Identify The Solute With The Highest Van't Hoff Factor.
Alright, gather ‘round, you lovely caffeine fiends and existential ponderers! We’re diving headfirst into the wild, wacky world of chemistry today, but fear not, this ain't your high school lab report. We’re talking about something called the Van’t Hoff factor. Sounds fancy, right? Like a brand of artisanal beard oil or a secret handshake for particle physicists. But stick with me, because this little number can tell us some seriously cool stuff about… well, about how things dissolve!
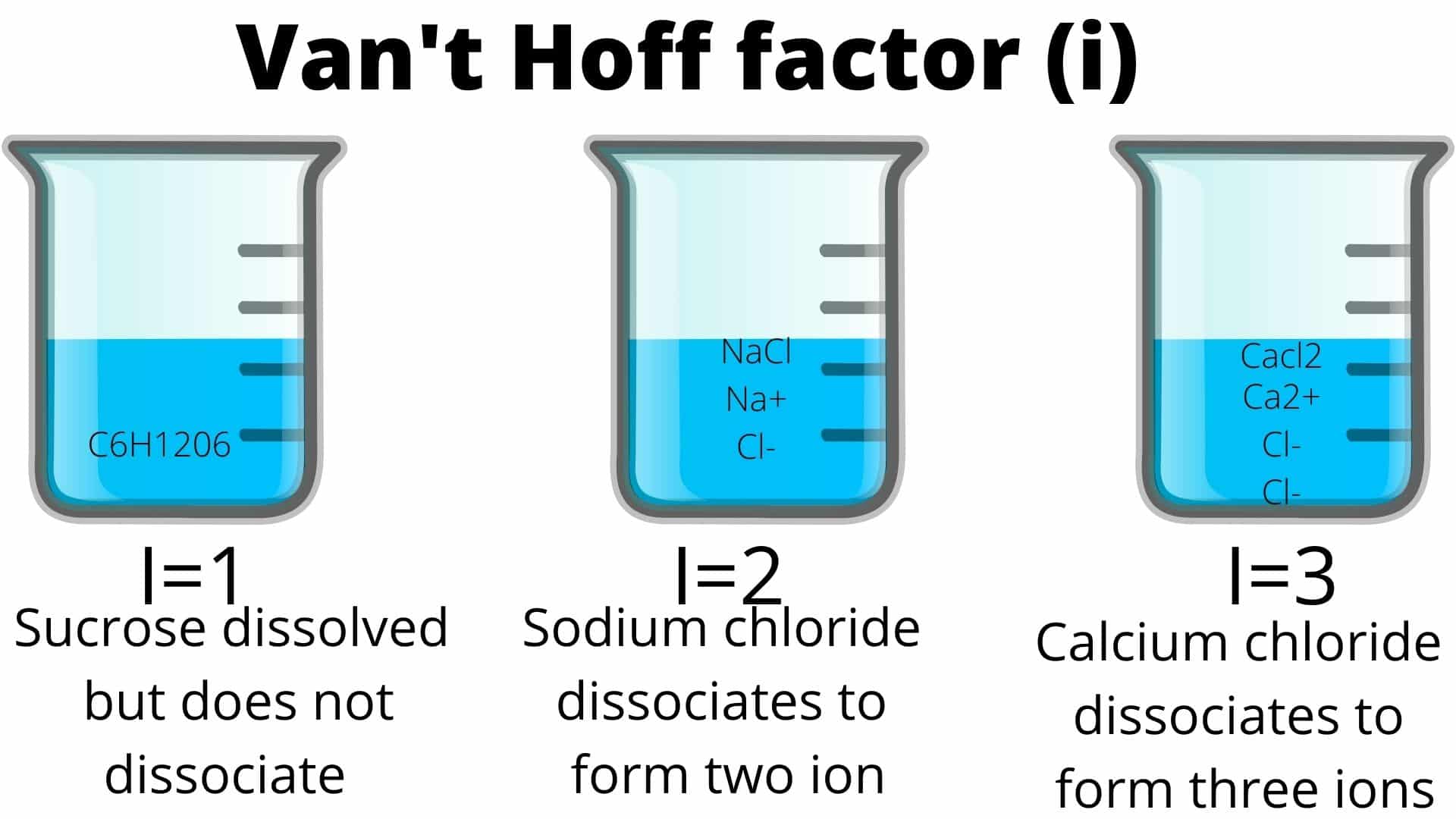
Imagine you’ve got a glass of water. Innocent enough. Now, you start tossing things in. Sugar? Salt? Maybe a rogue gummy bear (don’t judge)? What happens is that these little guys break apart, or at least try to, when they hit the H2O. The Van’t Hoff factor, this magical ‘i’ number, is basically our chemist’s scorecard for how many pieces of our original stuff we end up with floating around. Think of it like this: if you throw a whole LEGO brick into water, it’s still one brick, right? Van’t Hoff factor of 1. Easy peasy.
But then you get to the real party animals of the dissolving world. We’re talking about ionic compounds. These are the salts, the acids, the bases – the ones that are basically built like tiny, charged magnets. When you chuck them into water, bam! They don't just chill; they dissociate. They split up into their individual charged bits, called ions. And this is where the Van’t Hoff factor starts to get really interesting, and frankly, a bit mischievous.
