Identify The True Statements Regarding Disulfide Bridges Disulfide Bonds

Hey there, science explorers! Ever wonder what keeps those tiny building blocks of life, called proteins, in their super cool shapes? Well, let me tell you about a little something called a disulfide bridge. It's like a secret handshake for certain proteins, a way they stick together and stay in line.
Think of proteins as long, wiggly chains. To do their jobs properly, these chains need to fold up into very specific, often complex, 3D structures. It’s not just about being a long string; it’s about how that string twists and turns.
And that's where our star player, the disulfide bridge, comes in! These bridges are formed between two specific parts of a protein chain, usually involving a sulfur atom. It’s like a tiny, strong bond that helps lock certain parts of the protein in place.
Imagine you have a piece of string that you want to tie into a fancy knot. You might use a little piece of thread to hold the knot tight. A disulfide bridge acts a lot like that thread, providing extra stability to the protein's folded form.
So, what makes these disulfide bridges so special? They are incredibly important for the stability and function of many proteins. Without them, some proteins would just fall apart, like a deflated balloon!
Let's dive into some of the true statements about these fascinating little connections. Get ready to be amazed by how these tiny bonds do such big jobs in the world of biology!
The Mighty Bond: What's the Deal?
First off, a disulfide bridge is a type of covalent bond. This means it’s a strong, shared connection between atoms, not just a weak attraction. It’s a serious commitment, you could say!
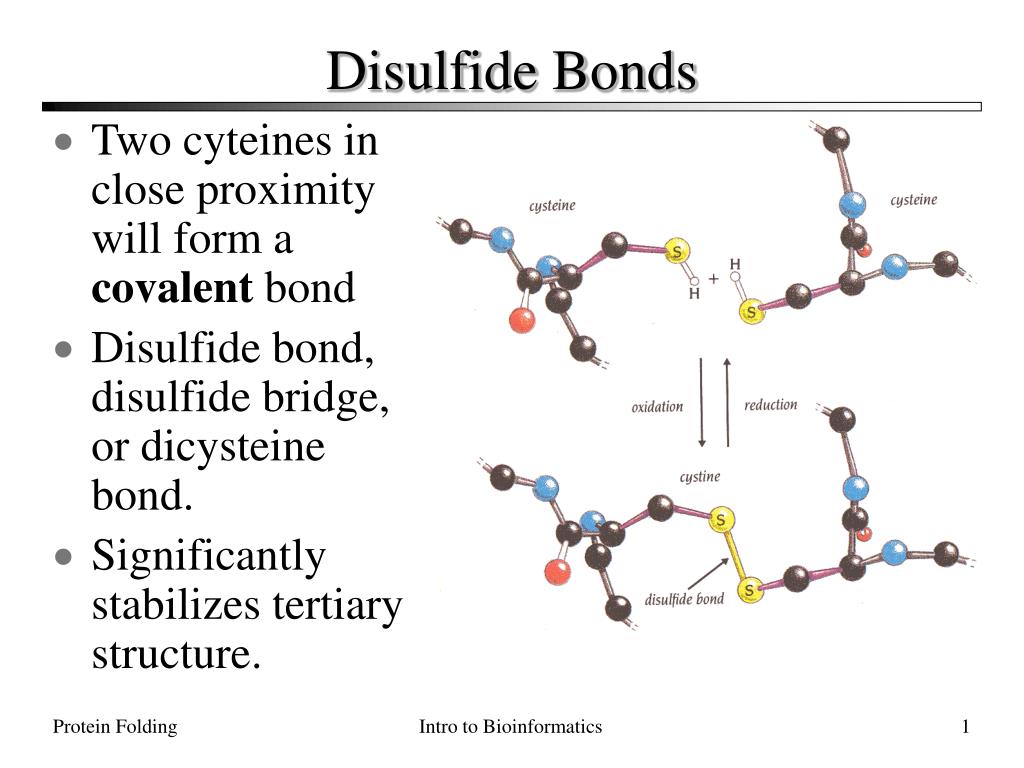
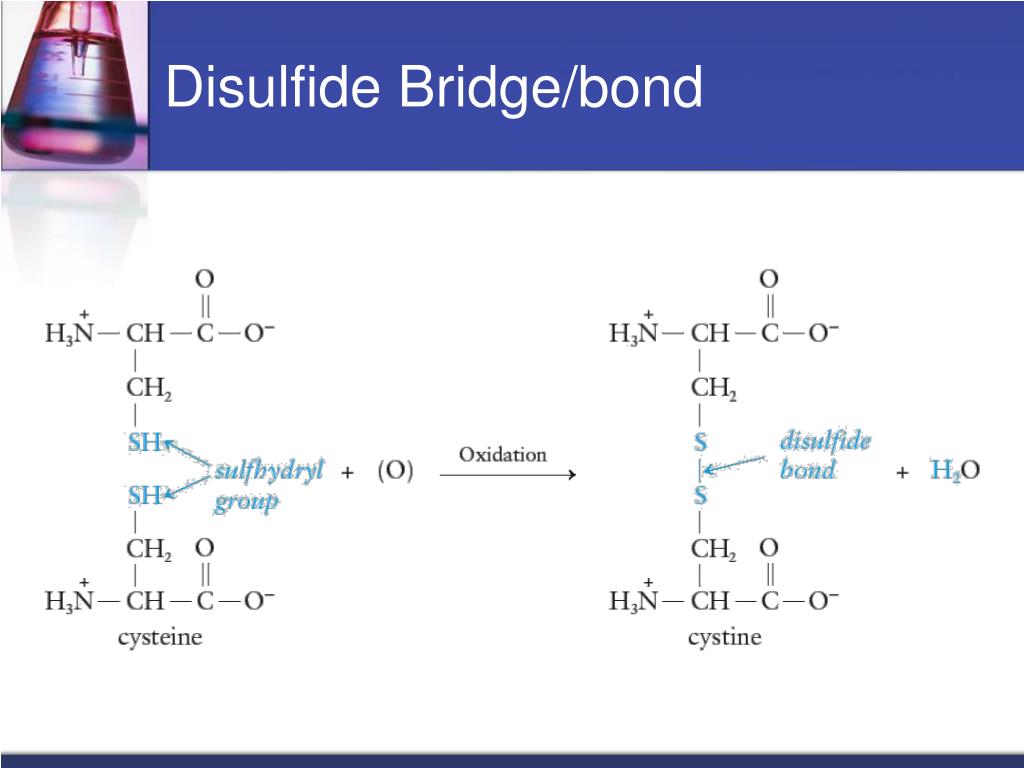
These bridges are specifically formed between the sulfur atoms of two cysteine amino acid residues. Cysteine is a special building block in the protein world, and when two of them get close enough and the conditions are right, they can form this super bond.
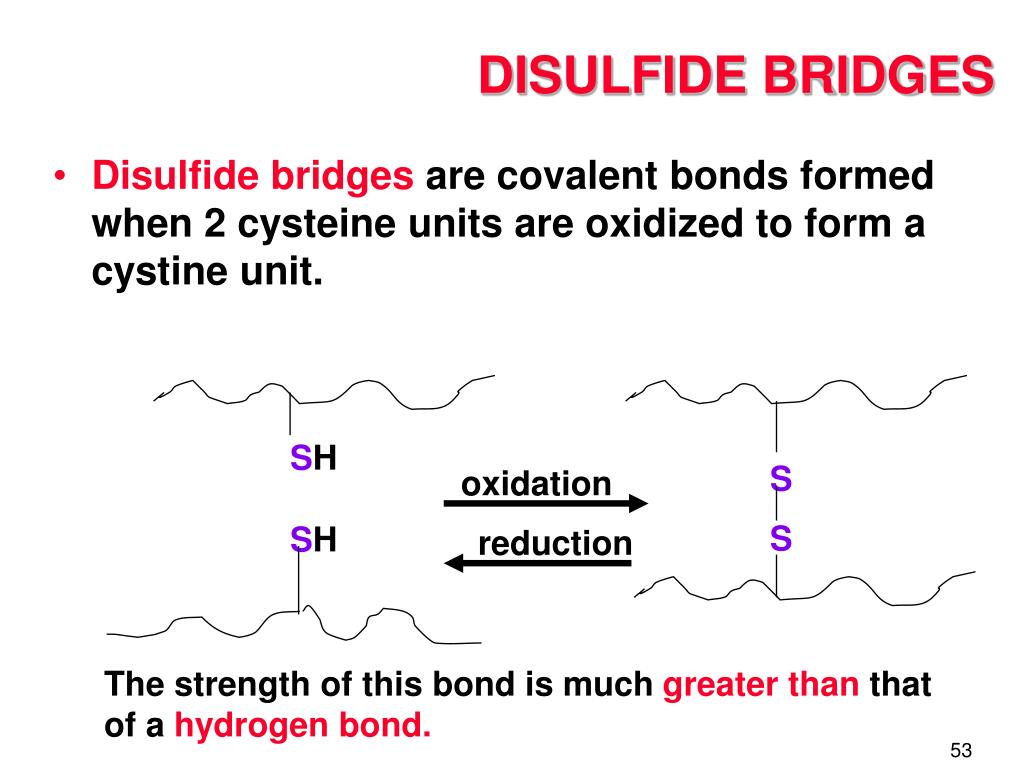
The formation of a disulfide bridge is called oxidation. It’s like a chemical reaction where the two sulfur atoms essentially "pair up" and share electrons, creating that strong link. This process usually happens in certain cellular environments.
