If Equal Masses Of The Following Compounds Undergo Complete Combustion

Imagine a world where science meets your everyday vibe. Today, we're diving into something that sounds a little… sciency, but trust us, it’s all about living smarter and maybe even a little greener. Think of it as your ultimate guide to understanding what happens when things go up in smoke, but with a laid-back, magazine-ready twist. We’re talking about complete combustion – that moment when a substance burns all the way through, leaving behind nothing but its fundamental building blocks. And the best part? We’re exploring this from a purely equal mass perspective. No complex calculations here, just pure, unadulterated curiosity for the modern minimalist.
So, what’s the big deal about equal masses? It’s a bit like comparing apples and oranges, but when you’re talking about burning things, it helps us understand the efficiency of different fuels. If you have the same weight of, say, wood versus gasoline, which one gives you more bang for your buck, energetically speaking? It’s a question that might pop up when you’re planning a backyard BBQ, thinking about car choices, or even just appreciating the power in a roaring fireplace. We’re going to break down how some common compounds stack up when they’re pushed to their fiery limits, all while keeping it light and breezy.
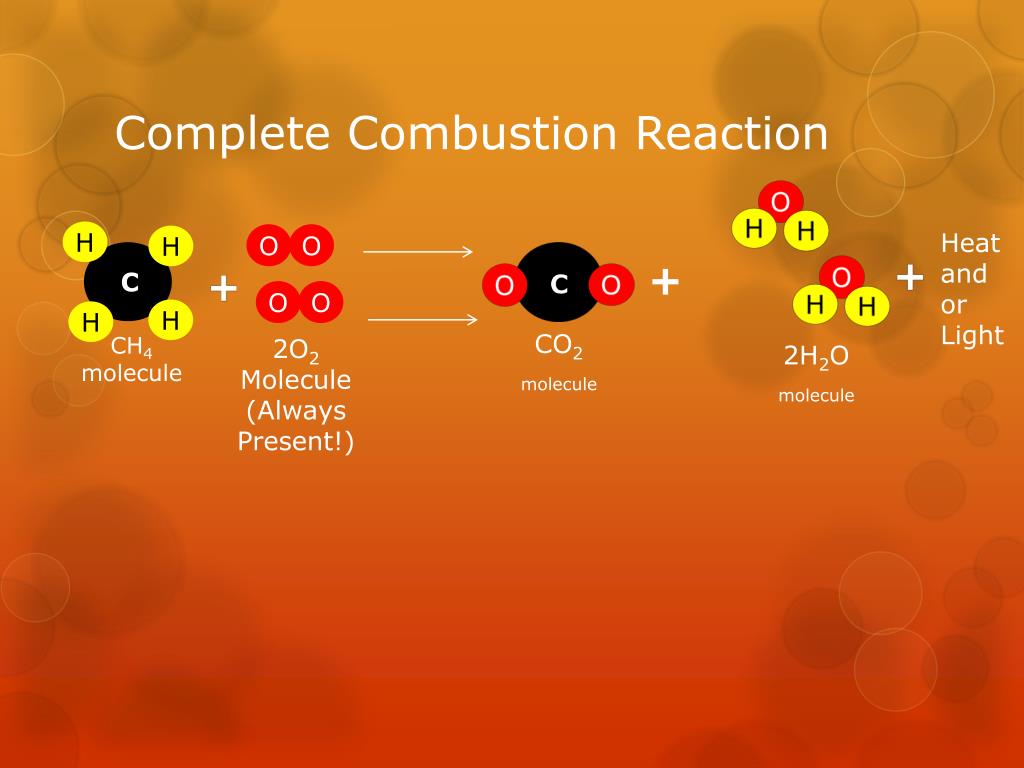
Let’s start with a household name: methane. You know it as the main ingredient in natural gas, the stuff that heats your home and cooks your dinner. When methane (CH₄) goes through complete combustion, it teams up with oxygen (O₂) to produce carbon dioxide (CO₂) and water (H₂O). It’s a pretty clean burn, relatively speaking. For every molecule of methane, you get a good amount of energy released. Think of it as the reliable friend in the fuel world – it does its job efficiently and doesn't leave too much of a mess behind. This is why natural gas is such a popular choice for heating and cooking in many homes. It’s readily available and, when burned completely, has a lower impact compared to some other fossil fuels.
Now, let’s talk about its slightly more complex cousin, propane. Propane (C₃H₈) is often found in those big tanks for barbecues or RVs. It’s a hydrocarbon, just like methane, but with more carbon atoms. When propane burns, it also produces carbon dioxide and water. However, because it has more carbon atoms per molecule, equal masses of propane will actually release more energy than equal masses of methane. It’s like upgrading from a trusty sedan to a slightly more powerful SUV – same principle, but with a bit more oomph. This extra energy density makes propane a great portable fuel source for outdoor cooking and heating.
Ever wonder about that classic campfire smell? That’s often the scent of wood undergoing combustion. While wood isn’t a single chemical compound, its primary combustible component is cellulose, which is a complex carbohydrate. When wood burns, it’s a bit more complicated than burning a pure gas. You get carbon dioxide, water, and a whole host of other things, including soot and ash. Because wood is less dense and contains a lot of water and other non-combustible materials, equal masses of wood will yield significantly less usable energy compared to methane or propane. It’s the romantic choice, the nostalgic fuel, but not necessarily the most energetically efficient pound-for-pound. This is why you need a lot more wood to generate the same amount of heat as a smaller amount of natural gas.
Moving onto a liquid superstar: ethanol. This is the alcohol you might find in some biofuels. Ethanol (C₂H₅OH) also burns cleanly, producing carbon dioxide and water. When you compare equal masses, ethanol is actually quite a good performer, often releasing more energy than methane. It’s a bit of a crowd-pleaser in the fuel world, offering a balance of energy output and a relatively cleaner burn profile. Many people are interested in ethanol as a renewable fuel source, and its combustion characteristics make it a viable option. It’s the kind of fuel that feels good to use, knowing it can be derived from plants. Think of it as the sustainably-minded performer, hitting a sweet spot.
What about something we encounter in a different context, like sugar? Yes, that sweet stuff we love in our desserts, chemically speaking, is a carbohydrate like cellulose. Sucrose (C₁₂H₂₂O₁₁), common table sugar, will combust to produce carbon dioxide and water. However, when you compare equal masses of sugar to something like methane, you'll find that sugar releases less energy. It’s not as potent a fuel source as the hydrocarbons. While it’s a source of energy for our bodies, it’s not the first thing you’d reach for to power a generator. It’s more about that immediate energy boost for your afternoon slump, not for industrial-scale power.
Let's introduce a bit of a curveball: hydrogen. Hydrogen gas (H₂) is often touted as the fuel of the future because its combustion produces only water. That’s right, only water! This makes it incredibly clean from an emissions standpoint. However, when we talk about equal masses, hydrogen is an absolute powerhouse. For the same weight, hydrogen releases a staggering amount of energy compared to any of the hydrocarbons. It's the lightweight champion that packs the biggest punch. The challenge with hydrogen isn't its energy output, but rather its storage and production. It's like the genius with a brilliant idea who needs a bit of help getting it off the ground.
Now, let’s touch on something a bit less common in everyday combustion scenarios but important for understanding the spectrum: carbon monoxide (CO). This is a partial combustion product of carbon-containing materials, and it’s notoriously dangerous. If equal masses of carbon monoxide were to undergo complete combustion (meaning it reacts further to form carbon dioxide), it would release a significant amount of energy. However, the real-world concern with CO isn't its potential energy release, but its presence as a toxic gas. It’s a reminder that incomplete combustion can be a real problem, turning a useful fuel into a harmful byproduct. This highlights the importance of proper ventilation and ensuring your appliances are functioning correctly.

