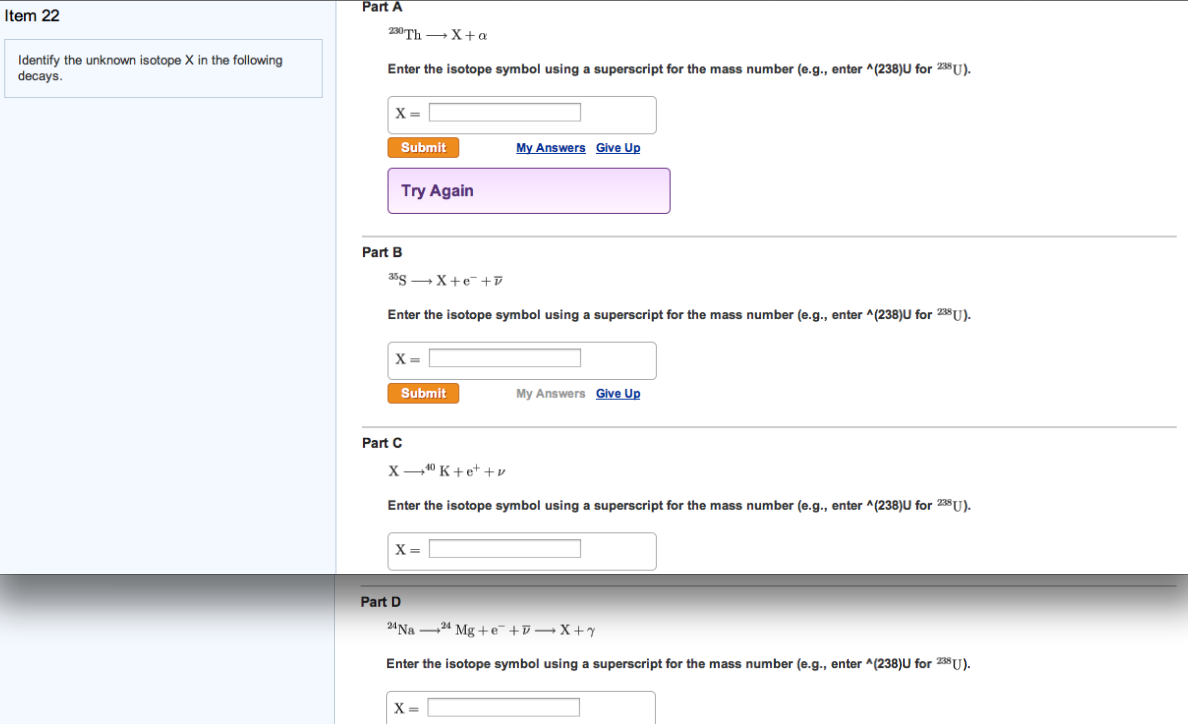
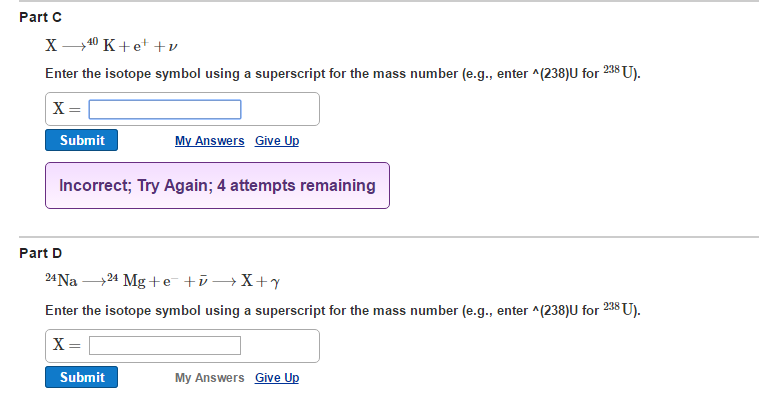
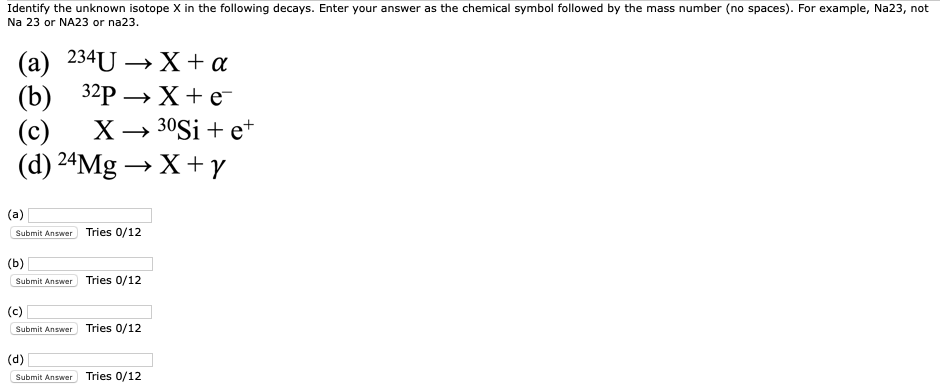
Identify The Unknown Isotope X In The Following Decays

Ever feel like you're staring at a cosmic mystery, a little puzzle that just won't click? Life, and by extension, the universe, often throws us these intriguing enigmas. Today, we're diving into one of those fascinating "whodunits," but instead of a stolen jewel, we're tracking down an unknown isotope. Think of it like being a detective for the atomic world, piecing together clues from the evidence left behind – the resulting particles and energy. It’s less about magnifying glasses and more about understanding the fundamental building blocks of everything around us. So, grab your favorite comfy beverage, maybe a cup of that artisan coffee you’ve been meaning to try, and let’s unravel the secrets of Isotope X!
Unlocking the Atomic Enigma: The Art of Isotope Sleuthing
Now, before you start picturing mad scientists in bubbling labs (though there's a certain charm to that image!), let's demystify what we're dealing with. Isotopes are essentially variations of the same element. Imagine a family: they all share the same last name (the element's atomic number, determined by the number of protons), but they might have slightly different characteristics – in this case, a different number of neutrons. This difference in neutrons affects their atomic mass and, crucially, their stability. Some isotopes are perfectly happy, chilling out for eons, while others are a bit more… energetic. They undergo radioactive decay, transforming into something else and releasing particles and energy along the way. It's nature's way of finding balance, like tidying up your digital clutter or finally organizing that overflowing closet. We're going to be looking at these transformations, these decays, to figure out what our mysterious Isotope X was hiding.
The Clues: What the Decays Tell Us
