Identify The Types Of Intermolecular Forces Present In C6h14
So, let's talk about something super chill. We're diving into the tiny world of molecules. Specifically, C6H14. Sounds fancy, right? But don't sweat it. It's just a bunch of carbon and hydrogen atoms hanging out. Think of it like a little molecular party!
And just like any good party, there's a whole lot of interaction going on. Not the awkward "what do I say now?" kind of interaction. More like the gentle nudges and magnetic pulls between these little dudes. These are called intermolecular forces. Super important stuff, even if it sounds a bit like jargon.
Imagine you've got a bunch of tiny, slightly sticky balloons. They're not super sticky, just enough to kind of drift towards each other. That's kind of what happens with molecules. They have these invisible attractions. And for C6H14, it's a really fun mix of these forces.
So, what exactly is C6H14? Well, it's an alkane. Fancy word again, I know. Basically, it means it's made of only carbon and hydrogen, and all the bonds are single. No double or triple bonds to get all wild. It's a pretty straightforward molecule.
Think of it like a straight chain of carbon atoms, with hydrogen atoms all over it, like little fuzzy friends. Or, it could be a bit branched. Like a tiny molecular dendrite. It can exist in a few different forms, called isomers. But no matter the shape, the intermolecular forces are the same. It's all about how those atoms are arranged and how they interact.
The Main Players in the Molecular Mingle
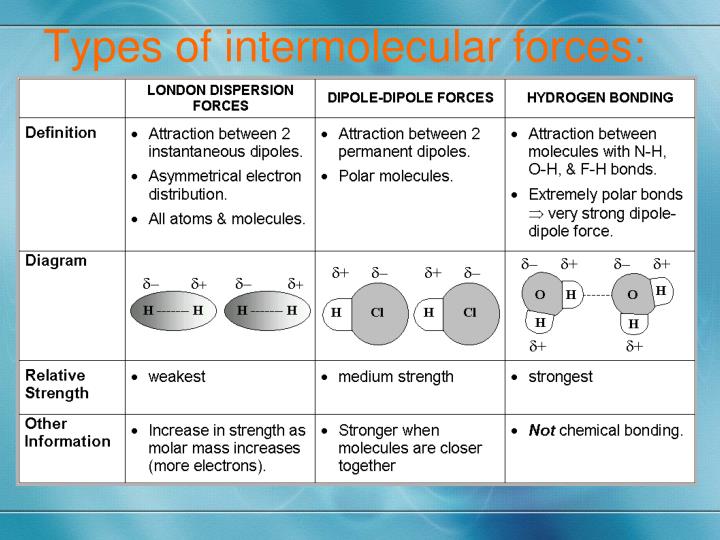
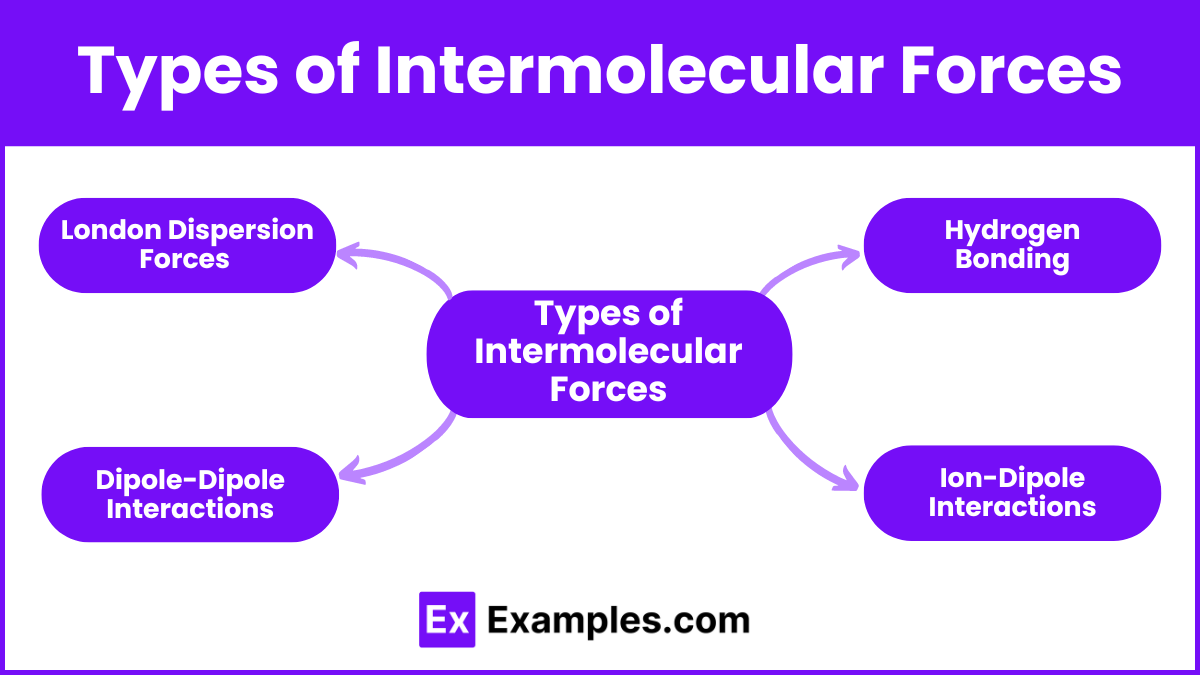
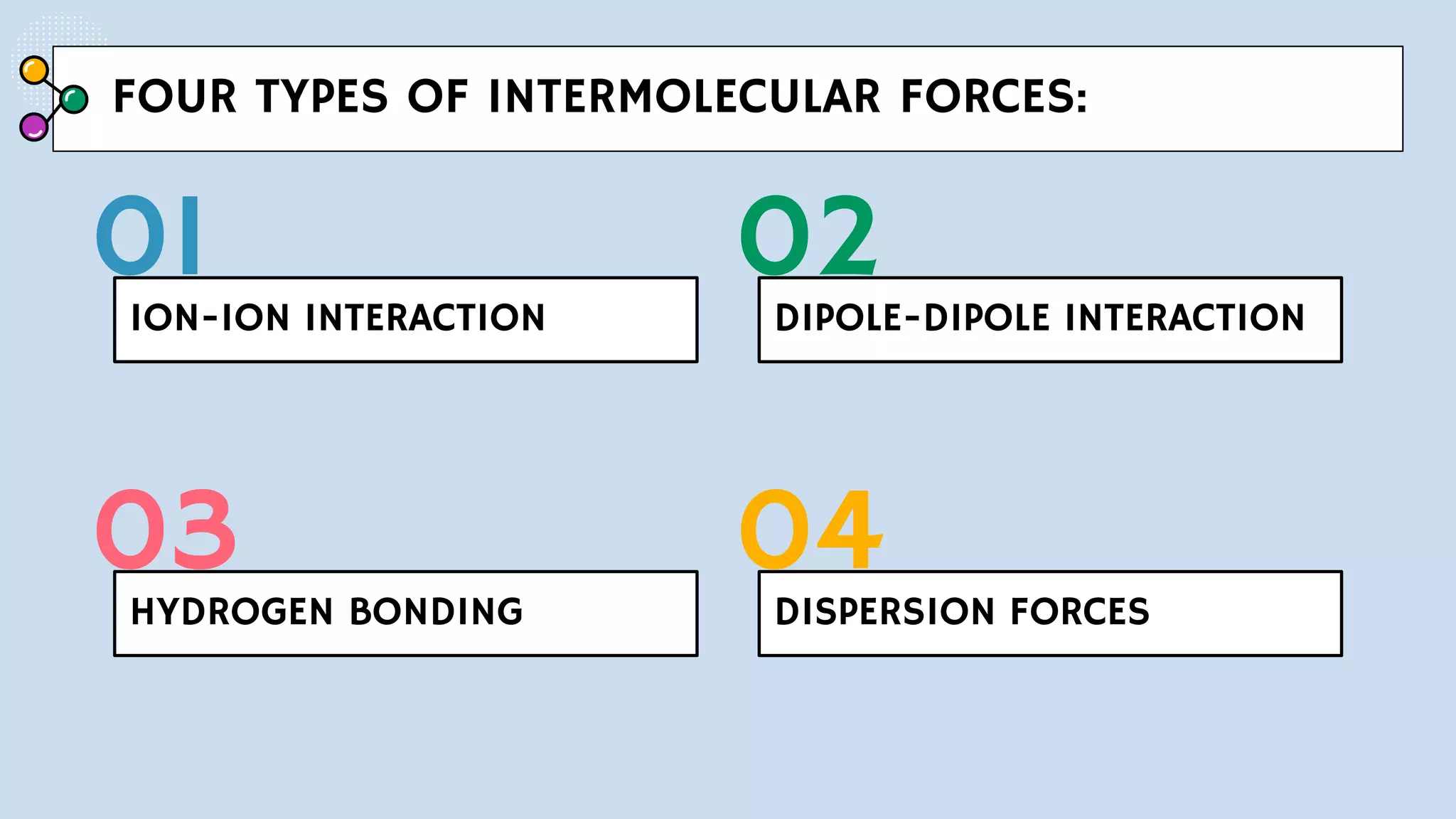
Now, for the good stuff! What forces are actually making these C6H14 molecules want to hang out? We've got a few stars in this show. And the biggest, most prominent one? Drumroll please... London dispersion forces!