And here’s a fun fact: breaking a disulfide bridge usually requires a reduction process. So, it's a bit like a reversible lock and key. You can lock it with oxidation, and unlock it with reduction.
These bridges are not found in all proteins. They are particularly common in proteins that are found outside of the cell, in environments that might be a bit harsher. Think of it as extra armor for proteins working in tough conditions.
Proteins That Love Their Bridges
Many of the proteins you've probably heard of, like antibodies, rely heavily on disulfide bridges. Antibodies are the tiny soldiers that fight off invaders in your body, and they need to be super stable to do their job.
Proteins that travel outside the cell, like those in your skin or your digestive system, often have these bridges. This is because the environment outside the cell can be very different from the inside, and the bridges help the proteins survive and keep their shape.
Think about your skin! The structural proteins in your skin, like keratin (that's what your hair and nails are made of too!), are packed with disulfide bridges. They give your skin its strength and resilience. Pretty neat, huh?
Even some enzymes, the workhorses of many biological reactions, use disulfide bridges to maintain their precise shapes. If an enzyme's shape is wrong, it can't do its job effectively, and that can cause all sorts of problems.
It’s not just about holding things together; disulfide bridges can also play a role in how proteins interact with each other. They can be part of the "puzzle pieces" that allow different proteins to recognize and bind to one another.
The Coolest Part: Function and Stability
The main superpower of disulfide bridges is providing structural stability. They act like internal scaffolding, preventing the protein from unfolding too easily, especially when exposed to heat or changes in pH.
This stability is crucial for a protein’s biological activity. If a protein’s shape is compromised, it often loses its ability to perform its specific function, which can be anything from carrying oxygen to signaling between cells.
Consider this: some proteins are folded so intricately that without their disulfide bridges, they would be like a tangled mess of yarn. The bridges are what keep that beautiful, functional structure intact.
You might have heard of certain diseases linked to protein misfolding. In many cases, the inability to form or maintain proper disulfide bridges can be a contributing factor to these issues. It highlights just how vital these bonds are.
So, the next time you think about the complex world inside your body, remember the unsung heroes: the disulfide bridges. They are the quiet guardians of protein shape, ensuring everything runs smoothly and efficiently.
A Little Twist: Where They Are NOT Found
It's equally important to know where you won't typically find these bridges. Inside the cell, in the main watery environment called the cytoplasm, the conditions are generally reducing. This means that disulfide bridges are much less common there.
Why is this the case? Well, if disulfide bridges were constantly forming and breaking in the cytoplasm, it would be chaotic! Proteins in the cytoplasm often have different ways of maintaining their structure, relying on other types of interactions.
The endoplasmic reticulum, an organelle inside the cell, is a bit of an exception. It's where many proteins destined for secretion or insertion into membranes are processed, and it's a more oxidizing environment where disulfide bonds can form. So, there are special zones for these bonds!
This difference in where they are found tells us a lot about the environments different proteins operate in. It’s a clever biological strategy to ensure proteins are stable where they need to be.
So, while they are essential in many places, they are not everywhere. This selective presence makes their role even more fascinating.

The Grand Finale: Why Should You Care?
Understanding disulfide bridges is like unlocking a secret level in the game of biology. It helps us appreciate the incredible complexity and elegance of life at its most fundamental level.
These tiny bonds are crucial for everything from how your immune system works to the strength of your hair. They are a testament to the power of chemistry in making life possible.
If you're curious about how your body functions, or even how medicines are designed to target specific proteins, learning about these bonds is a fantastic starting point. It’s a peek behind the curtain of life!
So, the next time you see something about proteins and their shapes, you'll know about the superhero bond that often holds them together. It's a small thing, but it makes a world of difference!
Keep exploring, keep asking questions, and you might just discover more amazing biological secrets like the mighty disulfide bridge! It’s a wild and wonderful world out there.
Disulfide bridges are truly one of nature's clever solutions for protein stability!
