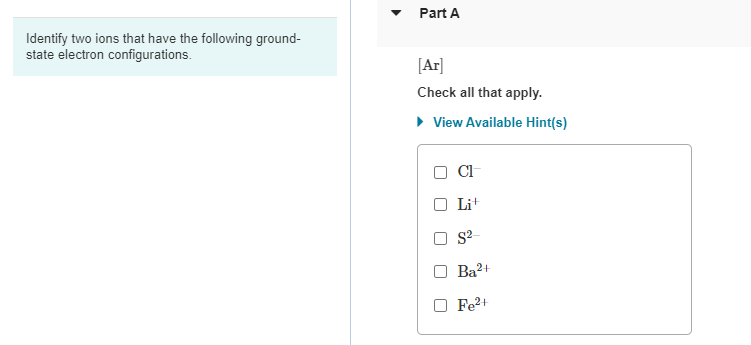
Identify Two Ions That Have The Following Ground-state Electron Configurations
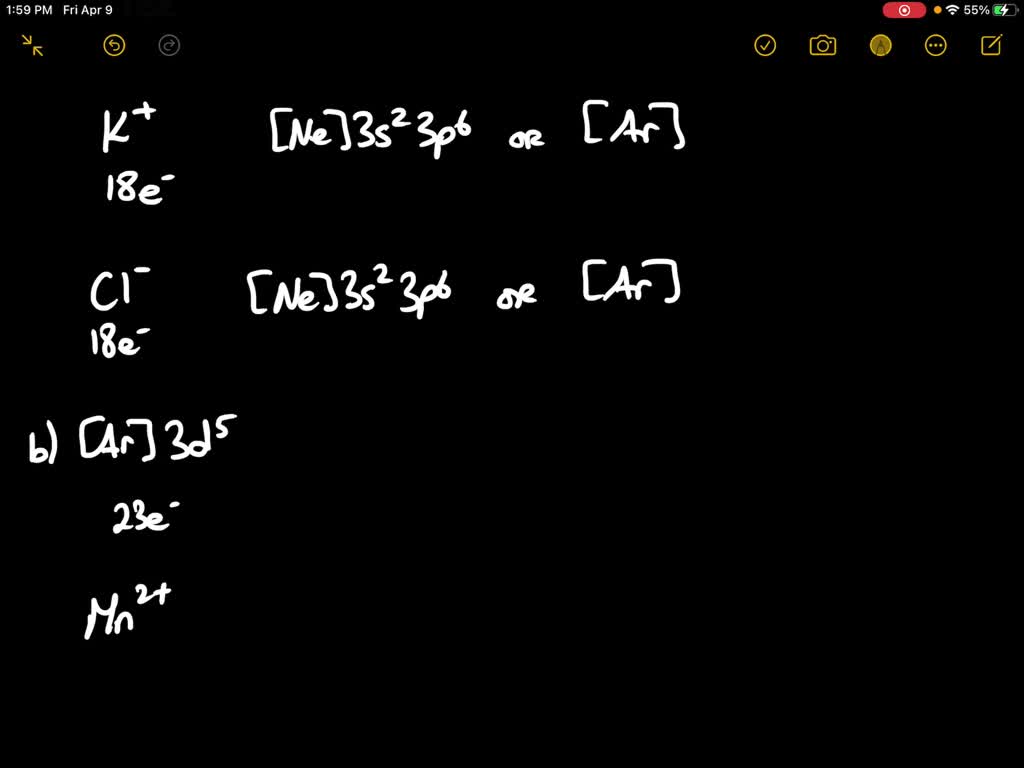
Alright, so imagine you're at a party, right? And everyone's got their own little vibe, their own way of being. Some folks are just happy to chill in the corner with a snack, others are the life of the party, and then you've got the ones who are a little bit… extra. Chemistry is kinda like that, but with atoms and their electrons.
Today, we're talking about ions. Think of an ion as an atom that's decided to either borrow an electron or lend one out. It’s like that friend who always forgets their charger and asks to use yours, or the one who’s got way too many pens and just hands one over. It all boils down to achieving a state of happy balance, just like we all crave that good vibe at a party.
So, what’s this "ground-state electron configuration" thing? Easy peasy. Electrons are those tiny things buzzing around the center of an atom. They hang out in different energy levels, like different floors in a building. The "ground state" is just the most stable and lowest energy arrangement of these electrons. It's like everyone settling into their favorite spot on the couch, not wanting to move unless they absolutely have to. No unnecessary fidgeting!