The Sugar vs. Salt Showdown: A Classic Comedy of Errors
Let’s start with our old friend, sugar. When you stir sugar into your tea, it’s like a polite guest at a party. It dissolves, sure, but the sugar molecules mostly stay intact. They’re like those introverts who just nod politely from the corner. So, for sugar, the Van’t Hoff factor is pretty much 1. It doesn't really break into smaller pieces.
Now, introduce table salt, or sodium chloride (NaCl). This guy is the life of the party, but in a slightly more… dramatic way. When NaCl hits water, it’s like a flash mob of tiny, charged dancers! The sodium ions (Na+) and chloride ions (Cl-) go their separate ways. So, one unit of NaCl becomes two separate entities floating around. Ding ding ding! Our Van’t Hoff factor for salt is approximately 2.
This is why your food tastes saltier when you add salt than it does when you add the same amount of sugar. More “things” are hitting your taste buds, even if they started out as the same weight!
Enter The Big Guns: The Multi-Part Wonders
But wait, there’s more! The world of ionic compounds is like a family reunion with a lot of cousins. Some molecules are just built to split into more pieces than a toddler with a pack of crayons. We’re talking about compounds that are made of multiple types of ions, and each of those ions is often a little cluster of atoms itself. It’s like a nesting doll, but with electrical charges!
Let’s consider something like calcium chloride (CaCl2). When this stuff hits water, it doesn’t just split into two. Oh no. The calcium ion (Ca2+) is one piece, and then you’ve got two chloride ions (Cl-) tagging along. So, one CaCl2 unit turns into 1 calcium ion + 2 chloride ions = 3 individual pieces! Its Van’t Hoff factor is around 3. It’s like a really enthusiastic magician making things disappear… and then reappear as more things!
This is where things start to get really fun, and by fun, I mean incredibly useful for things like de-icing roads. That’s right, the stuff they spray on the roads in winter? Often contains things like calcium chloride precisely because it lowers the freezing point of water more than just salt does, thanks to its higher Van’t Hoff factor. More pieces, more freezing point depression. Science!
The Ultimate Dissociation Champion: Hunting for the Highest ‘i’
So, the million-dollar question, the riddle wrapped in an enigma, the reason you might be losing sleep at 3 AM (or not, that’s okay too): which solute has the highest Van’t Hoff factor? We’re looking for a molecule that goes absolutely bonkers in water, breaking into the maximum number of charged pieces. We’re talking about the ultimate dissociator, the king of splitting!
To get a really high Van’t Hoff factor, we need a compound that, when it dissolves, yields a large number of ions. This usually means a compound with multiple cations and multiple anions, and ideally, those anions are also polyatomic ions (meaning they are groups of atoms that act as a single charged unit).
Let’s throw in some more complex examples. Take sodium sulfate (Na2SO4). When this dissolves, you get two sodium ions (Na+) and one sulfate ion (SO4^2-). That’s 2 + 1 = 3 pieces. So, its Van’t Hoff factor is around 3.

But what about something like aluminum sulfate (Al2(SO4)3)? This is where we start playing in the big leagues. When Al2(SO4)3 dissolves, you get two aluminum ions (Al^3+) and three sulfate ions (SO4^2-). That’s 2 + 3 = 5 pieces! Its theoretical Van’t Hoff factor is around 5. Now we’re talking! This is like a chemical rave, with multiple dancers splitting off in every direction.
The Theoretical vs. The Real World: Where Chemistry Gets Wiggly
Now, here’s a little secret from the chemistry underground: the Van’t Hoff factor we calculate theoretically (like our 5 for aluminum sulfate) isn’t always the exact number we see in real life. Why? Because those tiny charged ions, when they’re all floating around and bumping into each other, can get a bit… clingy. They don’t always behave as perfectly separate entities. They can form little clusters, or interact with the water molecules in complex ways. It’s like at that party – some people make new friends, others just find someone to stand with.
This means the actual Van’t Hoff factor is often a little lower than the theoretical one, especially at higher concentrations. It’s like the party gets so crowded that people start to feel a bit overwhelmed and stick closer together. This is often represented by a slightly different symbol, or by acknowledging that the factor is an approximation.

So, Who’s the Undisputed Champion?
While compounds like aluminum sulfate, with a theoretical Van’t Hoff factor of 5, are definitely up there, the real question for the absolute highest often leads us to some pretty exotic or specially designed molecules. However, for commonly encountered substances that we might actually use, compounds that break into four or five ions are generally considered to have very high Van’t Hoff factors.
Think about complex salts like potassium ferricyanide (K3[Fe(CN)6]). This bad boy can dissociate into 3 potassium ions (K+) and the complex ferricyanide ion ([Fe(CN)6]^3-). That’s 3 + 1 = 4 pieces. Pretty good!
But if we're looking for something truly spectacular, we might need to consider substances that are designed to be highly dissociative, or those that have very large, complex polyatomic ions that themselves might have some internal structure. However, sticking to more common solutes, salts that yield a total of 4 or 5 ions upon dissociation, like aluminum sulfate (Al2(SO4)3) or perhaps some of the more complex sulfates or phosphates, are usually the contenders for the highest Van’t Hoff factors you'll encounter in general chemistry.
So, the next time you’re stirring something into your drink, just remember the invisible dance party happening in your cup. Some molecules are shy wallflowers, others are flamenco dancers, and a few are… well, they’re the ones you want to watch out for if you’re trying to freeze something or, you know, de-ice a highway. Chemistry, folks. It’s a weird and wonderful world!