Sounds like something out of a sci-fi movie, right? "Beware the London dispersion forces!" But in reality, they're the most common type of intermolecular force. And they're kind of, well, accidental.
Here's the quirky fact: molecules aren't perfectly symmetrical all the time. Even nonpolar molecules like C6H14 have tiny, temporary fluctuations in their electron clouds. Imagine electrons zipping around. Sometimes, just by chance, a few more electrons might be on one side of the molecule than the other. This creates a tiny, temporary imbalance of charge. A little "electron oopsie!"
This temporary imbalance in one molecule can then nudge the electrons in a neighboring molecule. It's like a domino effect of electron wobbles! This induced dipole then attracts the positive side of the first molecule. And voila! You've got a weak attraction. London dispersion forces are all about these temporary, induced dipoles. Super subtle, but super important.
And for C6H14, which is a relatively large molecule with lots of electrons, these London dispersion forces are actually quite significant. More electrons mean more chances for these tiny wobbles to happen. It's like having a bigger party with more people, so there are more chances for spontaneous interactions. That's why C6H14, even though it's nonpolar, has a liquid state at room temperature. These forces are strong enough to keep the molecules from just flying off into space as a gas.
Why are London Dispersion Forces Fun?
They're fun because they're so universal! Every molecule has them. Even the most polar molecules, which have stronger forces, still have London dispersion forces going on. It's like the background music to all molecular interactions. And the fact that they arise from such random electron movement is just... neat! It’s a beautiful example of how order can emerge from apparent chaos at the molecular level.
Think about it: the whole universe of molecular attractions is partly based on tiny, accidental electron hiccups. How cool is that? It's a reminder that even the most fundamental forces can have a bit of whimsy to them.
Are There Any Other Forces at Play?
Now, here’s where things get really interesting for C6H14. Since it's an alkane, it's considered nonpolar. This is a crucial detail. Nonpolar molecules don't have permanent positive and negative ends. Unlike, say, water, which has a strong positive and negative side (making it polar and able to form hydrogen bonds).

Because C6H14 is nonpolar, it doesn't have the stronger types of intermolecular forces. We're talking about dipole-dipole forces and hydrogen bonding. Nope, not here. These require permanent dipoles, which our happy little C6H14 molecules just don't have.
So, the story for C6H14 is largely about the power of London dispersion forces. They are the sole, or at least the overwhelmingly dominant, intermolecular force. It's like a party where everyone is really good at just chatting and enjoying each other's company, without any intense romantic entanglements or dramatic arguments. Just a chill, friendly vibe.
This lack of stronger forces is why things like hexane (which is a type of C6H14) are often used as solvents for nonpolar substances. They can dissolve greasy things because they have similar intermolecular forces. It's a bit of a "like dissolves like" situation. Polar solvents dissolve polar things, and nonpolar solvents dissolve nonpolar things.
The Simplicity is the Charm
Some might say, "Oh, it's just London dispersion forces. That's boring!" But I disagree! The beauty of C6H14's intermolecular forces lies in their elegant simplicity. It highlights how even weak, temporary attractions can collectively create observable properties.
It's like a massive orchestra playing a single, beautiful note for a very long time. It might not have the complex harmonies of other pieces, but its purity and resonance are captivating. The strength of London dispersion forces in C6H14 is a testament to the sheer number of interactions happening. Billions upon billions of molecules, all subtly influencing each other.
And the fact that this molecule is so common! Hexane is in gasoline, it's used in labs, it's all around us. Understanding its intermolecular forces helps us understand why it behaves the way it does. Why it’s a liquid, why it dissolves certain things, and why it's so useful.
So, next time you hear about C6H14, don't just think of a chemical formula. Think of a molecular party, powered by the charmingly accidental, yet surprisingly powerful, London dispersion forces. It’s a little peek into the hidden, playful world of chemistry, where even the smallest fluctuations can have big consequences.
It’s a reminder that science isn’t always about giant equations and complex theories. Sometimes, it's about understanding the gentle nudges and magnetic whispers between the tiniest particles that make up our world. And for C6H14, those whispers are all about London dispersion forces. Pretty cool, right?
