Identify The Unknown Isotope Xx In The Following Decays.

Hey there, science explorers and curious cats! Ever feel like you're playing a super-secret spy game, but with tiny, invisible particles instead of trench coats and dark alleys? Well, buckle up, because we're about to dive into the thrilling world of radioactive decay, where we get to be detectives and figure out the identity of a mysterious "Isotope Xx"! Think of it like a cosmic scavenger hunt, and we've got the clues!
So, imagine we have this super elusive character, our dear Isotope Xx. It's a bit of a drama queen, you see. It can't just sit still and be its stable, chill self. Nope, Isotope Xx loves to change! It undergoes these amazing transformations, called decays, where it spits out bits of itself and turns into something else entirely. It's like a caterpillar becoming a butterfly, but with way more "POOF!" and a sprinkle of energetic particles. Pretty neat, right?
Now, the universe, in its infinite wisdom (and sometimes mischief!), has a whole bunch of different ways for Isotope Xx to change. We've got the alpha decay, where it throws out a couple of protons and neutrons – like it's chucking a tiny helium nucleus at you! Then there's the beta decay, which is a bit more sneaky. It can either spit out an electron (a beta-minus decay, imagine a tiny negative firecracker) or capture one (a beta-plus decay, like it's giving a tiny positive hug to an electron). And let's not forget the gamma decay, where it just sheds some excess energy as a super-powerful light ray – think of it as a cosmic yawn that’s incredibly energetic!
Our mission, should we choose to accept it (and we totally do because it's awesome!), is to figure out exactly who Isotope Xx is. It’s like trying to guess a person's favorite ice cream flavor based on the sprinkles they leave behind. We're given a list of these transformations, these decays, and with each one, we get a tiny peek behind the curtain. It's like solving a really cool, atom-sized puzzle!
Let’s say we see our Isotope Xx doing its thing. It’s like, "Okay, I'm feeling a bit volatile today, so I’m going to do an alpha decay!" POOF! Out pops a little something, and our Isotope Xx turns into a new element. We look at what came out, and we can start piecing things together. It’s like saying, "Aha! If it threw out that specific bunch of protons and neutrons, then it must have been this element before!"
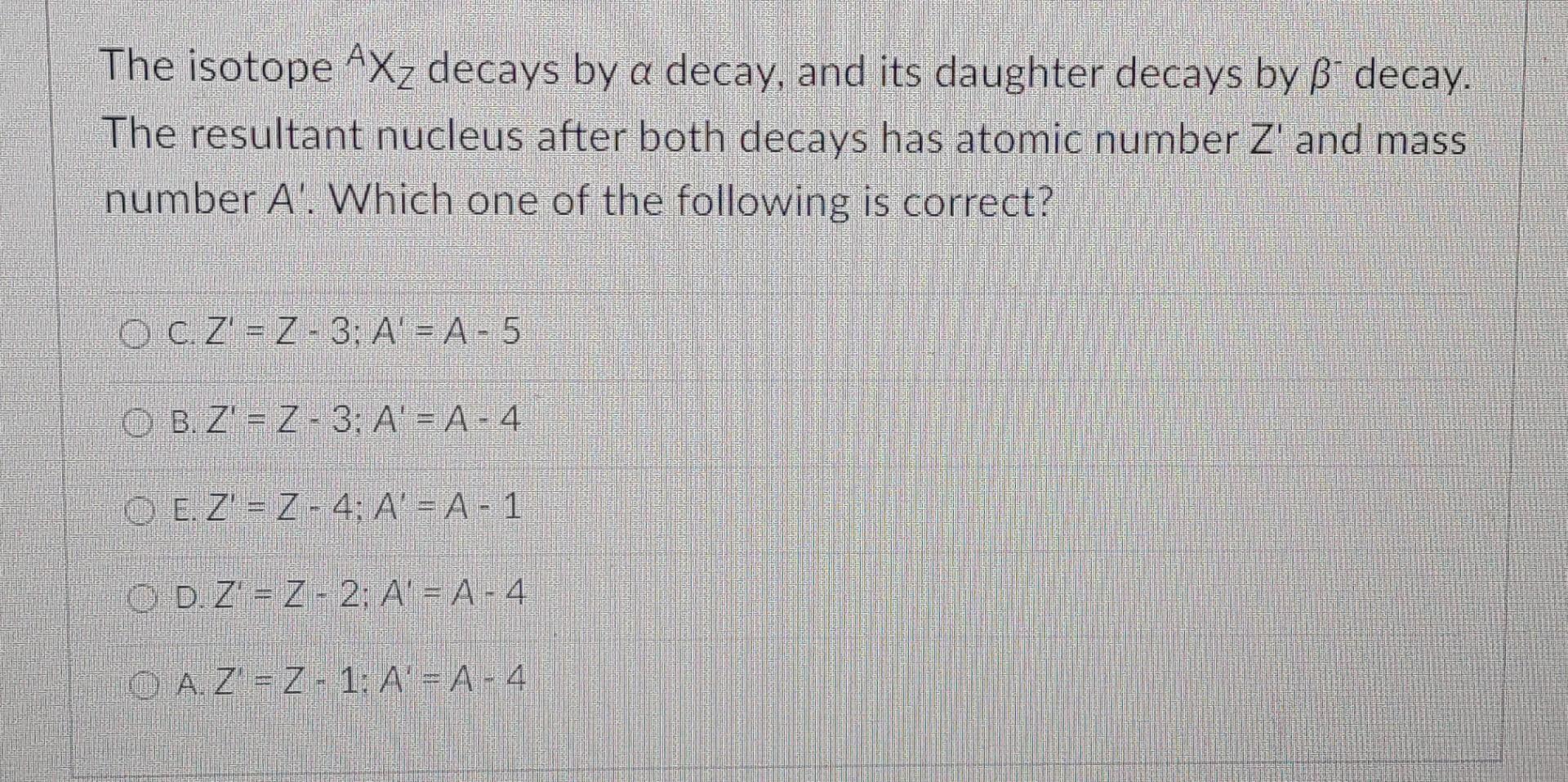
Then, maybe the next clue is that our, now slightly different, element decides to do a beta-minus decay. ZAP! Another particle flies off, and it transforms again. This is where it gets really exciting. Each decay tells us a story. It’s like having a secret code that reveals the identity of our mystery element. Imagine you're trying to identify a brand of candy. If you see red wrapping paper, it could be strawberry. If you then see it's chewy, it narrows it down. But if you see red wrapping paper, it's chewy, and it has a sour coating, well, you're pretty darn close to guessing that specific sour cherry gummy worm, aren't you?