Now, the trick is that atoms love to have a full outer shell of electrons. It's their version of having a perfectly organized closet. This full shell makes them feel all snug and content. When an atom doesn't have a full outer shell, it’s a bit like having a single sock missing from a pair – it just feels incomplete. So, to achieve that perfect, full outer shell, they might gain or lose electrons, and poof! they become an ion.
Our mission today is to play detective and sniff out two specific ions. We're looking for ions whose ground-state electron configurations are identical. Think of it as finding two people at that party who have the exact same obscure hobby. "Wait, you also collect vintage sporks? No way!" It’s that moment of unexpected connection.
So, how do we find these electron-configuration twins? We need to peek at the electron configuration of neutral atoms first. A neutral atom has a perfectly balanced number of protons (the positive guys in the nucleus) and electrons (the negative guys zipping around). No net charge, just chilling.
Let's start with a common element, something you might even have in your kitchen: sodium (Na). Sodium is in Group 1 of the periodic table, that column of elements that are all a bit reactive. It has 11 protons and, in its neutral state, 11 electrons. Its electron configuration is like this:
1s² 2s² 2p⁶ 3s¹
See that last "3s¹"? That single electron in the outermost shell is like a little kid with a single toy, desperately wanting a friend to play with it. It's a bit of a loner, and it's much easier for sodium to just give away that one electron than to try and find seven more to fill up its outer shell. It's like deciding to declutter by giving away that one item you never use.
When sodium loses that one electron, it becomes a sodium ion (Na⁺). It now has 10 electrons. And guess what its electron configuration becomes?
1s² 2s² 2p⁶
Notice anything? That outer shell (the second shell) is now full! It's got those eight electrons, a complete set. It's like finally finding the matching sock, or organizing your spice rack alphabetically. Pure bliss. This is its stable configuration. No more unwanted fidgeting.
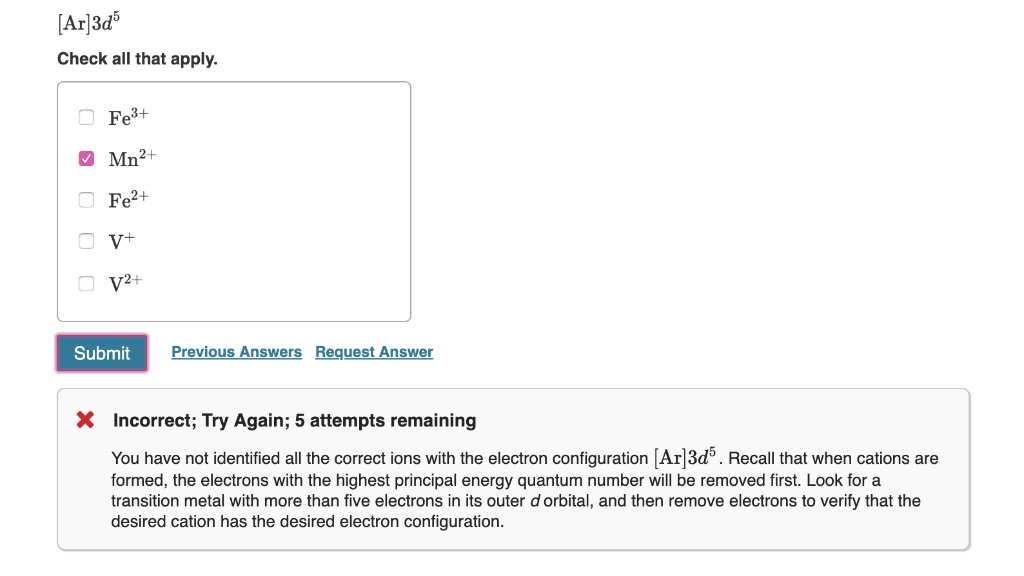
So, we've found one ion: Na⁺. Now, we need to find another ion that has the exact same electron configuration: 1s² 2s² 2p⁶. Where else can we find this perfectly contented arrangement of electrons?
Let's think about elements that gain electrons to achieve that full outer shell. Remember how sodium lost one electron to get to this configuration? Maybe there's an element that's just one electron short of having a full outer shell of its own. That way, if it gains that one electron, it'll end up with the same number of electrons as our happy sodium ion.
Consider chlorine (Cl). Chlorine is in Group 17 of the periodic table, known as the halogens. These guys are the ultimate electron-grabbers. A neutral chlorine atom has 17 protons and 17 electrons. Its electron configuration looks like this:
1s² 2s² 2p⁶ 3s² 3p⁵
Look at that outer shell (the third shell). It’s got 2 + 5 = 7 electrons. It’s so close to being full! It’s like being one ingredient away from making that perfect batch of cookies. It’s yearning for just one more electron to complete its set. It's practically begging someone to pass it that last egg.
