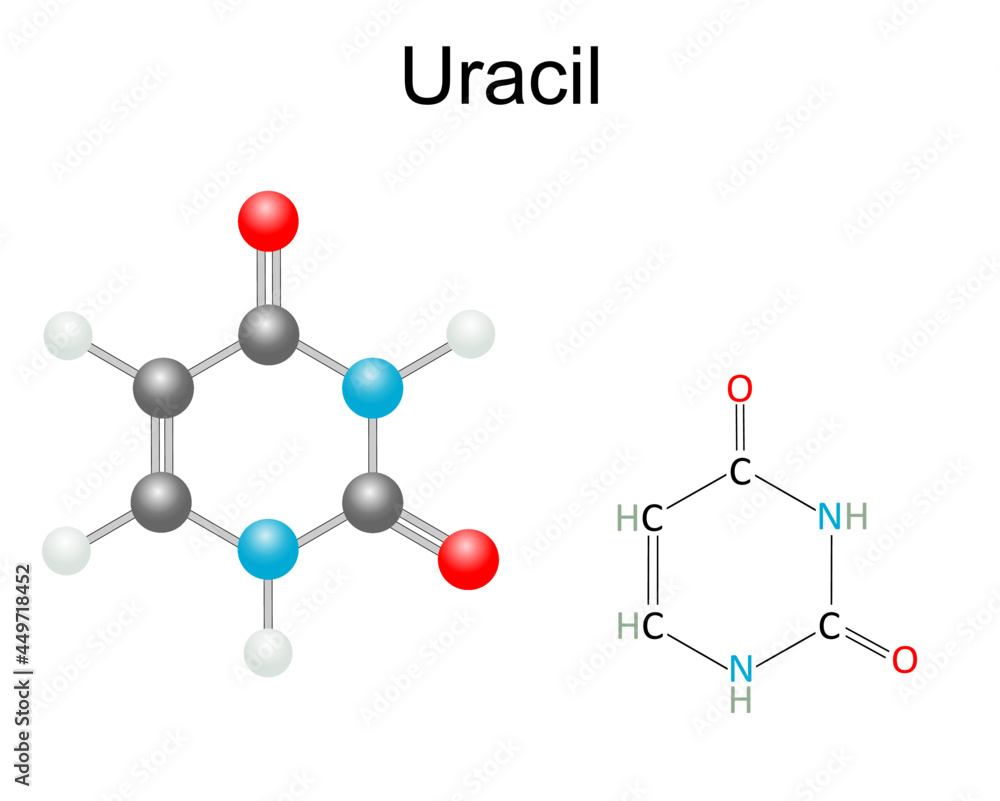
So, imagine you’re tinkering around in your kitchen, right? You’ve got all sorts of bits and bobs, spices, flour, maybe even some questionable leftovers lurking in the back of the fridge. Now, what if your mission, should you choose to accept it (and let’s be honest, who doesn’t love a good kitchen science experiment?), was to isolate just one specific ingredient from that whole glorious mess? Not just any ingredient, mind you, but something super particular, like… say, uracil.
Yeah, uracil. Sounds a bit fancy, doesn’t it? Like something you’d find on the ingredient list of a gourmet dog food or a space helmet cleaning kit. But here’s the kicker: uracil is actually a pretty darn important little molecule. It’s one of the building blocks of RNA, which is like the delivery guy of your cells, carrying all sorts of vital instructions around. Think of it as the cellular equivalent of a pizza order arriving at your door – essential for survival, and nobody wants a soggy, late pizza, right?
Now, isolating uracil is a bit like trying to find that one specific, slightly-off-color sprinkle on top of a mountain of cupcakes. You know it’s there, you think you know what it looks like, but getting just that one little guy without accidentally scooping up a whole bunch of frosting and cake? Tricky business. It’s not like you can just pull it out with your fingers, unless your fingers happen to be equipped with molecular-level tweezers. And let’s be real, most of us are still trying to master the art of not dropping our toast butter-side down.
The whole process of isolating something like uracil is basically a highly scientific, super-sophisticated version of playing "Where's Waldo?" but with molecules instead of that horizontally-striped guy. And trust me, Waldo is way easier to spot than a single uracil molecule. At least Waldo doesn't dissolve into a hundred other bits if you breathe on him wrong.
