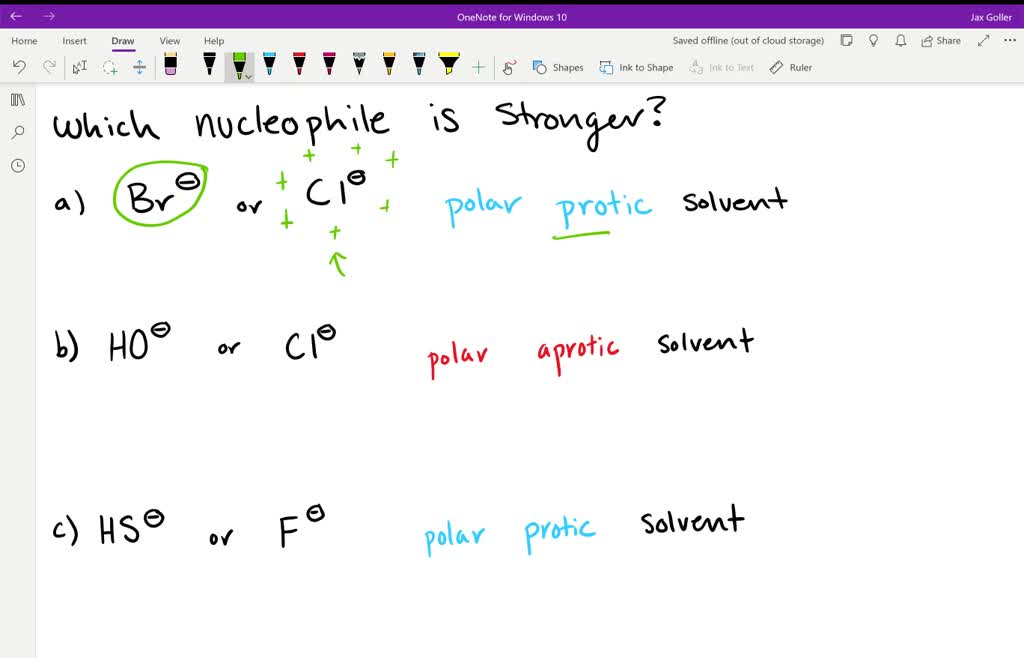
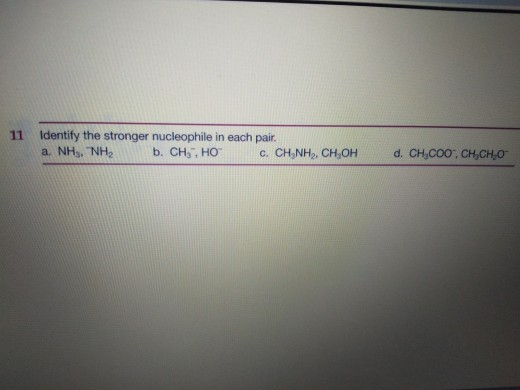
Identify The Stronger Nucleophile In The Following Pair.
Hey there, life explorers! Ever feel like you’re navigating a world of… well, stuff? From choosing the perfect avocado (a true art form, wouldn't you agree?) to understanding the subtle differences in your oat milk options, life is a constant stream of decisions. Today, we're diving into a topic that might sound a little technical, but trust me, it’s got a surprisingly chill vibe and some neat parallels to our everyday choices. We’re talking about identifying the stronger nucleophile in a pair.
Now, before you picture mad scientists in lab coats, let's reframe this. Think of nucleophiles as the “givers” or the “lovers” in the chemical world. They’re all about sharing their electron goodness with something that’s a little electron-deficient, kind of like a friendly stranger offering to help you carry your groceries when your arms are full. It’s all about attraction, connection, and making things happen!
The Heart of the Matter: What Exactly IS a Nucleophile?
So, a nucleophile, or "nucleus-loving" species, is basically an atom or a molecule that has a surplus of electrons. They’re the generous ones, always ready to donate a pair of electrons to an electron-poor center (we call that an electrophile – think of them as the "takers"). This electron-sharing dance is fundamental to how molecules interact and form new substances, much like how different personalities come together to create vibrant social circles.