When a chlorine atom gains one electron, it becomes a chloride ion (Cl⁻). It now has 17 protons and 18 electrons (the original 17 plus the one it gained). Its electron configuration is:
1s² 2s² 2p⁶ 3s² 3p⁶
Wait a minute. That's not the same as our sodium ion. My bad! I got a little excited about chlorine's eagerness. Let's backtrack. We're looking for an ion with 1s² 2s² 2p⁶. This configuration has a total of 2 + 2 + 6 = 10 electrons.
So, we need an ion that ends up with exactly 10 electrons. We already know Na⁺ has 10 electrons. It started with 11 and lost 1. What other element, when it becomes an ion, can end up with 10 electrons?
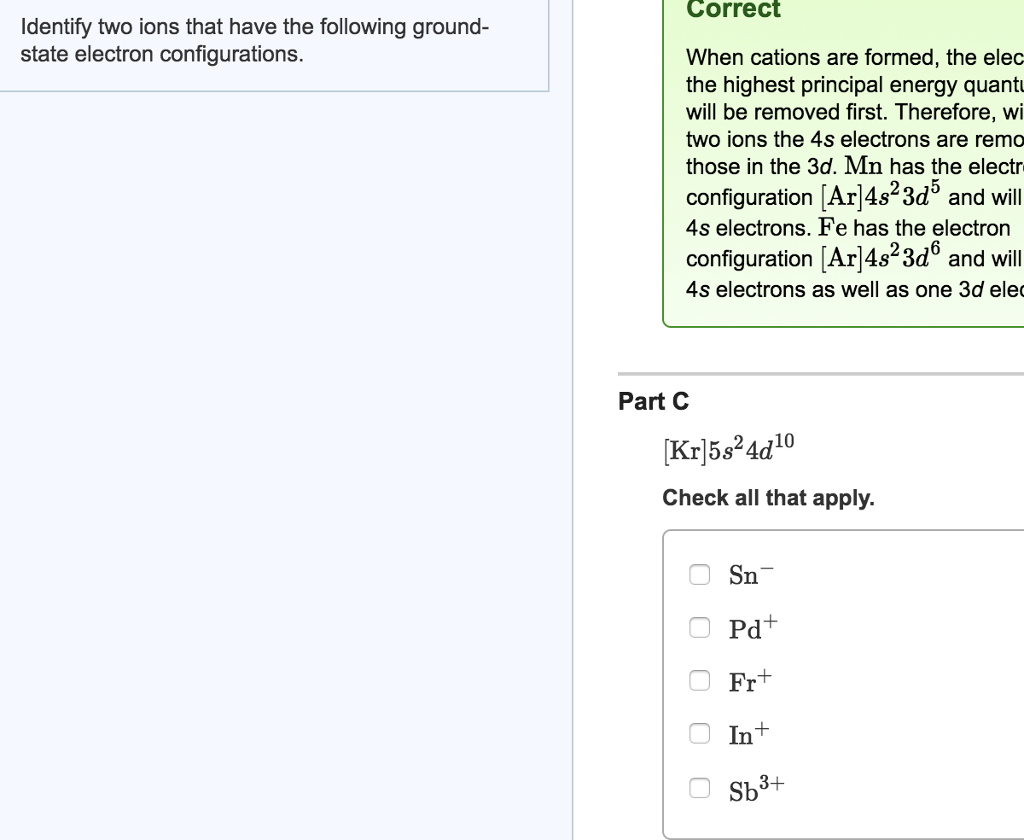
Let’s think about elements that are close to having 10 electrons in their neutral state. What if we look at something with fewer electrons than sodium, but it gains some?
