Identify The Sugar Carbons In This Figure Of Ssdna.

So, picture this: I'm hunched over my laptop, probably fueled by questionable amounts of caffeine and an even more questionable diet, trying to make sense of this ridiculously complicated diagram. It’s of, you guessed it, ssDNA. Single-stranded DNA. Sounds simple enough, right? Well, it’s not. Not when you’re trying to pinpoint specific atoms in a molecule that looks like a tangled ball of yarn after a cat has had its way with it. My eyes were starting to cross, and I swear I could hear the letters on the screen start to… well, dance. And there I was, staring at this ssDNA figure, thinking, “Okay, brain, what exactly are we looking for here?”
Then it hit me, like a rogue ribosome in a protein-folding factory. The prompt! Identify the sugar carbons. Ah. Of course. Because while the whole DNA thing is a marvel of biological engineering, it’s built from these fundamental building blocks, and the sugars are, well, pretty darn important. They form the backbone, you know? The stuff that holds the whole darn thing together. So, forget the wild goose chase of identifying every atom. Let’s zoom in on these sugary bits. Because understanding the sugar carbons in ssDNA is like understanding the foundation of a skyscraper. You can’t appreciate the dazzling architecture without knowing what’s holding it up, can you?
And honestly, sometimes the most complex things are best understood by breaking them down into their most basic, sometimes surprisingly sweet, components. Speaking of sweet… that’s where the sugar part comes in. Get it? Sugar? DNA? Okay, okay, I'll stop with the bad puns. Probably. But seriously, these aren’t the kind of sugars you’d find in a cookie, much as I’d like to believe that sometimes. These are deoxyribose sugars, and they’re the unsung heroes of our genetic code.
The Backbone of the Beast: Deoxyribose Revealed!
So, let’s get down to brass tacks, or rather, carbon atoms. We’re looking at a single strand of DNA, and within this strand, the sugars are like the beads on a very, very long necklace. Each nucleotide – that’s the basic unit of DNA – has a sugar molecule. And it’s these sugars that link up to form the famous DNA backbone. Pretty neat, huh?
In our ssDNA figure, you’ll see these repeating ring structures. Those are your deoxyribose sugars. They’re five-membered rings, meaning they have four carbon atoms and one oxygen atom making up the ring itself. But here’s the kicker: the carbons are where all the action happens. They’re the attachment points for everything else. The phosphate groups, the nitrogenous bases… it’s all about these carbons!
Now, to be precise, we're talking about deoxyribose sugar. The “deoxy” part is a hint that it’s a modified sugar. Specifically, it’s missing an oxygen atom compared to ribose sugar (which you find in RNA). This tiny difference is actually a big deal for DNA’s stability. Less reactive, you see. More robust. Perfect for storing all our precious genetic information long-term. So, next time you’re thinking about DNA, remember it’s not just a double helix; it’s a carefully designed molecule, and those sugars play a crucial role in its elegance and durability.
Let’s Talk Carbon Numbers: The Key Identifiers
Alright, time to get a bit more technical, but don’t worry, I’ll hold your hand through this. Each carbon atom in the deoxyribose ring is numbered. This numbering system is super important for chemists and biologists because it tells us exactly where things are attached. Think of it like street addresses for atoms. Without these numbers, it would be utter chaos.
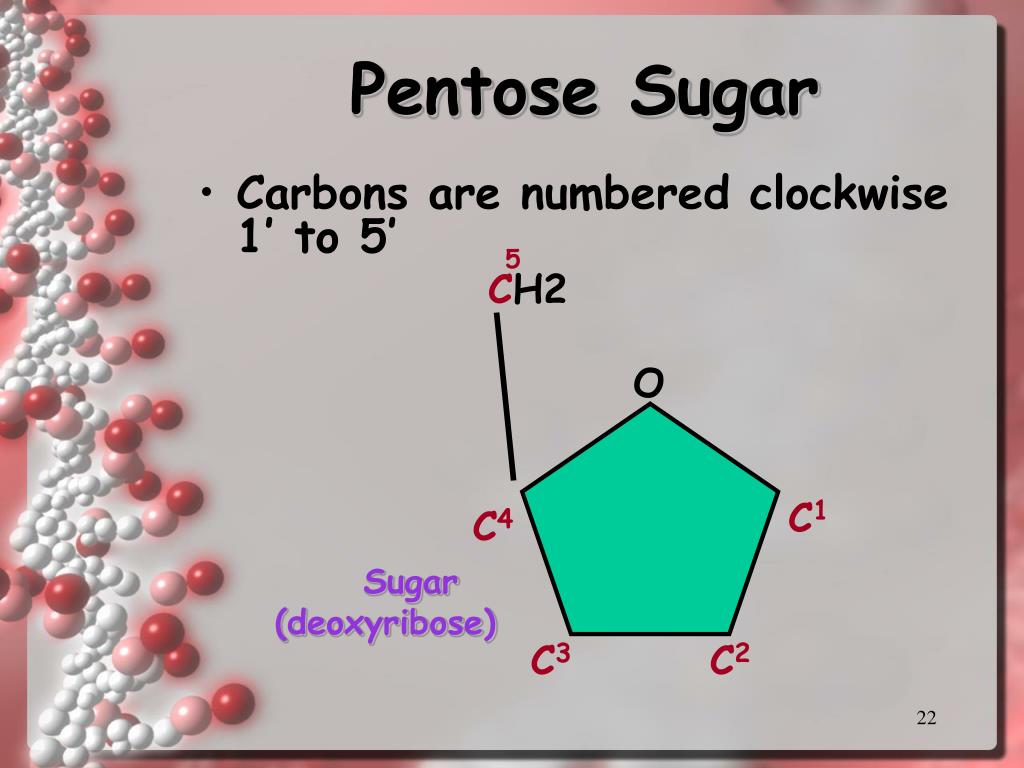
So, in the deoxyribose sugar ring, we have carbons labeled 1’ (pronounced “one prime”) through 5’. The prime symbol is there to distinguish these carbons from the carbons in the nitrogenous base, which also have numbers. See? Even the numbering has its own rules! It’s like a secret code we’re trying to crack.
Let’s break them down:
The 1’ Carbon: This is the carbon that’s directly attached to the nitrogenous base (A, T, C, or G). This is where the genetic information really gets “written,” in a way. It’s the link between the sugar and the code itself. Imagine this carbon is the podium where the base stands to deliver its message.
The 2’ Carbon: This is where our deoxyribose differs from regular ribose. Remember how I said it’s “deoxy”? Well, at the 2’ position, deoxyribose has a hydrogen atom (H) instead of a hydroxyl group (-OH). This is the “missing” oxygen. This seemingly small difference makes DNA much more stable, which is crucial for its role as the long-term archive of our genetic instructions.