Think about it: you’ve got your sample – let’s call it your "molecular soup." This soup could be anything. It could be some ancient, forgotten bacterial culture from the depths of your compost bin (don’t judge, we’ve all been there), or perhaps it’s a carefully prepared solution of cellular goo from a lab. Either way, it’s a complex concoction, a real medley of different molecules all chilling together. You’ve got proteins, fats, sugars, and of course, a whole bunch of other nucleic acid building blocks that are practically uracil’s identical twins, just with slight personality quirks. You know, like that cousin who looks exactly like you but insists on wearing socks with sandals. They're related, but different.
So, how do you even begin to pull out your target molecule, our shy little uracil? It’s not like you can just offer it a tiny bribe, like "Psst, uracil, I've got some extra ATP over here, fancy a trade?" Nope. You need to be a bit more strategic. It’s like trying to catch a specific butterfly in a field absolutely buzzing with them. You need patience, precision, and a whole lot of scientific know-how. And probably a very fine net, or in this case, some very clever chemical tricks.
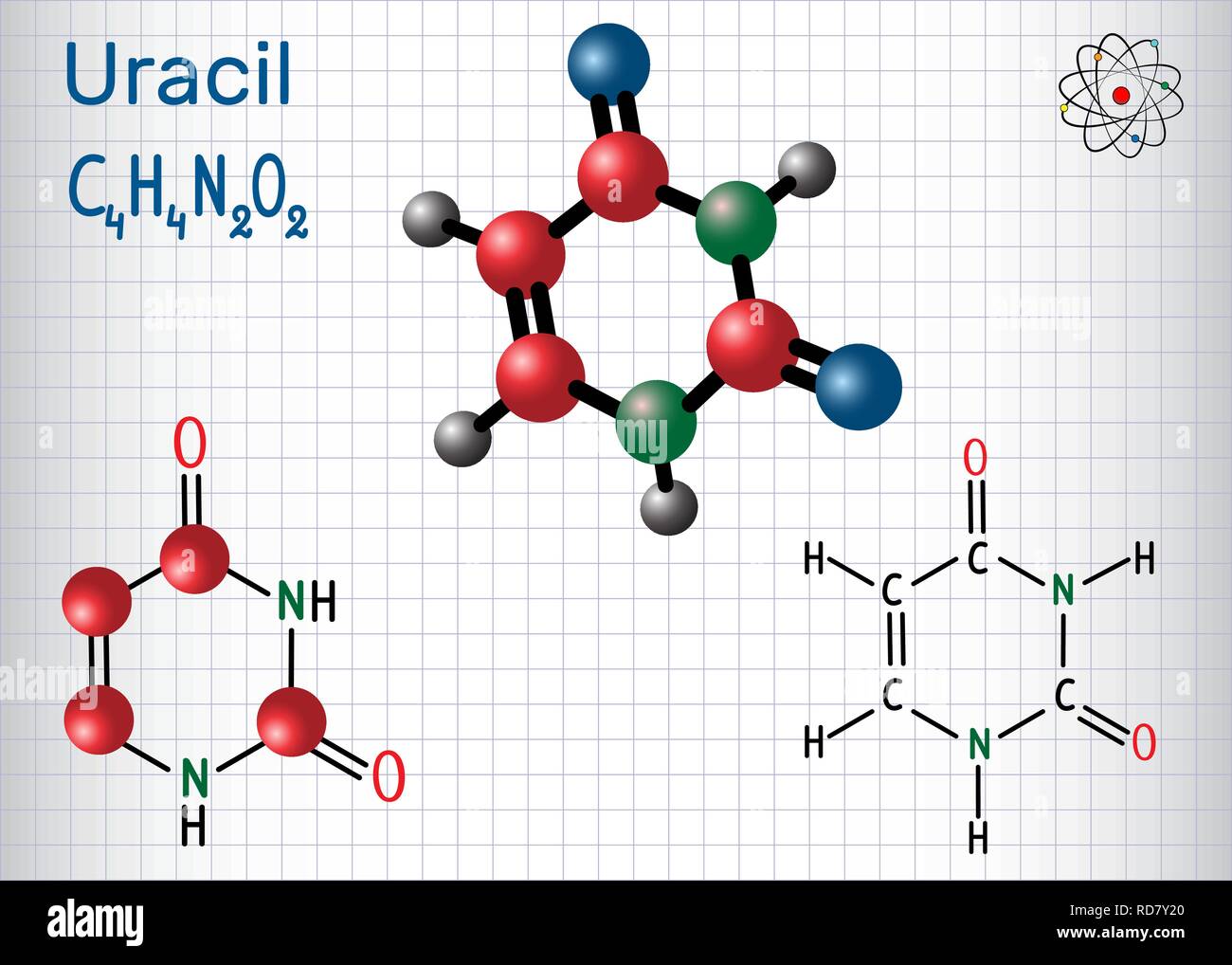
One of the classic moves in the molecular isolation playbook is called chromatography. Don't let the fancy name scare you. Think of it like a highly organized race where the molecules have to run through a special obstacle course. This obstacle course is usually a column packed with some sort of material, like tiny beads or a special gel. As your molecular soup flows through, different molecules interact with the packing material in different ways. Some get stuck more easily, some zoom right through. It’s all about their stickiness and their size.