In the grand scheme of things, radioactive decay is a pretty reliable process. It follows predictable rules, much like the laws of grammar in a well-written novel or the rules of your favorite board game. When an unstable nucleus decides to shed some excess energy, it can do so in a few primary ways. We’ve got alpha decay, where the nucleus ejects a particle made of two protons and two neutrons (essentially a helium nucleus – cute, right?). Then there's beta decay. This one's a bit more nuanced. In beta-minus decay, a neutron transforms into a proton, emitting an electron (the beta particle) and an antineutrino. In beta-plus decay, a proton becomes a neutron, releasing a positron (the antiparticle of an electron) and a neutrino. Finally, there's gamma decay, which is more about releasing excess energy in the form of high-energy photons, like a cosmic sigh of relief.
Each of these decay modes comes with a specific signature, a fingerprint that helps us identify the parent and daughter nuclei. It's like a witness description: "It was wearing a proton, an electron, and a lot of energy!" By analyzing the particles emitted and the resulting nucleus, we can work backward, like a seasoned detective reconstructing a crime scene, to identify the original suspect – our Isotope X.
Case File 1: The Alpha Trail
Let's imagine our first clue. We observe a nucleus undergoing alpha decay. This means our unknown Isotope X had a certain atomic number and mass. When it emits an alpha particle (which has an atomic number of 2 and a mass number of 4), the resulting daughter nucleus will have an atomic number two less than Isotope X and a mass number four less than Isotope X. Think of it like shedding a small, dense backpack. You’re lighter and have less stuff.
So, if the daughter isotope we detect is, say, Helium-4 (which has 2 protons and 2 neutrons), and we know it came from an alpha decay, we can deduce that Isotope X was a nucleus with 2 more protons and 4 more nucleons (protons + neutrons) than Helium-4. Helium-4 itself is pretty stable, a bit like that reliably chill friend who’s always there. But its parents in the radioactive world are often quite the characters!
Practical Tip: When you're trying to identify something unknown in your own life, whether it's a mysterious ingredient in a recipe or a strange noise your car is making, try to isolate the variables. What's the most obvious change? What's the most significant difference? Focus on those key indicators, just like we focus on the atomic number and mass number in decay.