Consider the simple element, carbon itself, in its pure form, like charcoal. When charcoal burns completely, it forms carbon dioxide. If you were to compare equal masses of pure carbon to methane, methane would likely release more energy per unit mass because of the hydrogen atoms it contains. Carbon is a fundamental building block, and while it burns, it's the combination with hydrogen in hydrocarbons that often gives us that extra energy density we associate with fuels. Think of carbon as the essential foundation, and hydrocarbons as the elaborately constructed buildings.
We’ve talked a lot about energy release, but what about the "waste" products? Complete combustion of hydrocarbons primarily produces carbon dioxide and water. Carbon dioxide is a greenhouse gas, which is a major concern for our planet’s climate. Water, on the other hand, is harmless. The more carbon atoms in a fuel, the more carbon dioxide is produced for the same amount of energy released, generally speaking. This is why there's a push towards fuels with lower carbon content or fuels that produce fewer greenhouse gases overall.
This exploration into equal masses and combustion isn't just an academic exercise. It has real-world implications for how we choose our energy sources and understand our impact. When you're at the supermarket, thinking about packaged goods, or when you’re looking at your energy bills, these concepts are quietly at play. Understanding that different fuels have different energy densities and produce different byproducts helps us make more informed decisions.

Think about your morning coffee. The beans were grown, processed, and transported. All these processes involved energy. The mug you drink from was likely made using energy-intensive processes. Even the act of brewing your coffee requires energy. While we’re not directly burning coffee beans, the principle of energy conversion and efficiency is everywhere.
For instance, when we talk about biofuels like ethanol, we're looking at renewable resources that can offer a more sustainable energy pathway. The "equal mass" comparison helps us quantify their potential contribution compared to traditional fossil fuels. It’s about finding that sweet spot where power meets responsibility. It’s like choosing between a fast-fashion item and a well-made, ethically sourced piece. Both serve a purpose, but one has a different story and a different impact.
The cultural connection is fascinating too. For centuries, fire has been central to human civilization – for warmth, cooking, and storytelling. From the hearth fires of ancient homes to the roaring furnaces of industry, fire has shaped our world. Understanding the science behind its power, even in a simplified way, connects us to that long history. We’re still harnessing the same fundamental forces that our ancestors did, just with much more sophisticated tools and knowledge.

So, next time you're enjoying a campfire, firing up your grill, or simply flicking on a light switch, take a moment to appreciate the invisible dance of molecules happening behind the scenes. It’s a reminder that even the simplest acts have a complex and fascinating scientific story behind them. And who knows, maybe this little bit of knowledge will inspire you to explore more sustainable options or simply appreciate the energy that powers your daily life a little bit more. It's all about living with a bit more awareness, a bit more wonder, and a lot more ease.
Ultimately, this isn't about becoming a chemistry whiz overnight. It's about a gentle curiosity, a willingness to peek behind the curtain of everyday phenomena. It’s about appreciating that the world around us, from the fuel in our cars to the food on our plates, is a constant interplay of energy and matter. And when we understand that, even at a basic level, we can navigate our lives with a little more intention and a lot more appreciation for the remarkable processes that sustain us.
It's the same principle when you think about packing for a trip. Do you pack a few versatile, high-energy-density items that can be combined in multiple ways, or a lot of smaller, less efficient things? It’s about smart choices, about maximizing what you have. And in the grand scheme of things, when we talk about energy, those smart choices become even more crucial. So, let's keep exploring, keep questioning, and keep living our lives with a touch of scientific wonder.