Imagine you’re trying to sort M&Ms by color, but instead of just picking them up, you’re pouring them through a sieve that has different-sized holes, and some of the M&Ms are coated in a special, sticky substance. The ones that are less sticky and smaller might fall through faster, while the bigger, stickier ones get caught up for longer. Your uracil, with its unique chemical properties, will have its own particular pace through this molecular racecourse. It’s about exploiting those tiny differences that make our uracil, well, uracil.
Another approach, if you’re feeling particularly ambitious, is spectroscopy. This is like shining a special kind of light on your molecular soup and seeing how each molecule reacts to it. Different molecules absorb and reflect light in distinct ways. Think of it like holding up a prism to sunlight. You get a beautiful rainbow, right? That rainbow is made up of different wavelengths of light, and each molecule in your sample will have its own unique "light fingerprint." By analyzing which wavelengths are absorbed or emitted, you can identify and even quantify your uracil. It’s like giving each molecule a tiny, personal concert and then listening very, very carefully to the music.
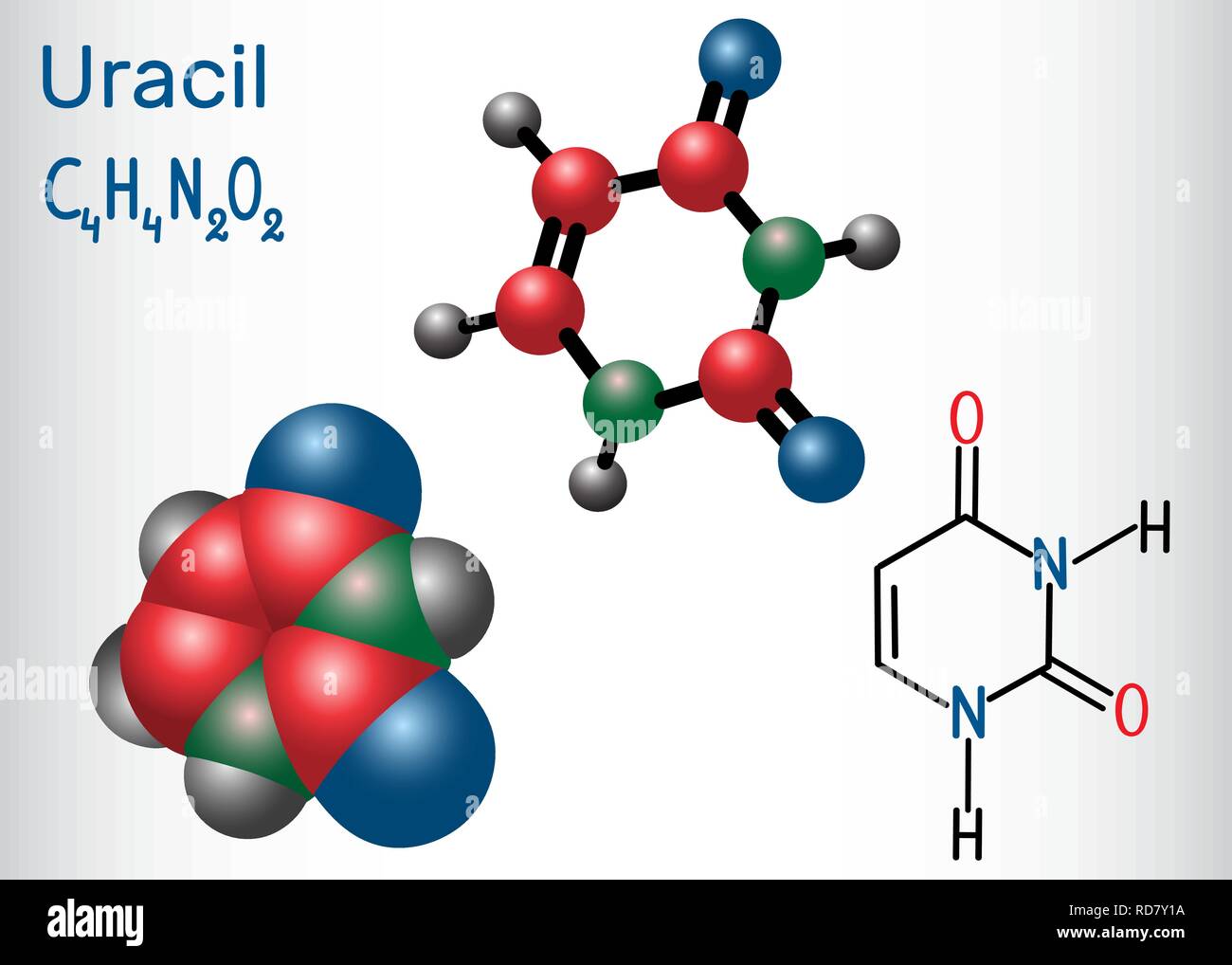
And then there’s electrophoresis. This one is a bit more dramatic. Imagine your molecules are like tiny, charged marbles, and you’re rolling them down a slope. Except, instead of gravity, you’re using an electric field to pull them along. Because molecules can have a positive or negative charge, and they come in different sizes, they’ll move at different speeds through a gel matrix (think of it as a microscopic maze). Your uracil, with its specific charge and size, will trundle along at its own unique speed. It’s like a microscopic traffic jam, and you’re watching to see who gets stuck where.
Honestly, the whole thing can feel a bit like being a molecular detective. You’ve got your clues – the chemical properties of uracil – and you’ve got your crime scene – the messy molecular soup. Your job is to piece together the evidence, use your fancy tools, and finally, aha! point to that single uracil molecule and say, "There you are!" It's a moment of pure scientific triumph, a bit like finding the remote control that’s been hiding under the sofa cushions for weeks.
The techniques used for isolation are all about finding that sweet spot where your uracil can be separated from everything else. It’s like trying to pick out your favorite song from a radio station that's playing about fifty different tracks at once, all mashed together. You need to tune in to the exact frequency of uracil. You’re looking for its unique chemical personality, its electronic charge, its molecular weight, and how it interacts with its neighbors. It’s like getting to know your neighbors really, really well, down to their molecular level.