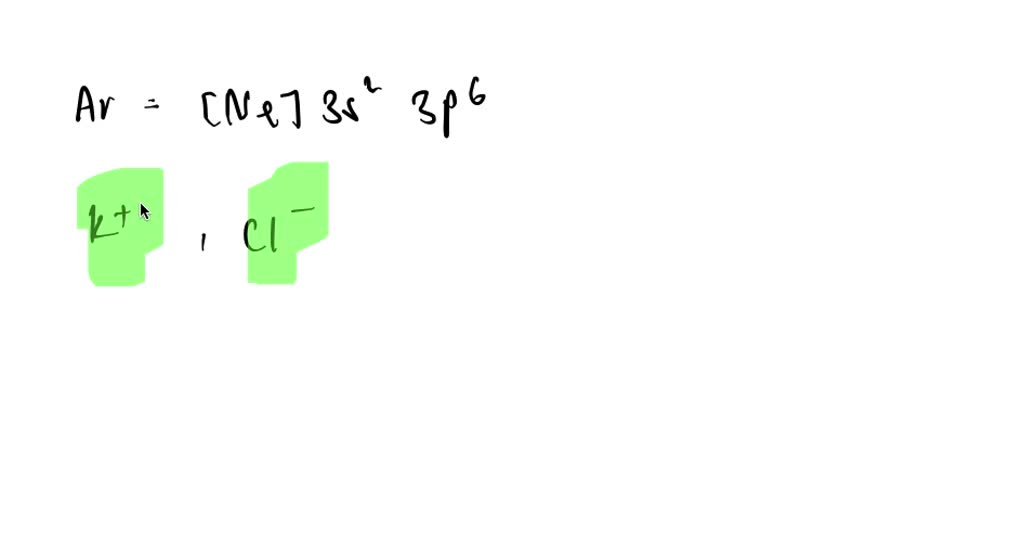
Consider oxygen (O). Oxygen has 8 protons and, in its neutral state, 8 electrons. Its electron configuration is:
1s² 2s² 2p⁴
Its outer shell (the second shell) has 2 + 4 = 6 electrons. It’s two electrons short of a full outer shell. It’s like a puzzle missing two key pieces. So, oxygen often gains two electrons to become an oxide ion (O²⁻). Its electron configuration then becomes:
1s² 2s² 2p⁶
Aha! We found a match! Oxygen, when it forms the oxide ion (O²⁻), has the same ground-state electron configuration as the sodium ion (Na⁺). Both have 10 electrons arranged as 1s² 2s² 2p⁶. It’s like finding two completely different people who both learned to play the ukulele and are now jamming together. They started at different places, but they ended up with the same musical output.
So, our two ions are Na⁺ and O²⁻. Both have achieved that coveted, stable, full outer shell by gaining or losing electrons, and in doing so, they ended up with the identical electron arrangement of 1s² 2s² 2p⁶. They're like two different paths leading to the same beautiful destination.
Why is this whole "full outer shell" thing such a big deal? Well, elements that have a full outer shell are incredibly stable and unreactive. They're like that person who’s completely content and doesn’t need any drama. They’ve got everything they need, and they’re not looking to make any sudden changes. Think of the noble gases, like Neon (Ne) or Argon (Ar). Their electron configurations are already like 1s² 2s² 2p⁶ or 1s² 2s² 2p⁶ 3s² 3p⁶. They are the ultimate chillers of the periodic table. They're the ones at the party who brought their own perfectly chilled beverage and are perfectly happy just observing.
So, when sodium loses an electron to become Na⁺, it’s essentially mimicking the electron configuration of Neon. It’s like adopting the coolness of the popular kid. Similarly, when oxygen gains two electrons to become O²⁻, it’s also mimicking Neon's electron configuration. They’re both trying to achieve that "noble gas" status. It's the ultimate goal, the chemist's version of winning the lottery.
It's fascinating, isn't it? How these tiny, invisible particles behave. They're constantly seeking balance, striving for that perfect electron arrangement. And in their quest for stability, they sometimes end up looking exactly alike, even if they started from completely different places.
Think of it like this: you might start your day feeling a bit scattered, maybe you forgot your keys or spilled coffee on your shirt. That's your "neutral atom" phase – a bit unorganized. But then you figure things out, you find your keys, you clean up the spill. By the end of the day, you feel put together and balanced, ready for whatever comes next. That's your "ion" phase, your state of achieved stability.
The electron configuration is just the blueprint of how those electrons are arranged. And when two different atoms, through the process of becoming ions, end up with the exact same blueprint for their electrons, well, that's just neat. It’s a little chemical high-five across the periodic table.
So next time you see a salt shaker (which is basically sodium chloride, NaCl), remember that behind that seemingly simple compound are ions like Na⁺ and Cl⁻, each with their own story of electron-gaining or losing. And if you ever encounter an oxide (like in rust or even breathing!), that involves O²⁻, which, as we discovered, shares a special electron-hugging secret with our friendly sodium ion. It's a reminder that even in the smallest building blocks of our universe, there's a constant dance of seeking balance and sometimes, achieving perfect, identical harmony.
It’s all about achieving that sweet spot, that perfect configuration where the atom (or ion, in this case) feels complete and at peace. It’s the chemical equivalent of finding your favorite comfy chair after a long day. No more worries, no more reaching, just pure, blissful stability. And that, my friends, is the beauty of ions and their electron configurations.