Imagine it like this: you’re at a party, and there are a few people looking a bit shy or isolated (the electrophiles). A nucleophile is that outgoing, friendly individual who notices them and initiates a conversation, sharing their positive energy and making everyone feel more connected. It’s that spark that gets things moving.
Why Should We Care About "Stronger" Nucleophiles?
Just like in human interactions, some "givers" are more enthusiastic and effective than others. Some are like a gentle nudge, while others are a full-on, enthusiastic embrace. In chemistry, a stronger nucleophile is simply more eager and able to donate its electron pair. This means it will react faster and more readily. Understanding this helps chemists predict how reactions will proceed, much like you might instinctively know who’s more likely to organize the next weekend getaway.
Think about it in terms of dating apps. Some profiles are just more appealing, right? They have that extra something that draws you in. Similarly, some nucleophiles have chemical characteristics that make them more attractive to electron-deficient centers.
Decoding the Nucleophile Strength Spectrum: The Key Players
So, how do we figure out which nucleophile is the rockstar of electron donation? It boils down to a few key factors that influence their electron cloud and their willingness to share. Let's break them down, easy peasy.
1. The "Atom" Factor: Who's Holding the Goods?
This is perhaps the most straightforward. We often compare nucleophiles based on the atom that's actually doing the donating. Generally, as you move down a group on the periodic table, nucleophilicity increases. Why? Because the atoms get larger, and their outermost electrons are further from the nucleus, making them easier to donate.
Let’s take a common example: the halide ions – F⁻, Cl⁻, Br⁻, I⁻. Fluoride (F⁻) is the smallest and its electrons are held quite tightly. Iodide (I⁻), on the other hand, is huge! Its electrons are way out there, practically begging to be shared. So, in this group, iodide (I⁻) is the stronger nucleophile compared to fluoride (F⁻).
Think of it like this: you have a bunch of friends offering to help you move. The friend with the really small, nimble hands (like fluoride) might be great for picking up tiny screws, but the friend with the larger, more spacious hands (like iodide) might be better at grabbing a whole stack of books in one go. The latter is more efficient for a big task!
Fun Fact: The periodic table is organized by atomic number, but the arrangement of elements into groups (columns) and periods (rows) reveals so much about their behavior. It’s like a cosmic cheat sheet for chemists!
2. The "Charge" Factor: Who's Feeling Generous?
This one's pretty intuitive. Negatively charged species are generally stronger nucleophiles than their neutral counterparts, assuming they're from the same row or have similar atoms involved.
Why? Because a negative charge means there's an excess of electrons already. It’s like someone already feeling super generous and having extra cookies to hand out. A neutral molecule might have electrons, but it's not actively "looking" to get rid of them in the same way.
Consider water (H₂O) versus the hydroxide ion (OH⁻). The hydroxide ion has a negative charge, making it a significantly stronger nucleophile than water. It’s already got that electron surplus ready to go!
Cultural Ref: Think of it as the difference between someone who might lend you a dollar versus someone who happily hands you five. The latter is clearly the more generous spirit.
3. The "Resonance" Factor: Spreading the Love
Ah, resonance. This is where things get a little more artistic. Resonance occurs when a molecule's electrons aren't confined to a single bond or atom but are instead delocalized, meaning they're spread out over several atoms.

When a nucleophile can spread its electron density through resonance, it tends to be a weaker nucleophile. Why? Because the electron pair isn't as concentrated and therefore not as readily available for donation.
Imagine you have a small, concentrated pile of gold coins (a strong nucleophile). Now imagine those same coins are spread thinly across a large treasure map (a resonance-stabilized nucleophile). It’s harder to grab a good handful of coins from the map, right? The concentration matters!