And once you’ve got it? Well, that’s when the real fun (or the next phase of really intense scientific work) begins. You’ve isolated your uracil! You can then study it, use it for further experiments, or just admire it for its sheer molecular persistence. It’s like finally catching that elusive Pokémon, except instead of virtual glory, you get the satisfaction of scientific discovery. And maybe a really cool paper to publish.
The beauty of these isolation techniques is that they are incredibly versatile. Whether you're a seasoned biochemist in a gleaming lab or a curious amateur trying to understand the building blocks of life from your garage (again, no judgment!), the underlying principles are the same. It’s all about understanding the subtle differences that make each molecule unique, and then cleverly exploiting those differences to get what you want.
It's a bit like being a master chef. You have a pantry full of ingredients, and you need to pick out the perfect spice to elevate your dish. You can't just throw everything in. You need to understand the unique flavor profile of each spice, how it interacts with other ingredients, and the exact moment to add it. Isolating uracil is the molecular equivalent of picking that perfect pinch of saffron to give your rice dish that special je ne sais quoi.

Sometimes, the process involves making your uracil change slightly so it becomes easier to spot. You might, for instance, tag it with something that glows under UV light. Then, it's like putting a tiny neon sign on your uracil, saying "Over here! I'm the special one!" This is a bit like adding a bright, flashing LED to your car keys when you know they’re about to go missing. Genius, really.
The ultimate goal of isolating uracil, or any other molecule for that matter, is to gain knowledge. We want to understand how these tiny components work, how they interact, and how they contribute to the vast, complex symphony of life. It’s like taking apart a clock to understand how the gears turn and how the springs provide the energy. Except instead of tiny cogs, we’re dealing with atoms and electrons, and instead of a tick-tock, we’re talking about the very essence of biological processes.
So, the next time you hear about a scientific breakthrough involving molecules, remember that behind those impressive headlines, there’s often a meticulous, sometimes painstaking, but always fascinating process of isolation. It’s the molecular equivalent of treasure hunting, where the treasure is a tiny, fundamental piece of the puzzle of life itself. And who knows, maybe one day, you'll be in your own kitchen, with your own molecular soup, embarking on your own quest to find that elusive little molecule. Just try not to spill the soup on the floor. That’s a whole different kind of cleanup mission.