Case File 2: The Beta Ripple
Now, let's consider a scenario involving beta-minus decay. This is where things get really interesting. A neutron in the nucleus transforms into a proton and an electron (plus an antineutrino). What does this mean for our isotope? The number of protons increases by one, meaning the atomic number goes up by one. However, since a neutron is essentially replaced by a proton (both having a mass number of roughly 1), the mass number remains the same. It’s like trading in a well-worn notebook for a slightly newer, but equally thick, one. The content might change a bit, but the overall volume stays consistent.
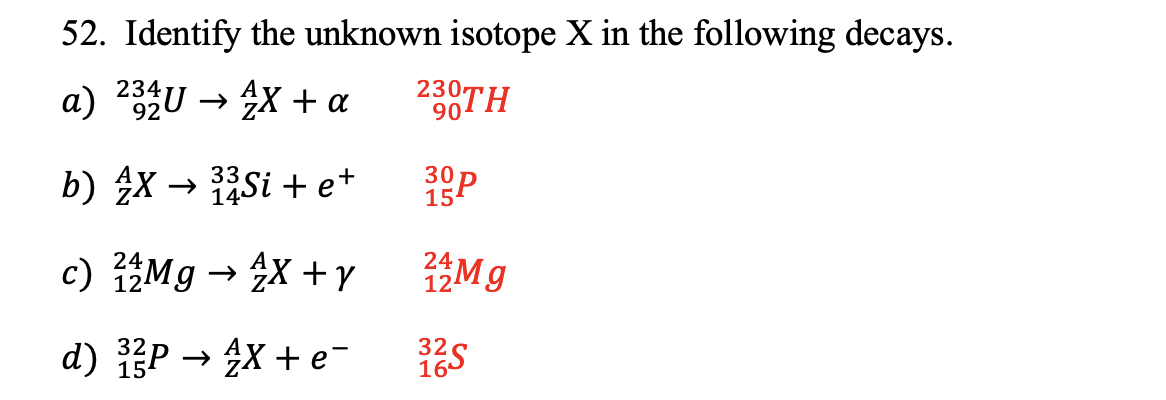
So, if we see a decay where the daughter isotope has one more proton than the parent, and the mass number is unchanged, we're likely looking at a beta-minus decay. This is a common pathway for elements that have too many neutrons to be stable. They’re essentially trying to get rid of that neutron surplus by converting it into a more stable proton. It’s a bit like decluttering your digital photo album – you find some blurry shots (neutrons) and convert them into something more presentable (protons).
Cultural Reference: Think of it like a good remix in music. The original track (the parent isotope) gets transformed, with some elements changing (neutron to proton), but the overall feel or "length" of the song (mass number) might stay the same, creating something new and interesting (the daughter isotope).
Conversely, beta-plus decay involves a proton transforming into a neutron, emitting a positron and a neutrino. In this case, the atomic number decreases by one, while the mass number, again, remains the same. This is typical for isotopes that have too many protons relative to their neutrons. They're nudging themselves towards a more balanced state.
Fun Fact: Positrons, the particles emitted in beta-plus decay, are the antimatter counterparts to electrons. When a positron meets an electron, they annihilate each other in a burst of energy – a little cosmic fireworks show!
Case File 3: The Gamma Glow
Sometimes, the primary emission isn't a particle, but a packet of energy. This is gamma decay. In this process, a nucleus is already in an excited state (perhaps after undergoing alpha or beta decay) and it releases this excess energy as a high-energy photon, a gamma ray. The crucial point here is that neither the atomic number nor the mass number changes. It's like a person taking a deep breath and relaxing after a stressful event. They don't fundamentally change, they just shed some of that pent-up tension.
So, if you observe a decay where the parent and daughter isotopes have the exact same atomic and mass numbers, but there's a significant release of energy, you're likely witnessing gamma decay. It's often a secondary event, a finishing touch, as the nucleus settles into its most stable configuration.
Practical Tip: In your own life, when you feel overwhelmed, consider what your "gamma decay" moment might be. Is it a good book, a walk in nature, or a chat with a friend? These are the ways we shed our own "excited states" and find a sense of calm and balance without fundamentally altering who we are.
Putting It All Together: Identifying Our Mysterious Isotope X
Now, let's apply these detective skills to our specific scenarios. We’re going to look at a couple of hypothetical decay chains to pinpoint our elusive Isotope X. Remember, we’re working backward from the knowns to find the unknown.