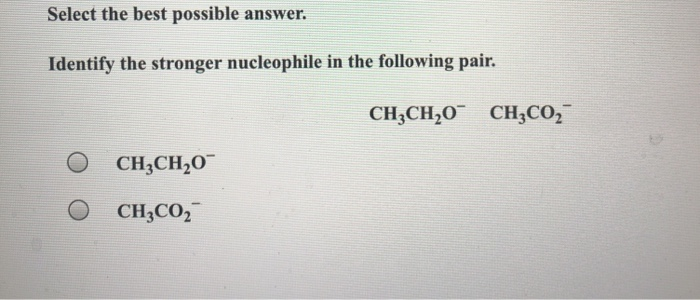
A classic example is comparing an alkoxide ion (like CH₃O⁻) with a carboxylate ion (like CH₃COO⁻). The negative charge in the alkoxide is localized on the oxygen. In the carboxylate, the negative charge is spread over two oxygen atoms through resonance. Therefore, the alkoxide ion is a stronger nucleophile than the carboxylate ion.
Practical Tip: When you see a negatively charged species, check if it can be stabilized by resonance. If it can, it's likely to be a weaker nucleophile than a similarly charged species where the charge is localized.
4. The "Steric Hindrance" Factor: Getting in the Way
This is where the "easy-going" part really comes into play. Steric hindrance refers to the spatial bulkiness of a molecule. If a nucleophile is too large and has a lot of "stuff" around the atom that's supposed to be donating electrons, it can have a harder time getting close to its target (the electrophile).
Think of trying to hug someone when you're both wearing giant, puffy winter coats. It's just… awkward and less effective than a hug between two people in t-shirts. The "coats" (bulky groups) get in the way.
So, between two nucleophiles with similar electronic properties, the one with less steric hindrance will generally be the stronger nucleophile because it can more easily approach and interact with the electrophile.

For instance, compare the tert-butoxide ion (a bulky, branched structure) with the methoxide ion (a small, simple structure). Even though both are alkoxides with a negative charge, the methoxide ion is a stronger nucleophile because it's much smaller and less sterically hindered.
Chill Fact: The concept of steric hindrance is also super relevant in everyday life! Think about trying to park a large SUV in a tight parallel parking spot versus a compact car. The smaller car has less "steric hindrance" and can maneuver more easily.
Putting It All Together: A Little Practice Makes Perfect
Let’s test your newfound knowledge with a quick scenario. Imagine we have a pair:
Based on what we’ve discussed:
- Charge: OH⁻ is negatively charged, H₂O is neutral.
The negative charge on OH⁻ makes it much more eager to share electrons. Therefore, hydroxide ion (OH⁻) is the stronger nucleophile.
Another one:
Here, we look at the atom:
- Atom: Both are halide ions, but iodine (I) is below fluorine (F) on the periodic table.
Iodine atoms are larger, and their electrons are less tightly held. Thus, iodide ion (I⁻) is the stronger nucleophile.
How about
Both are negatively charged alkoxides, so we consider other factors:
- Steric Hindrance: Methoxide is small and unhindered. Tert-butoxide is quite bulky due to the three methyl groups.
The lack of steric hindrance in methoxide makes it more accessible. Therefore, methoxide ion (CH₃O⁻) is the stronger nucleophile.
Beyond the Lab: The "Nucleophile" in Our Lives
So, what’s the takeaway from all this electron-sharing chatter? It’s a reminder that strength often comes from willingness and accessibility. The most impactful people, the most cherished friendships, the most fulfilling experiences – they often come from those who are most open, most generous, and most able to connect.
We all encounter situations where we can be the "nucleophile" – offering support, lending an ear, sharing our knowledge, or simply making someone smile. And just like in chemistry, our ability to do so effectively depends on our own "electronic structure," our willingness, and our lack of unnecessary "hindrance" that might prevent us from making a positive connection.
Next time you're faced with a decision, a conversation, or an opportunity to help, remember the humble nucleophile. Be the one with the available electron cloud, ready to make a positive impact. It's a simple concept, but its power to transform interactions, in the lab and in life, is truly profound. Keep sharing that good energy!