The 3’ Carbon: Ah, the 3’ carbon. This one is super important for building the DNA strand. It's where the next nucleotide will attach, via its phosphate group. The phosphodiester bond, as the fancy folks call it, forms between the phosphate on the 5’ carbon of one nucleotide and the 3’ carbon of the previous nucleotide. It’s the glue that holds the chain together, and this 3’ carbon is the critical docking station for that bond.
The 4’ Carbon: This carbon is part of the ring structure and is generally involved in holding the ring together. It’s kind of the structural support for the sugar molecule itself. Think of it as one of the main pillars in our skyscraper analogy.
The 5’ Carbon: This carbon is outside the ring. It’s attached to the ring at the 4’ carbon, and crucially, it’s where the phosphate group is attached. This phosphate group is what will link to the 3’ carbon of the next nucleotide. So, the 5’ carbon is essential for the linkage that forms the DNA backbone, extending the chain in the 5’ to 3’ direction.
Navigating the Figure: What to Look For
Now, let’s translate this knowledge to your figure of ssDNA. When you look at the diagram, you’ll see these pentagonal (five-sided) shapes. These are the deoxyribose sugars. They’re usually depicted with the oxygen atom forming one corner of the ring. The carbons are typically shown as vertices where lines meet.
Your task, should you choose to accept it (and you kind of have to if you want to understand this stuff!), is to find these rings and then identify the carbons based on their attachments.

Look for the Base: Find a nitrogenous base (a complex ring structure, usually colored differently from the sugar). The carbon atom that’s directly bonded to one of the nitrogen atoms in that base is your 1’ carbon. This is often the most straightforward to spot.
Follow the Linkages: * To find the 5’ carbon, look for the carbon atom that has a phosphate group attached to it. This phosphate group will then be shown connecting to the 3’ carbon of the previous sugar (if you were looking at a double strand or a longer strand segment, but in ssDNA it’s the attachment point for the next one). In a single strand, you’ll often see the phosphate hanging off the 5’ carbon, ready to connect to the next nucleotide.
* The 3’ carbon will be the one with a hydroxyl group (-OH) attached, which is then ready to form a bond with the phosphate of the incoming nucleotide. This is how the strand grows in the 5’ to 3’ direction. So, it’s the anchor point for the next step in the chain.* The 2’ and 4’ carbons are within the ring itself, forming the structure. The 2’ carbon will have a hydrogen atom (sometimes not explicitly drawn if it’s implied) where the oxygen would be in ribose. The 4’ carbon is the one that bridges the ring and is connected to the 5’ carbon and the oxygen in the ring.
It can be a bit like a scavenger hunt, can’t it? “Okay, where’s the base? Found it! Which carbon is attached to it? That’s the 1’!” Then you trace the chain. “Where’s the phosphate? On this carbon! That must be the 5’!” And so on.
Why Does This Even Matter? (Besides the Obvious!)
So, you might be thinking, “Why all this fuss over a few carbon atoms?” Well, my friend, these sugar carbons are the architects of the DNA structure and function. Their precise arrangement dictates how the bases can pair up, how the strand folds, and how enzymes interact with the DNA. It’s all about the geometry, and the geometry is determined by these atoms and their bonds.
Understanding these carbons is fundamental for grasping concepts like:
- Replication: When DNA copies itself, enzymes need to know exactly where to attach and read the sequence. The 3’ and 5’ carbons are crucial for the directionality of this process.
- Transcription: Making RNA from DNA also relies on the specific orientation provided by the sugar-phosphate backbone.
- Mutations: Changes in the DNA sequence can arise from errors in replication or damage to the DNA. Knowing the structure, including the sugar carbons, helps us understand how these changes occur and their consequences.
- Drug Design: Many medicines, particularly antivirals and chemotherapeutics, work by interfering with DNA or RNA synthesis. These drugs often target the sugar moiety or the linkages involving the sugar carbons. So, understanding these carbons can literally lead to life-saving treatments. Pretty impactful for a few little atoms, right?
It’s like understanding the alphabet to read a book. You can’t just look at the pretty pictures; you need to know what each letter represents and how they fit together to form words and sentences. The sugar carbons are the letters and grammar of the DNA language.
So, when you’re next faced with a diagram of ssDNA (or even dsDNA, for that matter), don’t just see a jumble of lines and shapes. See the elegant architecture. See the fundamental building blocks. And specifically, look for those five-membered rings, trace the attachments, and identify those crucial 1’, 2’, 3’, 4’, and 5’ carbon atoms. They are the silent, yet powerful, engineers of life itself. And hey, if you get stuck, just remember the cookie analogy… maybe that’ll help you remember the sugar!