Scenario A: The Alpha and Beta Double Play
Let's say we observe an isotope, which we'll call Daughter A, undergoing beta-minus decay to become Daughter B. Daughter B is identified as Carbon-14 (atomic number 6, mass number 14). We know that beta-minus decay increases the atomic number by 1 and keeps the mass number the same.
Working backward from Daughter B (Carbon-14) to Daughter A: * Since Daughter B has an atomic number of 6, and it was formed by beta-minus decay, Daughter A must have had an atomic number of 6 - 1 = 5. * Since the mass number remains the same, Daughter A also had a mass number of 14. * Therefore, Daughter A is Boron-14 (atomic number 5, mass number 14).
Now, let's say we observed our original unknown, Isotope X, undergoing alpha decay to become Daughter A (Boron-14).
Working backward from Daughter A (Boron-14) to Isotope X: * Since Daughter A has an atomic number of 5, and it was formed by alpha decay (which decreases atomic number by 2), Isotope X must have had an atomic number of 5 + 2 = 7. * Since Daughter A has a mass number of 14, and alpha decay decreases the mass number by 4, Isotope X must have had a mass number of 14 + 4 = 18. * An element with an atomic number of 7 is Nitrogen. * Therefore, Isotope X is Nitrogen-18.
Fun Fact: Nitrogen is crucial for life on Earth, making up about 78% of our atmosphere. It's in the proteins, DNA, and RNA that build us all!
Scenario B: A More Complex Transformation
Let's try another one. Suppose Isotope X undergoes beta-plus decay to form an intermediate isotope, let's call it Intermediate Y. Intermediate Y then undergoes gamma decay and remains unchanged. Finally, Intermediate Y undergoes alpha decay to form Daughter C, which is identified as Oxygen-16 (atomic number 8, mass number 16).
Let's break this down, starting from the known daughter isotope and working backward:
1. Intermediate Y to Daughter C (Oxygen-16): We know Daughter C is Oxygen-16. It was formed by the alpha decay of Intermediate Y. * Since Daughter C has an atomic number of 8 and alpha decay decreases it by 2, Intermediate Y had an atomic number of 8 + 2 = 10. * Since Daughter C has a mass number of 16 and alpha decay decreases it by 4, Intermediate Y had a mass number of 16 + 4 = 20. * An element with an atomic number of 10 is Neon. * Therefore, Intermediate Y is Neon-20.
2. Intermediate Y (Neon-20) and Gamma Decay: We are told Intermediate Y undergoes gamma decay and remains unchanged. This confirms that gamma decay doesn't alter the atomic or mass number, so it doesn't help us pinpoint Intermediate Y's identity beyond what we already deduced. It just means Neon-20 was in an excited state and shed some energy.
3. Isotope X to Intermediate Y (Neon-20): We know Isotope X underwent beta-plus decay to form Intermediate Y (Neon-20). Beta-plus decay decreases the atomic number by 1 and keeps the mass number the same.
Working backward from Intermediate Y (Neon-20) to Isotope X: * Since Intermediate Y has an atomic number of 10, and it was formed by beta-plus decay, Isotope X must have had an atomic number of 10 + 1 = 11. * Since the mass number remains the same, Isotope X also had a mass number of 20. * An element with an atomic number of 11 is Sodium. * Therefore, Isotope X is Sodium-20.
Cultural Reference: Think of these decay chains like a family tree, but with a twist. Each generation is a new element, transforming and evolving. It's a constant process of becoming, a cosmic evolution happening at the atomic level.
The Takeaway: Beyond the Atom
So, what does this atomic detective work have to do with our everyday lives? It’s all about understanding patterns and the fundamental laws that govern the universe, even if those laws are expressed in the language of protons, neutrons, and electrons. In our own lives, we're constantly identifying unknowns, whether it's deciphering a confusing email, understanding a complex problem at work, or even figuring out what your pet is trying to tell you.
Just like these isotopes transform to reach a more stable state, we too are in a constant process of growth and change. We learn from our experiences, adapt to new situations, and shed old habits that no longer serve us. The universe, in its infinite wisdom, is always seeking balance, and so are we, in our own unique ways. The next time you encounter a puzzle, big or small, remember the principles of isotope sleuthing: observe the evidence, understand the underlying rules, and work backward with a curious and persistent mind. You might just uncover something fascinating – perhaps even your own inner Isotope X, ready to reveal its true nature.