So, in our case, when Isotope Xx undergoes these decays, it's leaving behind a trail of breadcrumbs made of protons, neutrons, and energy. Our job is to follow that trail backwards. We look at what the element became and what particles came out. Using our trusty knowledge of the periodic table (which is basically a cheat sheet for elements!), we can work backwards and say, "Okay, if it ended up as this element after spitting out that, then before it must have been this other element!" It’s like reverse engineering a magnificent, tiny, radioactive masterpiece.
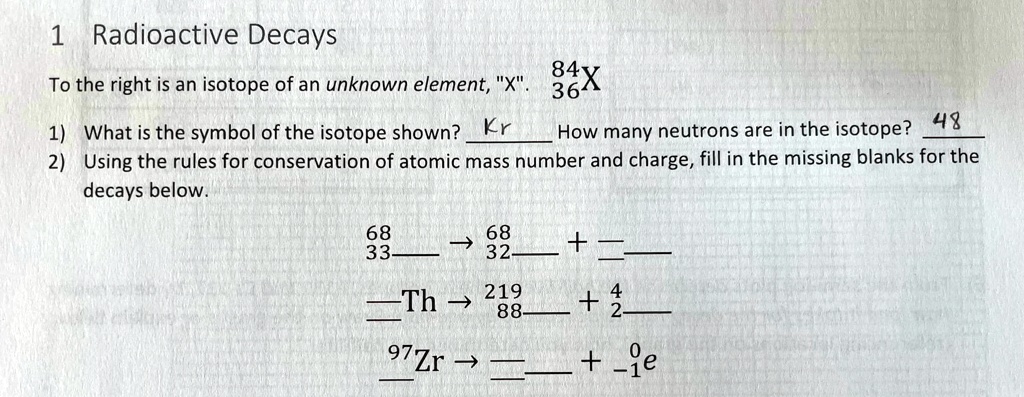
And sometimes, it's not just one step! Oh no, our Isotope Xx might go through a whole series of decays. It’s like a relay race where each element passes the baton to the next, getting a little closer to stability with each change. Our detective skills are put to the test as we decipher the sequence. We're looking at the initial state of Isotope Xx, and then the final state after a series of these transformations. It’s like seeing a messy room and trying to figure out who was the culprit, and what they did to make it so messy!
The beauty of it all is that these decays are governed by fundamental laws of nature. Protons and neutrons don't just magically appear or disappear. They are conserved! This is our superpower as isotope detectives. We know that the total number of protons and neutrons in the beginning has to equal the total number of protons and neutrons at the end, plus whatever was ejected. It’s like accounting, but with atomic particles!

So, when we're presented with a scenario where Isotope Xx undergoes a specific type of decay, we get to play the ultimate guessing game. We might see something like:
Isotope Xx → Element Y + alpha particleAnd our brains immediately go into overdrive! We know what an alpha particle is. We know what Element Y is (or we can look it up!). And with a little bit of atomic arithmetic, BAM! we can pinpoint the identity of our elusive Isotope Xx. It’s incredibly satisfying, like finally solving that one tricky Sudoku or finding the missing sock in the laundry!
So, the next time you hear about radioactive decay, don't think of it as scary or complicated. Think of it as a cosmic puzzle, a thrilling spy mission, and a fantastic opportunity for us to put on our detective hats and figure out the secret lives of atoms. It's all about observation, deduction, and a whole lot of scientific fun. Go forth, and may your isotopic investigations be ever victorious!
